Models Models are attempts to describe reality that

Models • “Models are attempts to describe reality, that doesn’t mean they necessarily have anything to do with reality” • Models describe some aspect(s) of a system governed by phenomena the model attempts to describe

Variables • In any model, looking at a process involves something that can change, a variable: • Extensive variable: depends on the amount present (mass, volume) • Intensive Variable: property is not additive, divisible (temperature) • Models describing energy transfer fall under the study called thermodynamics

Variables • For models, variables are key, and how some process changes a variable is the key to these models • ex. As we heat a pool of water how does the amount of mineral dissolved change, as our car burns gas, how does it’s position change • Describing these changes is done through differential calculus:

Review of calculus principles • Process (function) y driving changes in x: y=y(x), the derivative of this is dy/dx (or y’(x)), is the slope of y with x • By definition, if y changes an infinitesimally small amount, x will essentially not change: dy/dk= • This derivative describes how the function y(x) changes in response to a variable

Partial differentials • Most models are a little more complex, reflecting the fact that functions (processes) are often controlled by more than 1 variable • How fast Fe 2+ oxidizes to Fe 3+ is a process that is affected by temperature, p. H, how much O 2 is around, and how much Fe 2+ is present at any one time what does this function look like, how do we figure it out? ? ?

• Total differential, dy, describing changes in y affected by changes in all variables (more than one, none held constant)
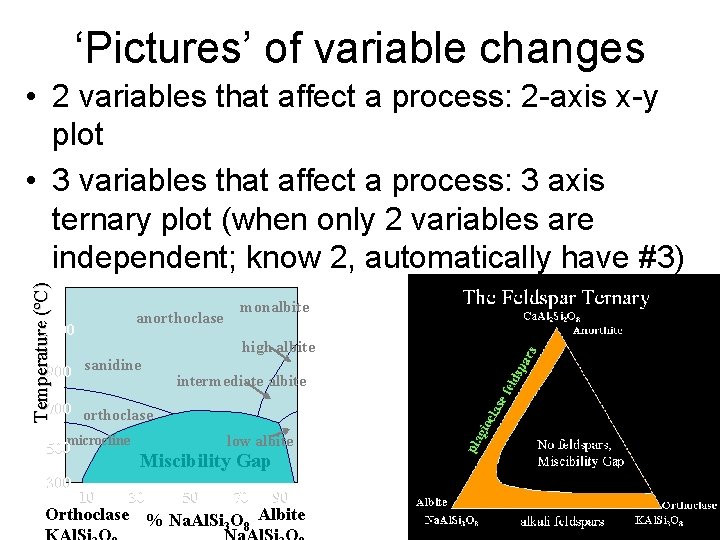
‘Pictures’ of variable changes Temperature (ºC) • 2 variables that affect a process: 2 -axis x-y plot • 3 variables that affect a process: 3 axis ternary plot (when only 2 variables are independent; know 2, automatically have #3) anorthoclase 1100 monalbite high albite 900 sanidine intermediate albite 700 orthoclase microcline 500 300 10 low albite Miscibility Gap 30 50 70 90 Orthoclase % Na. Al. Si O Albite 3 8
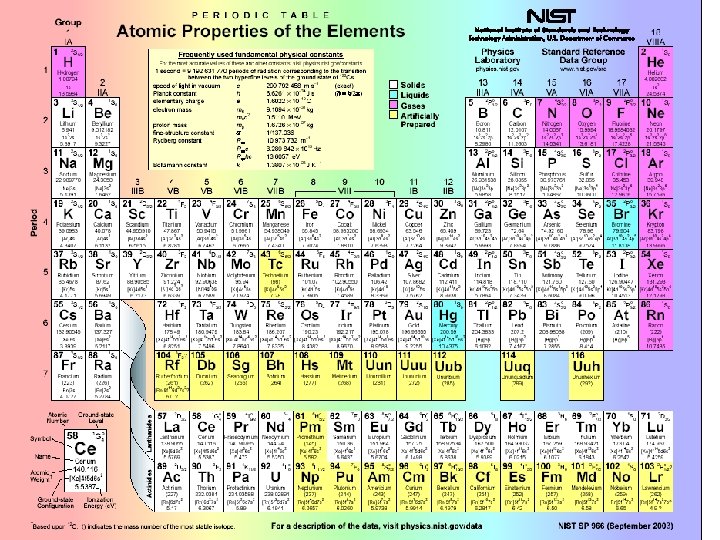

