Models of Interstellar Chemistry The TugofWar Between Astronomical

Models of Interstellar Chemistry: The Tug-of-War Between Astronomical and Chemical Complexity ERIC HERBST DEPARTMENTS OF PHYSICS, CHEMISTRY AND ASTRONOMY THE OHIO STATE UNIVERSITY

Some Collaborators • Former postdocs: Y. Aikawa, H. Cuppen, R. Garrod, V. Wakelam • Students: Q. Chang, D. Quan • And also: T. Millar, P. Caselli, D. Semenov
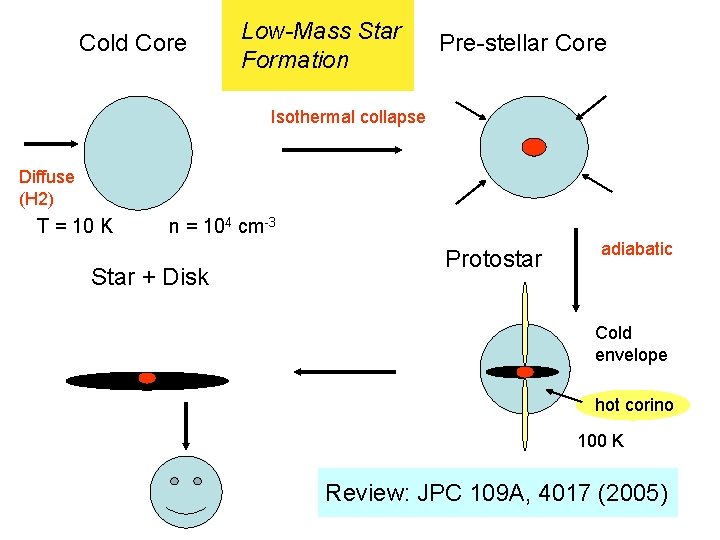
Cold Core Low-Mass Star Formation Pre-stellar Core Isothermal collapse Diffuse (H 2) T = 10 K n = 104 cm-3 Star + Disk Protostar adiabatic Cold envelope hot corino 100 K Review: JPC 109 A, 4017 (2005)
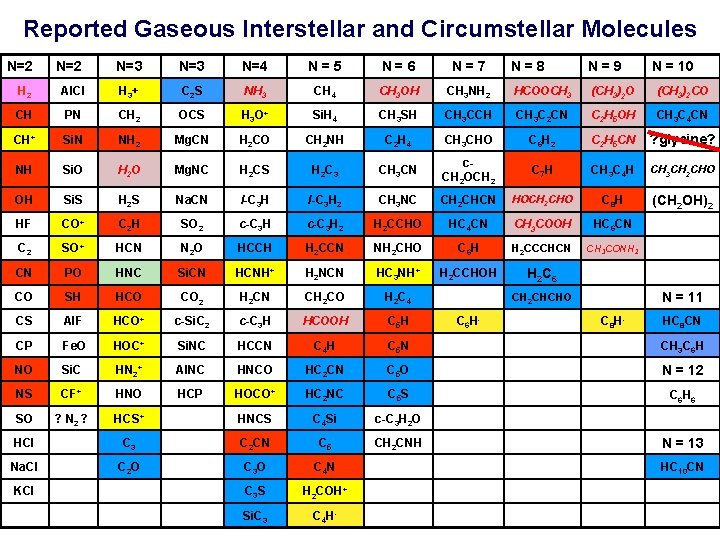
Reported Gaseous Interstellar and Circumstellar Molecules N=2 N=3 N=4 N = 5 N = 6 N = 7 N = 8 N = 9 H 2 Al. Cl H 3+ C 2 S NH 3 CH 4 CH 3 OH CH PN CH 2 OCS H 3 O+ Si. H 4 CH+ Si. N NH 2 Mg. CN H 2 CO NH Si. O H 2 O Mg. NC OH Si. S H 2 S HF CO+ C 2 CH 3 NH 2 HCOOCH 3 (CH 3)2 O (CH 3)2 CO CH 3 SH CH 3 CCH CH 3 C 2 CN C 2 H 5 OH CH 3 C 4 CN CH 2 NH C 2 H 4 CH 3 CHO C 6 H 2 C 2 H 5 CN ? glycine? H 2 CS H 2 C 3 CH 3 CN c. CH 2 OCH 2 C 7 H CH 3 C 4 H CH 3 CH 2 CHO Na. CN l-C 3 H 2 CH 3 NC CH 2 CHCN HOCH 2 CHO C 8 H (CH 2 OH)2 C 2 H SO 2 c-C 3 H 2 H 2 CCHO HC 4 CN CH 3 COOH HC 6 CN SO+ HCN N 2 O HCCH H 2 CCN NH 2 CHO C 6 H H 2 CCCHCN CH 3 CONH 2 CN PO HNC Si. CN HCNH+ H 2 NCN HC 3 NH+ H 2 CCHOH H 2 C 6 CO SH HCO CO 2 H 2 CN CH 2 CO H 2 C 4 CS Al. F HCO+ c-Si. C 2 c-C 3 H HCOOH C 5 H CP Fe. O HOC+ Si. NC HCCN C 4 H C 5 N CH 3 C 6 H NO Si. C HN 2+ Al. NC HNCO HC 2 CN C 5 O N = 12 NS CF+ HNO HCP HOCO+ HC 2 NC C 5 S C 6 H 6 SO ? N 2 ? HCS+ HNCS C 4 Si c-C 3 H 2 O HCl C 3 C 2 CN C 5 CH 2 CNH Na. Cl C 2 O C 3 O C 4 N KCl C 3 S H 2 COH+ Si. C 3 C 4 H- N = 11 CH 2 CHCHO C 6 H- N = 10 C 8 H- HC 8 CN N = 13 HC 10 CN

