Models of Atomic Structure Dalton Model Unbreakable neutrally

Models of Atomic Structure

Dalton Model • Unbreakable neutrally charged spheres.
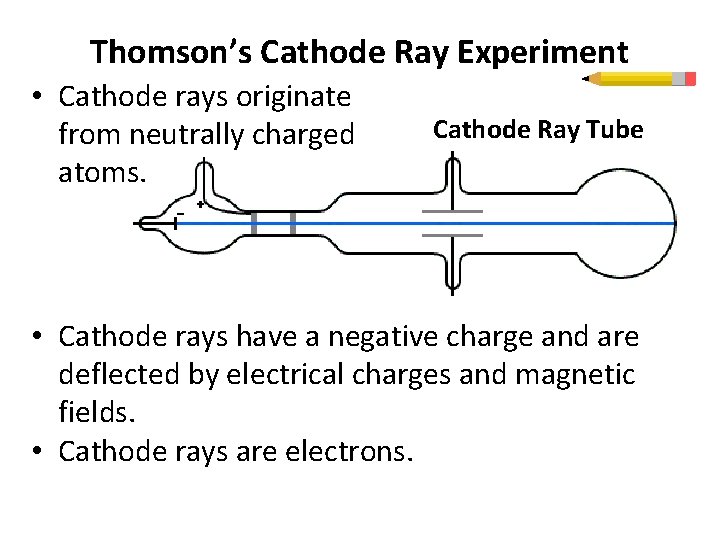
Thomson’s Cathode Ray Experiment • Cathode rays originate from neutrally charged atoms. Cathode Ray Tube • Cathode rays have a negative charge and are deflected by electrical charges and magnetic fields. • Cathode rays are electrons.
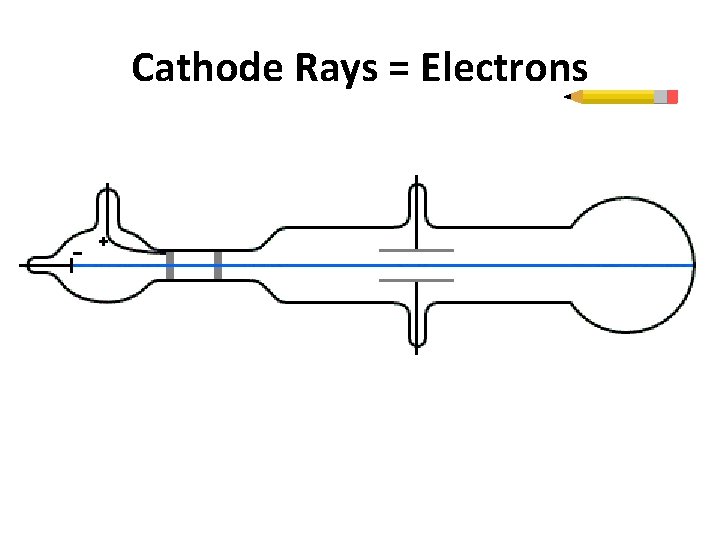
Cathode Rays = Electrons

Cathode Rays = Electrons

Cathode Rays = Electrons
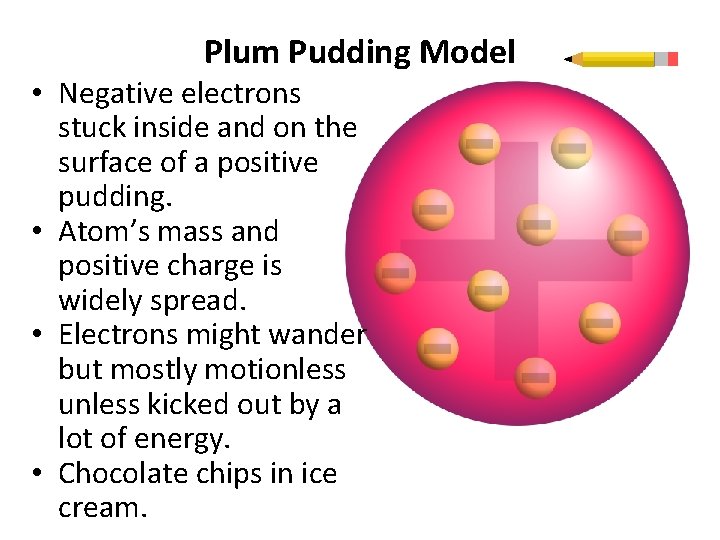
Plum Pudding Model • Negative electrons stuck inside and on the surface of a positive pudding. • Atom’s mass and positive charge is widely spread. • Electrons might wander but mostly motionless unless kicked out by a lot of energy. • Chocolate chips in ice cream.

