Models of Acids and Bases Arrhenius Concept Acids

Models of Acids and Bases Arrhenius Concept: Acids produce H+ in solution, bases produce OH ion. Brønsted-Lowry: Acids are H+ donors, bases are proton acceptors. HCl + H 2 O Cl + H 3 O+ acid base Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 1

Conjugate Acid/Base Pairs HA(aq) + H 2 O(l) H 3 O+(aq) + A (aq) conj acid 1 conj base 2 conj acid 2 conj base 1 conjugate base: everything that remains of the acid molecule after a proton is lost. conjugate acid: formed when the proton is transferred to the base. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 2

Acid Dissociation Constant (Ka) HA(aq) + H 2 O(l) H 3 O+(aq) + A (aq) Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 3

Acid Strength Strong Acid: 4 Its equilibrium position lies far to the right. (HNO 3) 4 Yields a weak conjugate base. (NO 3 ) Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 4

Acid Strength (continued) Weak Acid: 4 Its equilibrium lies far to the left. (CH 3 COOH) 4 Yields a much stronger (it is relatively strong) conjugate base than water. (CH 3 COO ) Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 5

Water as an Acid and a Base Water is amphoteric (it can behave either as an acid or a base). H 2 O + H 2 O H 3 O+ + OH acid 1 base 2 conj acid 2 conj base 1 Kw = 1 10 14 at 25°C Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 6
![The p. H Scale p. H log[H+] p. H in water ranges from 0 The p. H Scale p. H log[H+] p. H in water ranges from 0](http://slidetodoc.com/presentation_image_h/258cc23947249cbeb0c05725265221db/image-7.jpg)
The p. H Scale p. H log[H+] p. H in water ranges from 0 to 14. Kw = 1. 00 10 14 = [H+] [OH ] p. Kw = 14. 00 = p. H + p. OH As p. H rises, p. OH falls (sum = 14. 00). Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 7

Solving Weak Acid Equilibrium Problems 4 List major species in solution. 4 Choose species that can produce H+ and write reactions. 4 Based on K values, decide on dominant equilibrium. 4 Write equilibrium expression for dominant equilibrium. 4 List initial concentrations in dominant equilibrium. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 8

Solving Weak Acid Equilibrium Problems (continued) 4 Define change at equilibrium (as “x”). 4 Write equilibrium concentrations in terms of x. 4 Substitute equilibrium concentrations into equilibrium expression. 4 Solve for x the “easy way. ” 4 Verify assumptions using 5% rule. 4 Calculate [H+] and p. H. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 9

Percent Dissociation (Ionization) Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 10

Bases “Strong” and “weak” are used in the same sense for bases as for acids. strong = complete dissociation (hydroxide ion supplied to solution) Na. OH(s) Na+(aq) + OH (aq) Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 11

Bases (continued) weak = very little dissociation (or reaction with water) H 3 CNH 2(aq) + H 2 O(l) H 3 CNH 3+(aq) + OH (aq) Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 12

Polyprotic Acids . . . can furnish more than one proton (H+) to the solution. Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 13
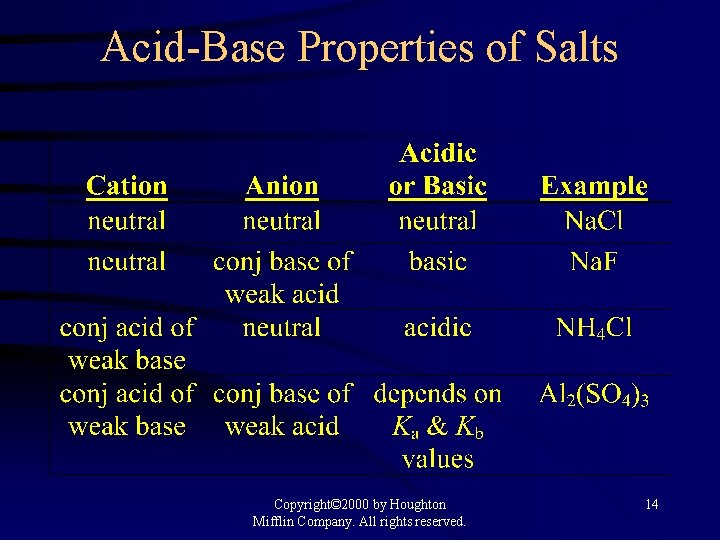
Acid-Base Properties of Salts Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 14

Structure and Acid-Base Properties Two factors for acidity in binary compounds: 4 Bond Polarity (high is good) 4 Bond Strength (low is good) Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 15

Oxides Acidic Oxides (Acid Anhydrides): 4 O X bond is strong and covalent. SO 2, NO 2, Cr. O 3 Basic Oxides (Basic Anhydrides): 4 O X bond is ionic. K 2 O, Ca. O Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 16
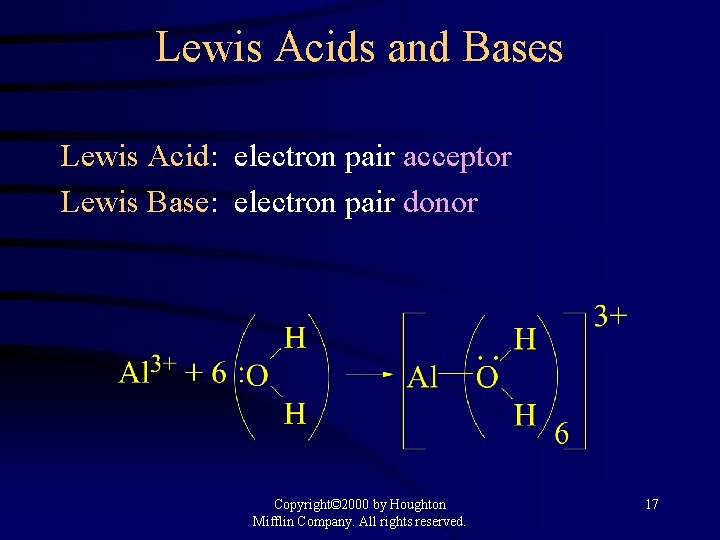
Lewis Acids and Bases Lewis Acid: electron pair acceptor Lewis Base: electron pair donor Copyright© 2000 by Houghton Mifflin Company. All rights reserved. 17
- Slides: 17