Ice Mantle Species in Cold Dense Clouds • • H 2 O CO 2 CO CH 3 OH • • • H 2 CO XCN HCOOH CH 4 NH 3 Water ice dominant Approx. 100 monolayers can develop on 0. 1 m grains

L 1527: continuum map

Highlights of Cold Phases • Ion-molecule gas-phase chemistry leading to unusual species including unsaturated (H-poor species) and deuterium fractionation. Networks include 4000+ reactions……. • Accretion tending to deplete heavy species from the gas, especially in centers of pre-stellar cores • Surface chemistry leading to ices, including water and methanol

Should we model surface chemistry? • PRO: obviously important, new experiments being undertaken, new theoretical approaches (Vidali et al. , Hornekaer et al. , Linnartz et al. , Mc. Coustra et al. , Fraser et al. , Watanabe et al. ) • CON: “last refuge of a scoundrel”; too complex to do anything but throw the stuff together in the lab (Tielens, Thaddeus) • “And all our yesterdays have lighted fools the way to dusty death” Macbeth

SURFACE CHEMISTRY BASED ON DIFFUSION & PHYSISORPTION

TYPES OF SURFACE REACTIONS: Low T REACTANTS: MAINLY MOBILE ATOMS AND RADICALS A + B AB association H + H H 2 H + X XH (X = O, C, N, CO, etc. ) WHICH CONVERTS O OH H 2 O C CH 2 CH 3 CH 4 N NH 2 NH 3 CO H 2 CO H 3 CO CH 3 OH C 2 CN CH 3 CH 2 CN ? ? ? ? Chemistry occurs mainly via Langmuir-Hinshelwood mechanism

Higher Temperatures (40 K) • Heavier species, such as atoms or radicals formed by photodissociation, can diffuse and react: • e. g. HCO + CH 3 O HCOOCH 3 • Garrod & Herbst (2006) Widicus-Weaver, Garrod, Herbst, in prep. , Charnley and co-workers Now thought to be important in hot cores and corinos…
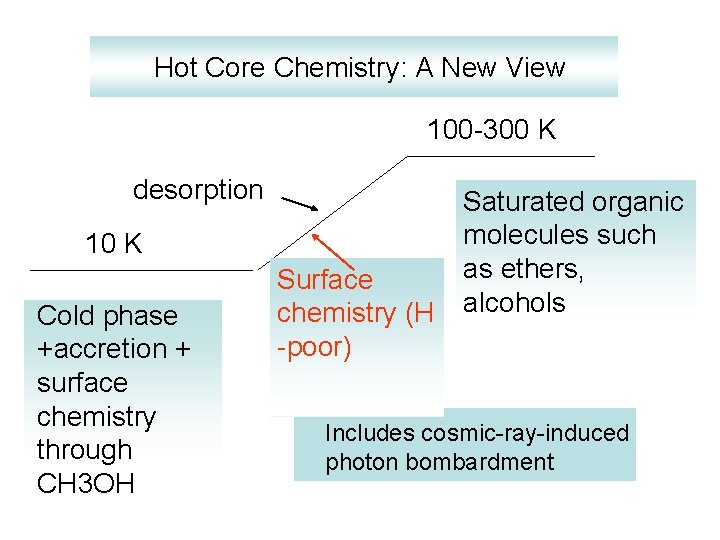
Hot Core Chemistry: A New View 100 -300 K desorption 10 K Cold phase +accretion + surface chemistry through CH 3 OH Surface chemistry (H -poor) Saturated organic molecules such as ethers, alcohols Includes cosmic-ray-induced photon bombardment