Plum Pudding Model • Negative electrons stuck inside and on the surface of a positive pudding. • Atom’s mass and positive charge is widely spread. • Electrons might wander but mostly motionless unless kicked out by a lot of energy. • Chocolate chips in ice cream.
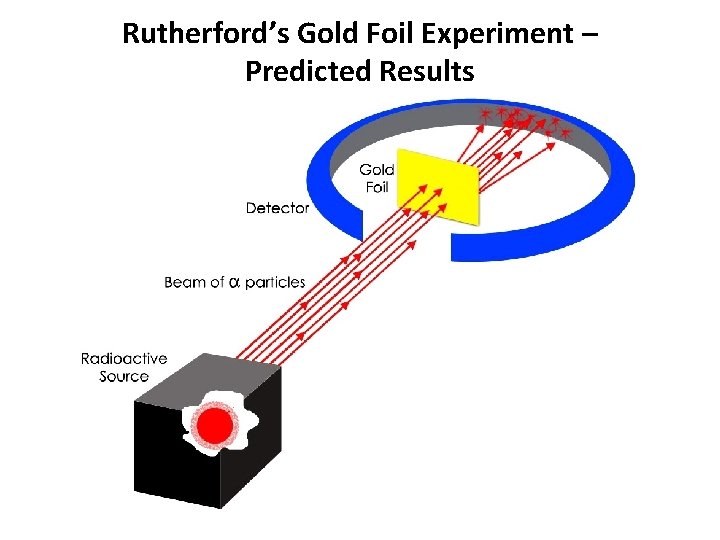
Rutherford’s Gold Foil Experiment – Predicted Results
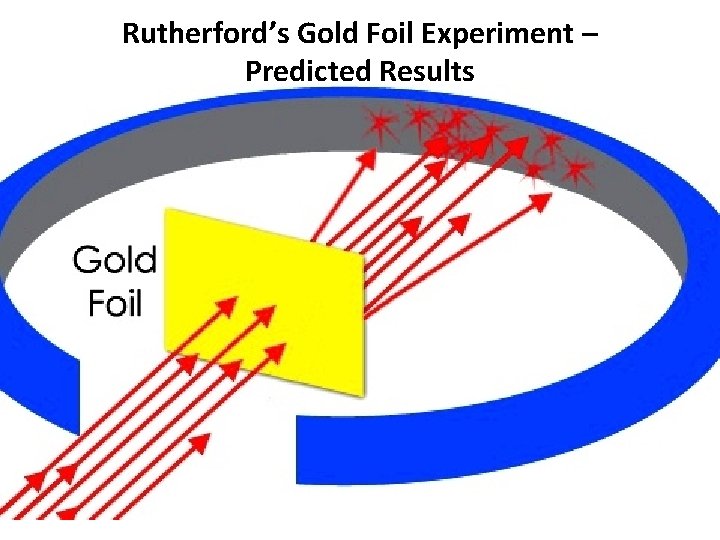
Rutherford’s Gold Foil Experiment – Predicted Results
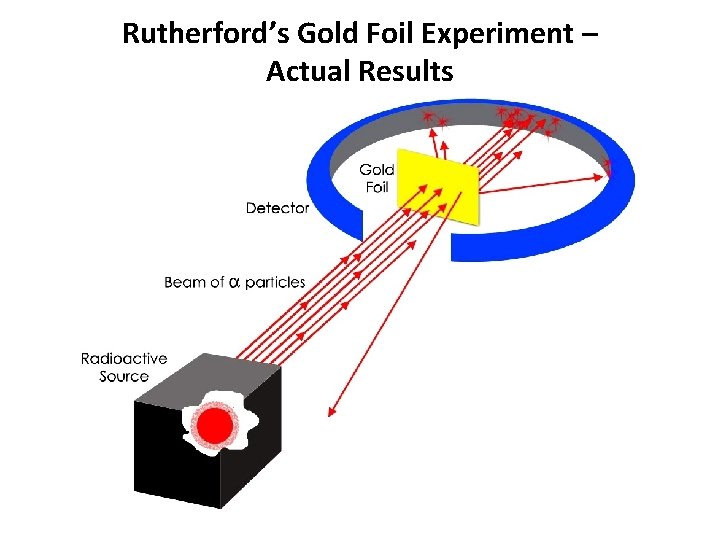
