Models of an Atom John Dalton 1766 1844












- Slides: 17

Models of an Atom

John Dalton (1766 – 1844) Experiment/Observations: • He studied partial pressures of the atmosphere • He noticed that the ratios in which elements combine remain in fixed, simple proportions.

Theory All matter is made up of individual particles called atoms, which cannot be divided: The four postulates: • All elements are made of atoms • All atoms of the same element have the same mass, and atoms of different elements have different masses • Compounds contain atoms of more than one element • In a particular compound, atoms of different elements always combine in the same way

Billiard Ball Model

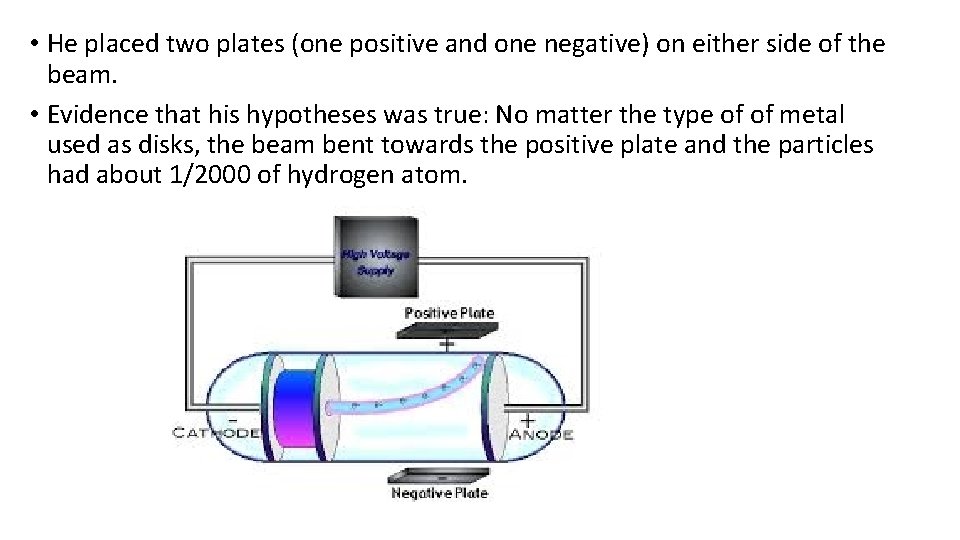
Thomson’s (1856 – 1940) Model of the Atom • Experiment/Observations: • Used a cathode ray tube – a sealed glass tube with metal disks at each end. Wires are connected as a source of electric current so that one disk is positively charged and one is negatively charged. A beam appears between the disks. • He hypothesized that the beam was made of negative particles

• He placed two plates (one positive and one negative) on either side of the beam. • Evidence that his hypotheses was true: No matter the type of of metal used as disks, the beam bent towards the positive plate and the particles had about 1/2000 of hydrogen atom.

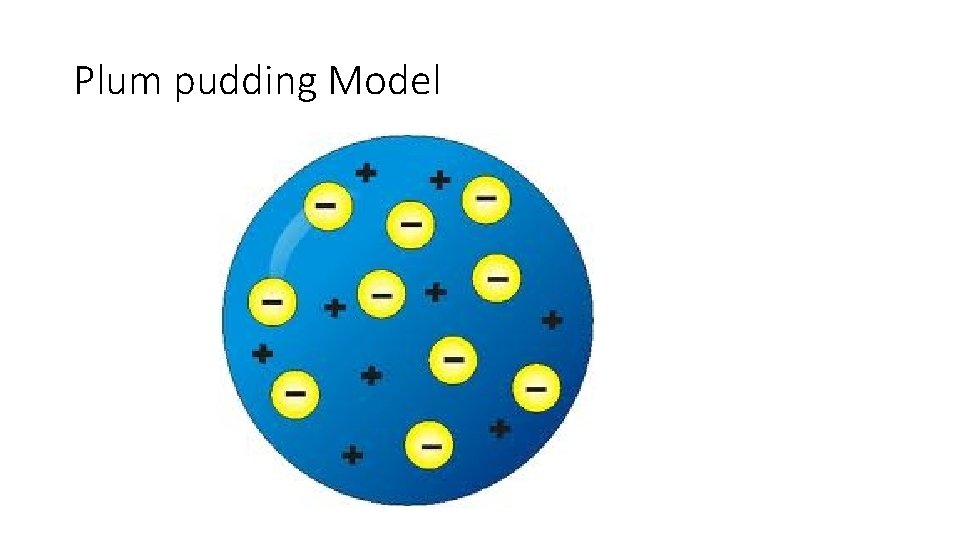
Theory • Atoms are made of subatomic particles – the plum pudding model.