Standard Gas-Grain Model • Gas and grain chemistries coupled by accretion and desorption, both thermal and non-thermal, including reactive desorption (Garrod, Wakelam, Herbst 2007) • Diffusive (LH) mechanism treated by modified rate equations. Uses one set of appropriate energy parameters per adsorbate/surface pair. • One size dust particle utilized • For cold sources, reasonable agreement with both gas and ice; low values for CO 2 and CO ice typically. (Garrod et al. 2007) • For hot cores, inclusion of surface chemistry during heatup phase seems to improve agreement with observation for organic molecules (Garrod & Herbst 2006).
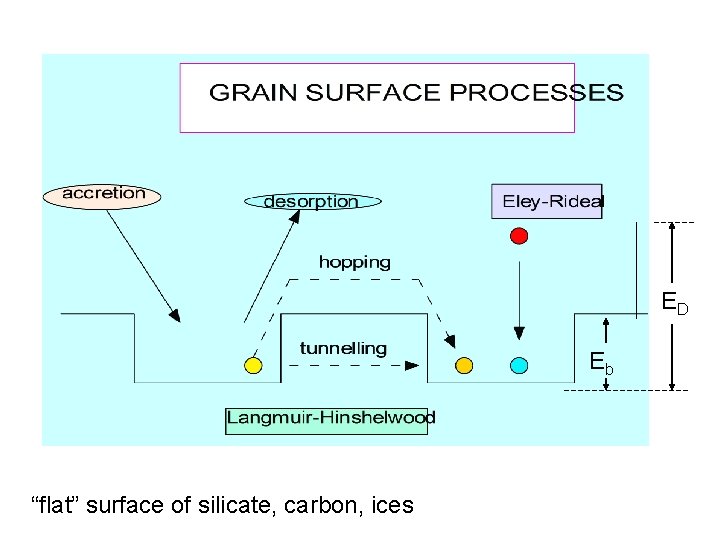
ED Eb “flat” surface of silicate, carbon, ices

Modelling Diffusive Surface Chemistry I Consider a species A that hops to the next site to find and react with a species B: Now consider a number of moving species A:

MODELLING DIFFUSIVE SURFACE CHEMISTRY II Rate Equations where Pickles & Williams 1977 Used in large gas-grain models.

Thermal Desorption & Diffusion for heavies at 10 K (hopping rate slowed down to agree with more detailed treatments…. . )

Desorption Mechanisms • Thermal, as temperature rises • Non-thermal at low temperature, includes reactive desorption, cosmic ray bombardment (inefficient) • Shock-wave sputtering, may be needed in galactic center • Photodesorption, pops up in models but seems to be very specific

Problems with Rate Equations • a) inaccurate treatment of random walk; problem known as “back diffusion” • b) overestimate of rate in “accretion limit” where average number of reactive particles less than unity and discreteness and fluctuations important (Tielens) • c) cannot easily take into account microscopic structure of surface (e. g. roughness, time dependence)