Rutherford’s Gold Foil Experiment – Actual Results
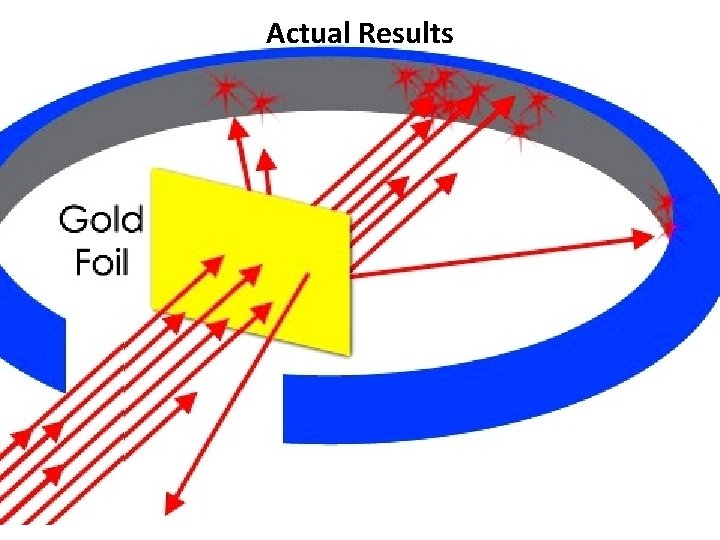
Actual Results
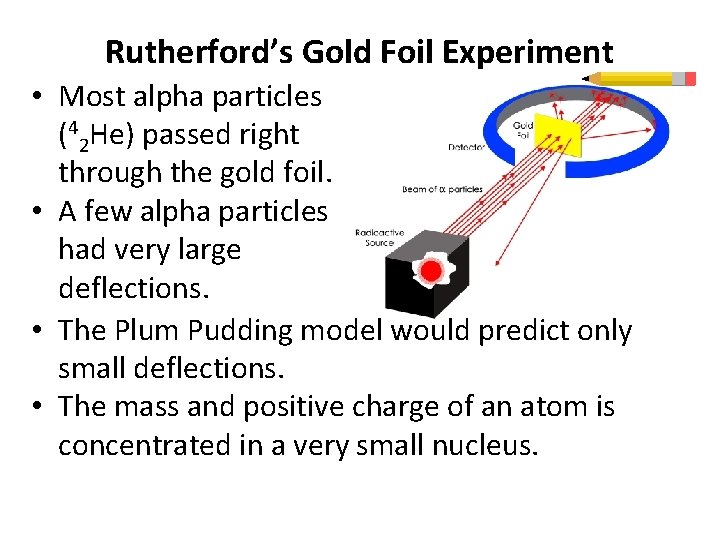
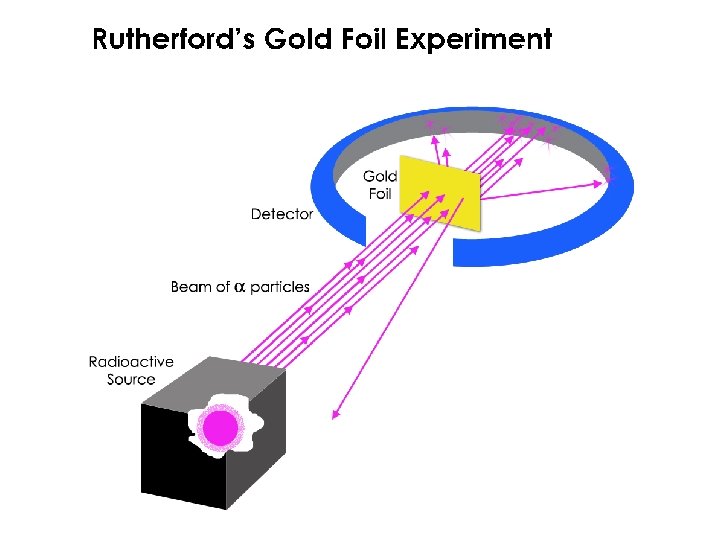
Rutherford’s Gold Foil Experiment • Most alpha particles (42 He) passed right through the gold foil. • A few alpha particles had very large deflections. • The Plum Pudding model would predict only small deflections. • The mass and positive charge of an atom is concentrated in a very small nucleus.