Plum pudding Model

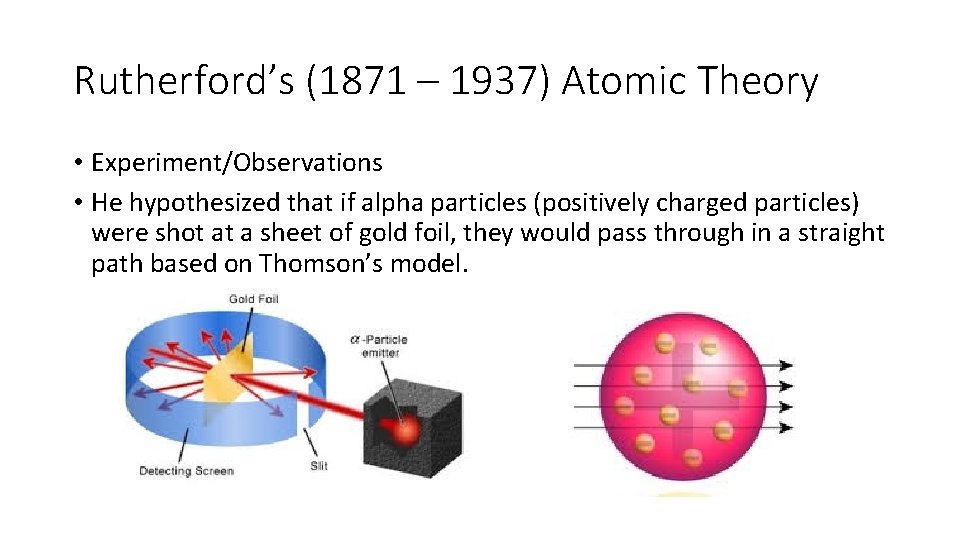
Rutherford’s (1871 – 1937) Atomic Theory • Experiment/Observations • He hypothesized that if alpha particles (positively charged particles) were shot at a sheet of gold foil, they would pass through in a straight path based on Thomson’s model.

• About one out of every 20, 000 alpha particles were deflected more than 90 degrees – they must have come close to a charged object. • The closer to this charge – the greater the deflection.

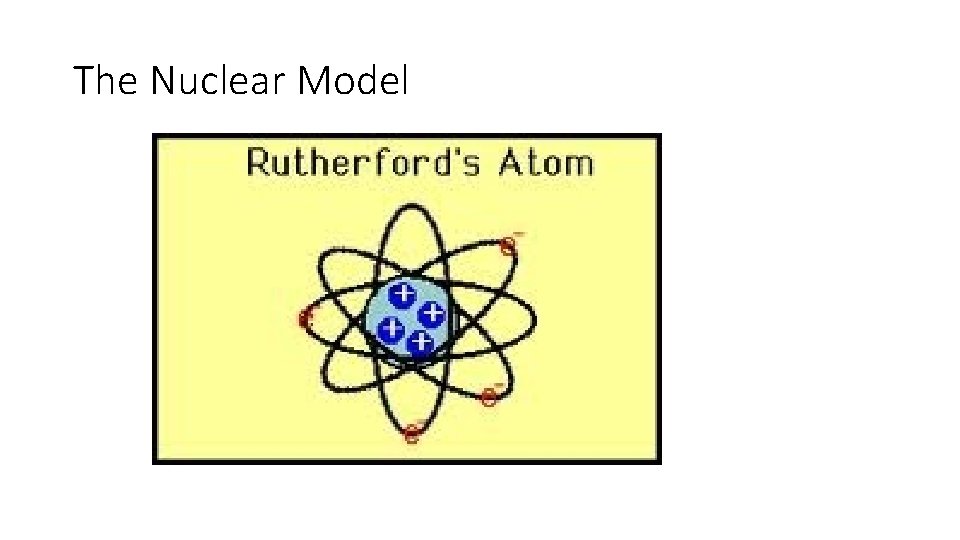
Theory • The positive charge of an atom is concentrated in it’s nucleus – the nuclear model.

The Nuclear Model

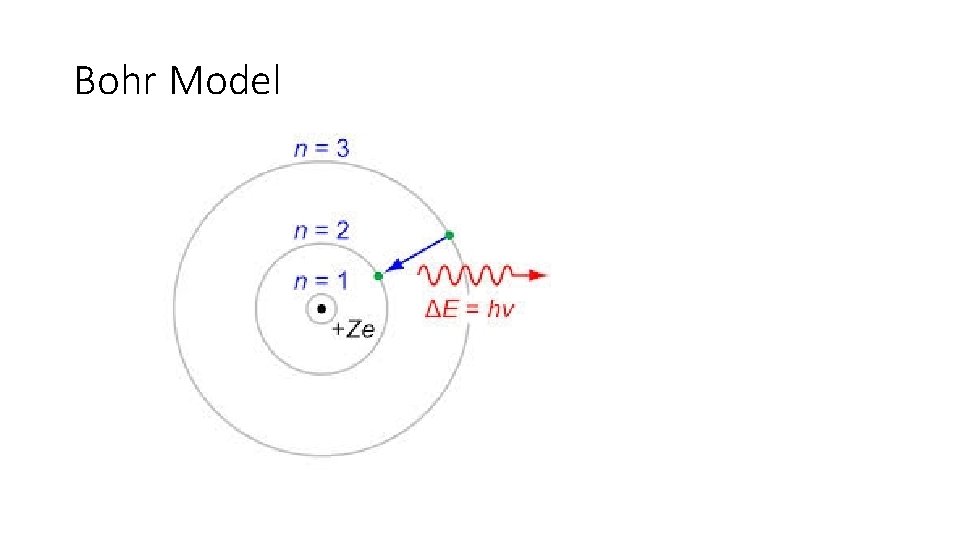
Neils Bohr (1885 – 1962) • Experiment/Observations: • In Rutherford’s model, the electrons would be constantly emitting energy according to classical physics, and the atom would be very unstable. • To fix this, Bohr theorized that electrons must have a fixed speed, and fixed energy. • Using Planck’s constant for light emissions, Bohr obtained the accurate formulas for energy levels of hydrogen atom.

Theory • Electrons orbit the nucleus at fixed spends and fixed energy. • Electrons may change energy levels by gaining or losing energy.

Bohr Model

Current – Electron Cloud Model • Schrodinger developed probability function of where an electron may be found – we cannot say with certainty where an electron actually is at any point.

Electron Cloud Model