MACROSCOPIC STOCHASTIC METHODS Idea is to follow probabilities that a certain number of atoms/molecules of a species are present on a particular grain as a function of time. Directed at accretion limit problem. MONTE CARLO METHOD: use random numbers (Charnley) MASTER EQUATION METHOD: propagate probabilities forward via differential equations (Biham et al. ; Green et al. ); more easily coupled to gas-phase rate equations (Stantcheva et al. ); approximations still necessary.
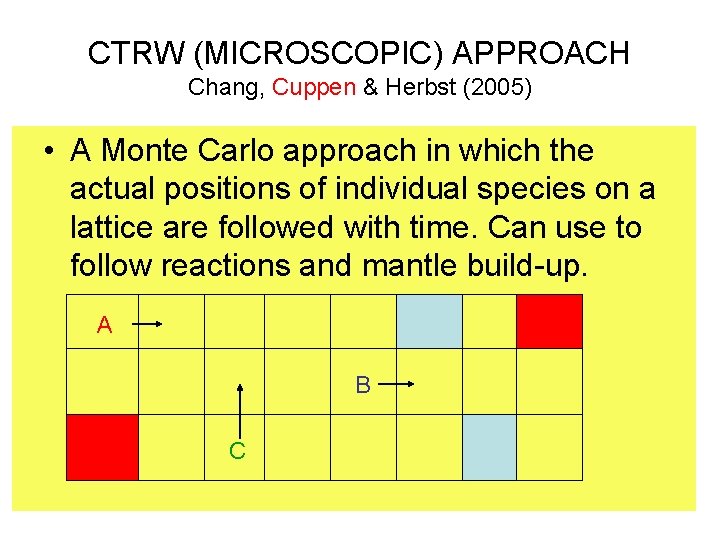
CTRW (MICROSCOPIC) APPROACH Chang, Cuppen & Herbst (2005) • A Monte Carlo approach in which the actual positions of individual species on a lattice are followed with time. Can use to follow reactions and mantle build-up. A B C

Complexity in one Form • Molecular Evolution and Star Formation: From Pre-stellar Cores to Protostellar Cores Aikawa, Wakelam, Garrod, Herbst: 1 -D hydrodynamics + standard gas-grain model • Gas-grain chemistry in cold interstellar cloud cores with a microscopic Monte Carlo approach to surface chemistry Chang, Cuppen, Herbst: gas-phase rate equations + microscopic Monte Carlo surface approach
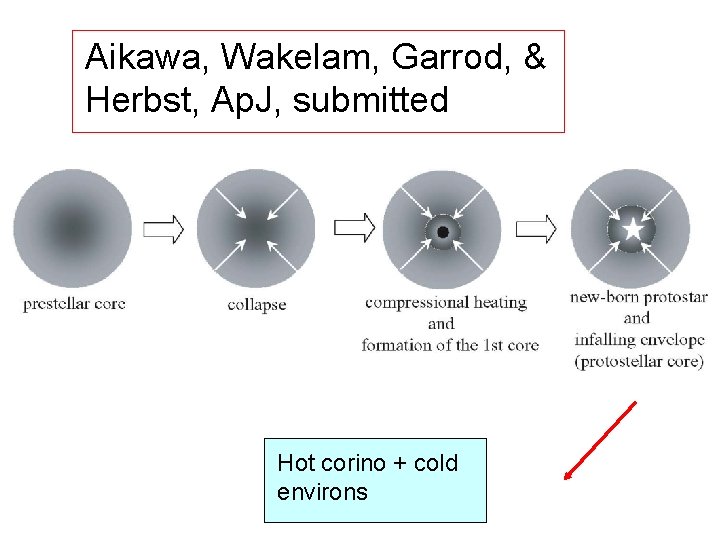
Aikawa, Wakelam, Garrod, & Herbst, Ap. J, submitted Hot corino + cold environs
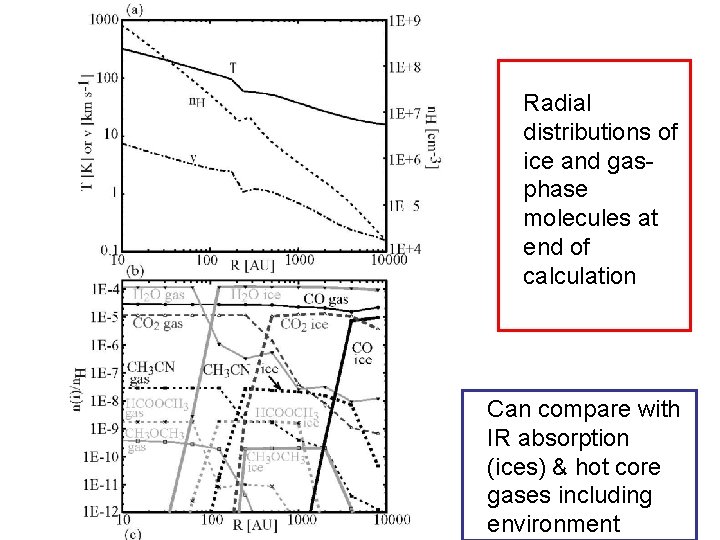
Radial distributions of ice and gasphase molecules at end of calculation Can compare with IR absorption (ices) & hot core gases including environment
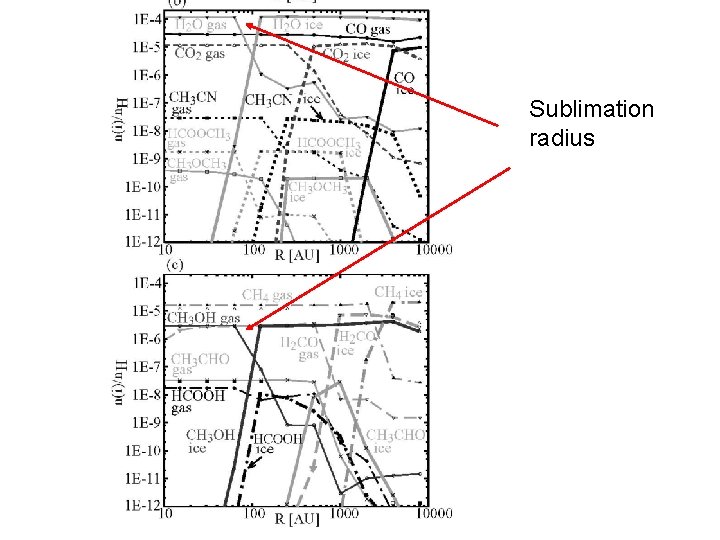
Sublimation radius