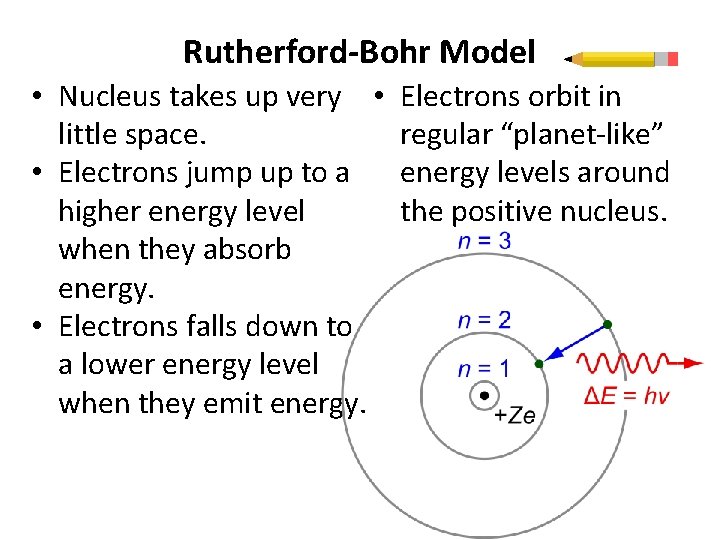
Rutherford-Bohr Model • Nucleus takes up very • little space. • Electrons jump up to a higher energy level when they absorb energy. • Electrons falls down to a lower energy level when they emit energy. Electrons orbit in regular “planet-like” energy levels around the positive nucleus.
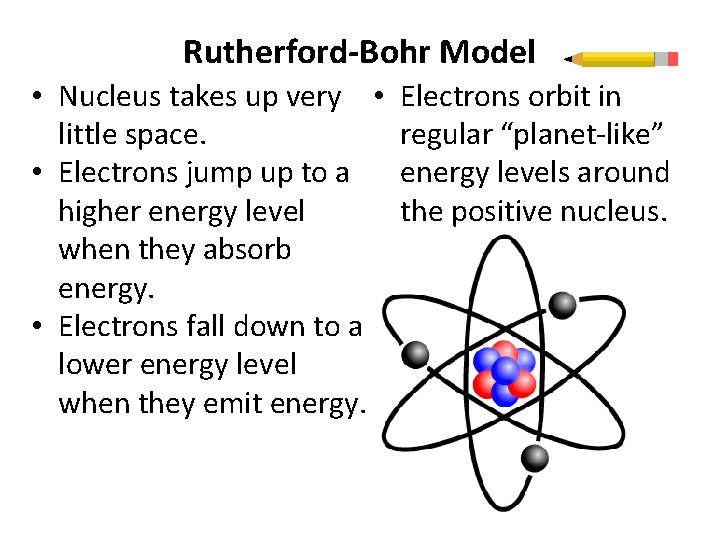
Rutherford-Bohr Model • Nucleus takes up very • little space. • Electrons jump up to a higher energy level when they absorb energy. • Electrons fall down to a lower energy level when they emit energy. Electrons orbit in regular “planet-like” energy levels around the positive nucleus.

Quantum Model • Electron clouds not orbits. • Electrons are not found in fixed locations, but rather probabilities to be in a location.