Stochastic Surface Model • Gas-phase proceeds via rate equations, while adsorption, desorption (of H), and surface reactions are handled stochastically with microscopic approach. Both LH and ER mechanisms included • So far, used for cold core only with thermal but non-thermal desorption. Chang, Cuppen, Herbst, Ap. J in press
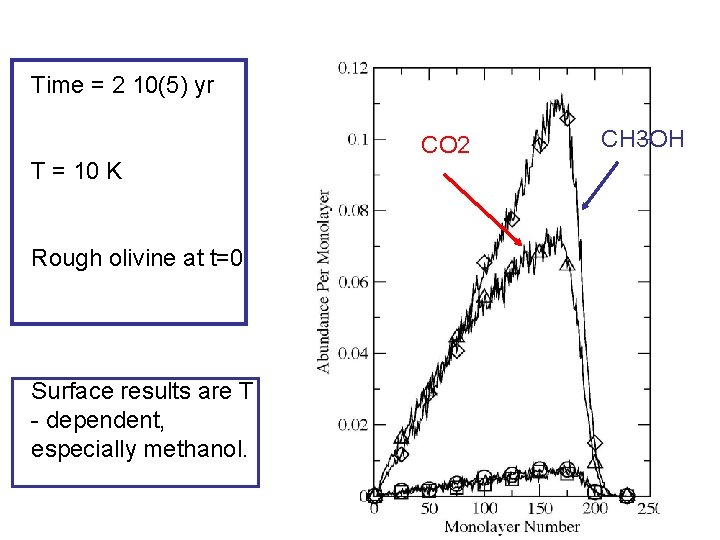
Time = 2 10(5) yr T = 10 K Rough olivine at t=0 Surface results are T - dependent, especially methanol. CO 2 CH 3 OH

TESTS OF COMPLEXITY • Both models can be compared with ices on grains surrounding a low-mass protostar. In the hydrodynamic case, the ices have gone through a thermal history whereas in the stochastic approach, the temperature and density are constant. Which one wins?

ELIAS 16; 29 ICES • • • Species Observed H 2 O 1. 3(-4) CO 2 18% (22%) CO 25% (6%) CH 3 OH <3 (<4%) Master equation model produces very little CO and CO 2. • Aikawa* Chang** • • ----- 2. 1(-4) 21 6. 2 23 2. 9 0. 04 3. 9 • *Isolated core at 8000 AU • ** rough silicate, 15 K

The Future • Better integration of gas-phase network and stochastic surface network (V. Shematovich) • Closer relation between lab work and modeling of surface reaction rates • Improvement in understanding of growth of ice mantles (Cuppen) • Inclusion of chemisorption • More detailed gas-grain models of hot cores, protoplanetary disks, shocked regions, PDR’s • Better appreciation of role of PAHs • More dynamics, heterogeneity, and radiative transfer in all models in the ALMA era
- Slides: 32