s-orbitals
p-orbitals
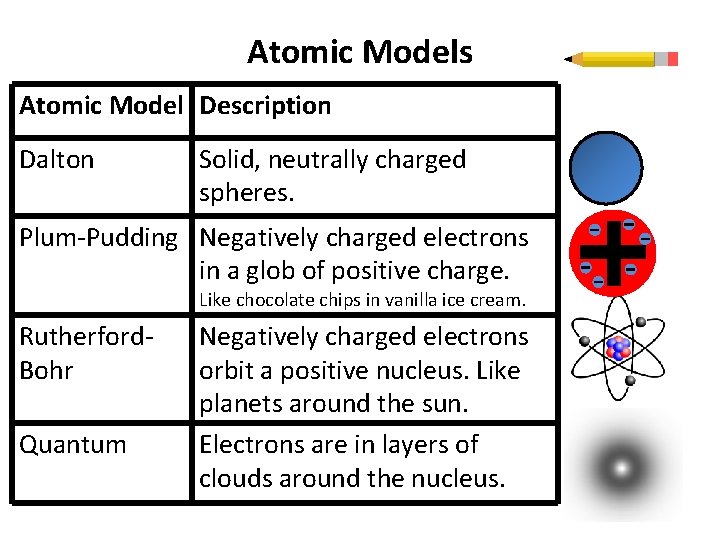
Atomic Models Atomic Model Description Dalton Solid, neutrally charged spheres. Plum-Pudding Negatively charged electrons in a glob of positive charge. Like chocolate chips in vanilla ice cream. Rutherford. Bohr Quantum Negatively charged electrons orbit a positive nucleus. Like planets around the sun. Electrons are in layers of clouds around the nucleus.
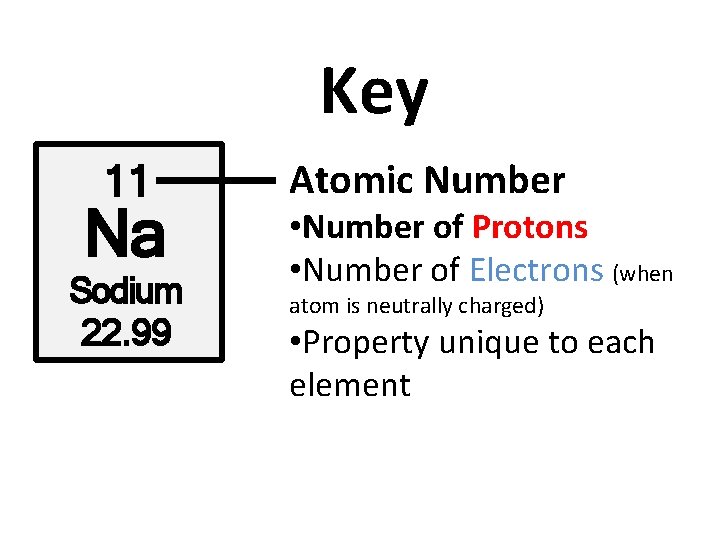
Key 11 Na Sodium 22. 99 Atomic Number • Number of Protons • Number of Electrons (when atom is neutrally charged) • Property unique to each element
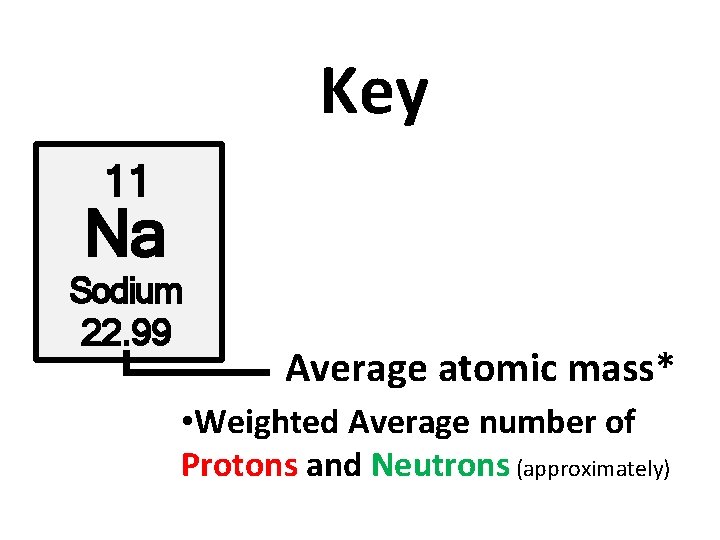
Key 11 Na Sodium 22. 99 Average atomic mass* • Weighted Average number of Protons and Neutrons (approximately)

Subatomic Particles the particles that make up an atom • Protons – high mass, positive charge. Found in nucleus. • Neutrons – high mass, no charge. Found in nucleus. • Electrons – low mass, negative charge. Found orbiting around nucleus. (abbreviated e– )
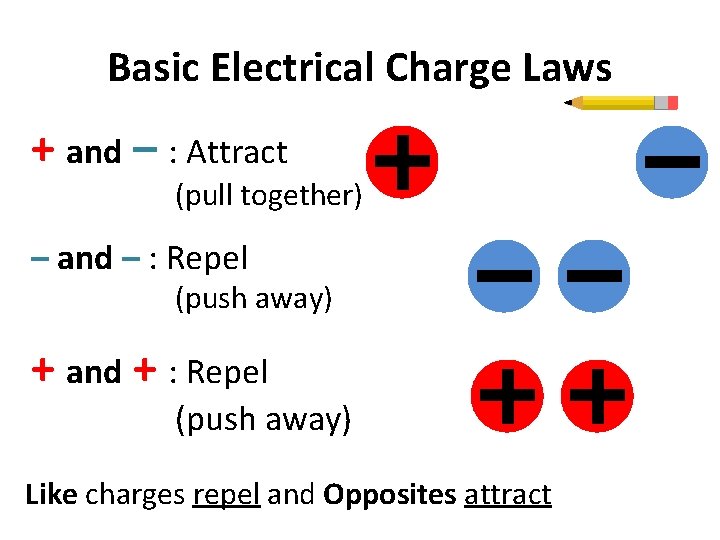
Basic Electrical Charge Laws + and – : Attract (pull together) – and – : Repel (push away) + and + : Repel (push away) Like charges repel and Opposites attract
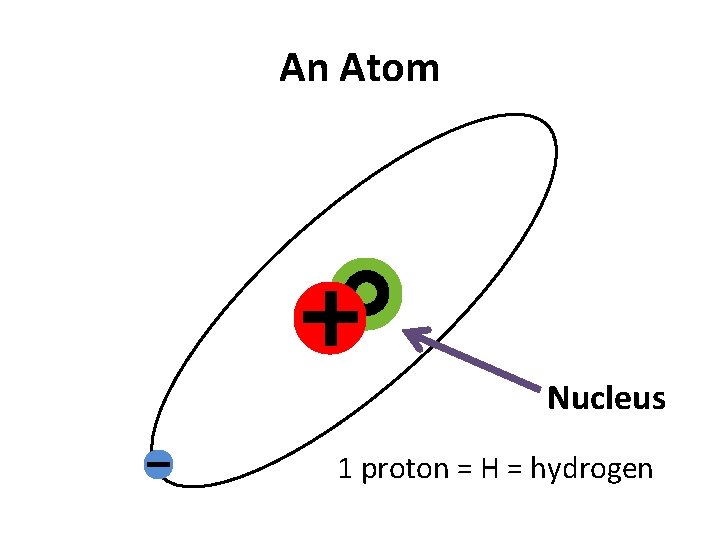
An Atom Nucleus 1 proton = H = hydrogen

Why doesn’t the electron fall into the nucleus? • It orbits because the electron is moving really fast around the nucleus. • Because the electron has such a low mass, even a small amount of energy makes it move very fast.
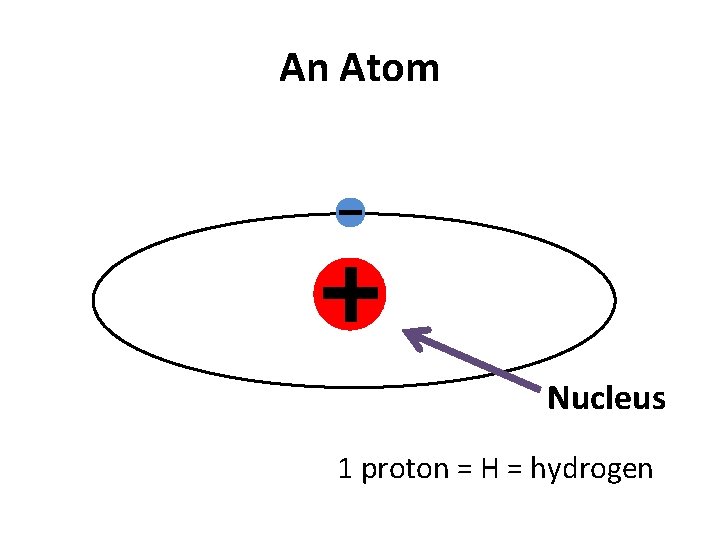
An Atom Nucleus 1 proton = H = hydrogen
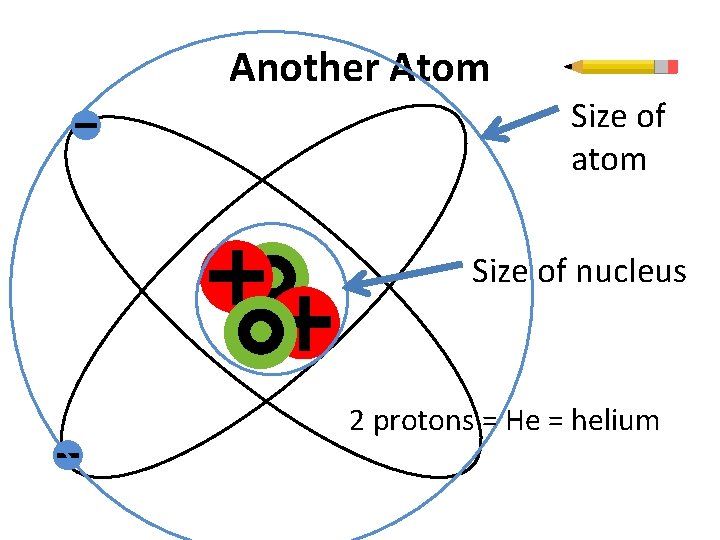
Another Atom Size of atom Size of nucleus 2 protons = He = helium
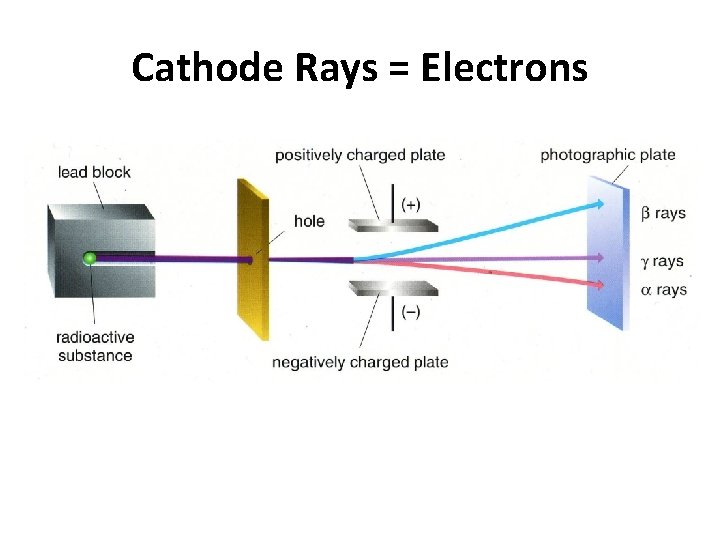
Cathode Rays = Electrons
H He Li Be B C N O F Na Mg Al Si P S Cl Ar K Ca Ne Br Kr I Xe
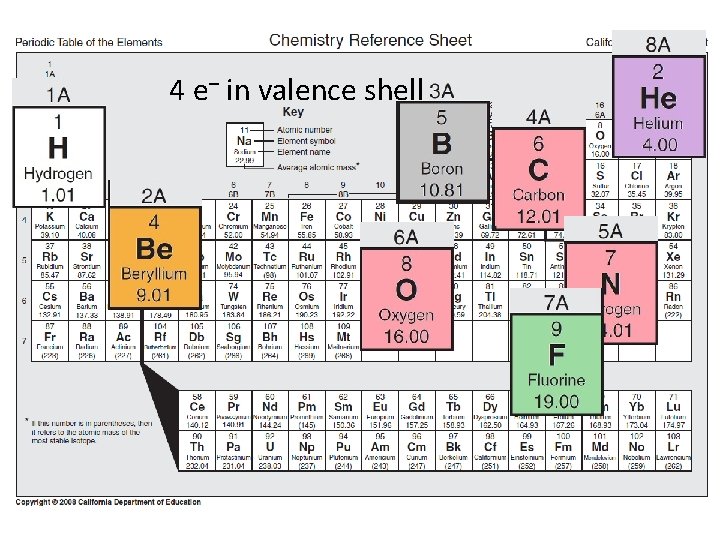
4 e– in valence shell
- Slides: 32