Properties derived from outer e • Ionization potential energy required to remove the least tightly bound electron • Electron affinity energy given up as an electron is added to an element • Electronegativity quantifies the tendency of an element to attract a shared electron when bonded to another element.
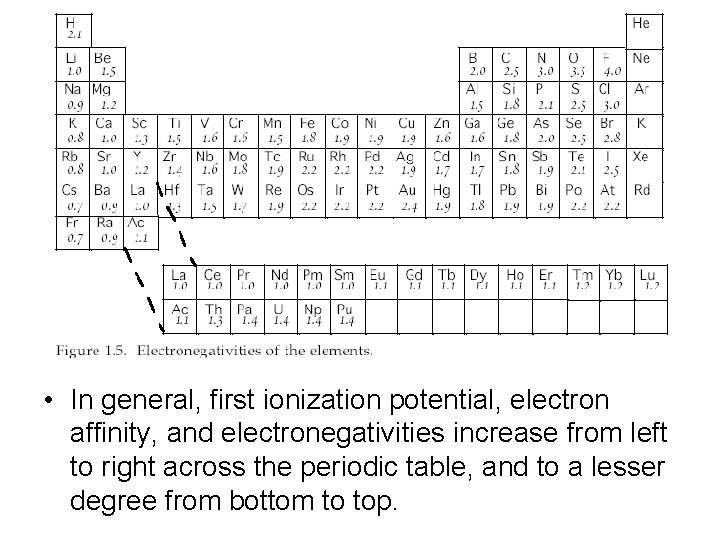
• In general, first ionization potential, electron affinity, and electronegativities increase from left to right across the periodic table, and to a lesser degree from bottom to top.

Ionic vs. Covalent • Elements on the right and top of the periodic table draw electrons strongly • Bonds between atoms from opposite ends more ionic, diatomics are 100% covalent • Bond strength Covalent>Ionic>metallic – Affects hardness, melting T, solubility • Bond type affects geometry of how ions are arranged – More ionic vs. covalent = higher symmetry
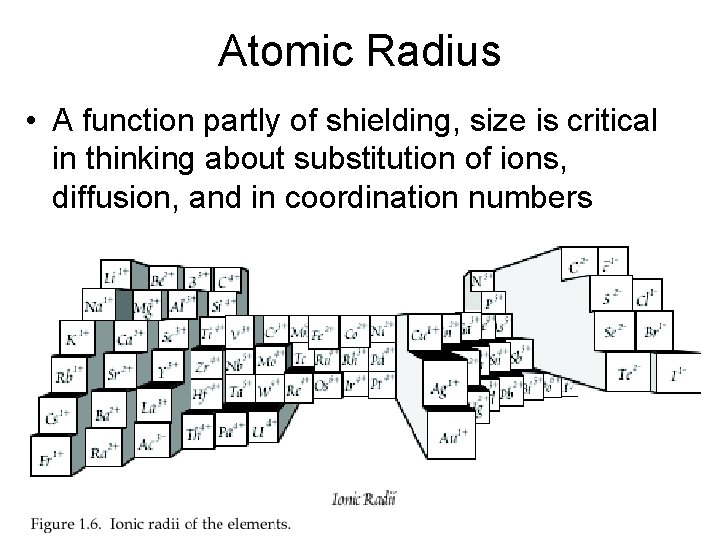
Atomic Radius • A function partly of shielding, size is critical in thinking about substitution of ions, diffusion, and in coordination numbers

Units review • Mole = 6. 02214 x 1023 ‘units’ make up 1 mole, 1 mole of H+= 6. 02214 x 1023 H+ ions, 10 mol Fe. OOH = 6. 02214 x 1024 moles Fe, 6. 02214 x 1024 moles OH. A mole of something is related to it’s mass by the gram formula weight Molecular weight of S = 32. 04 g, so 32. 04 grams S has 6. 02214 x 1023 S atoms. • Molarity = moles / liter solution • Molality = moles / kg solvent • ppm = 1 part in 1, 000, 00 (106) parts by mass or volume • Conversion of these units is a critical skill!!
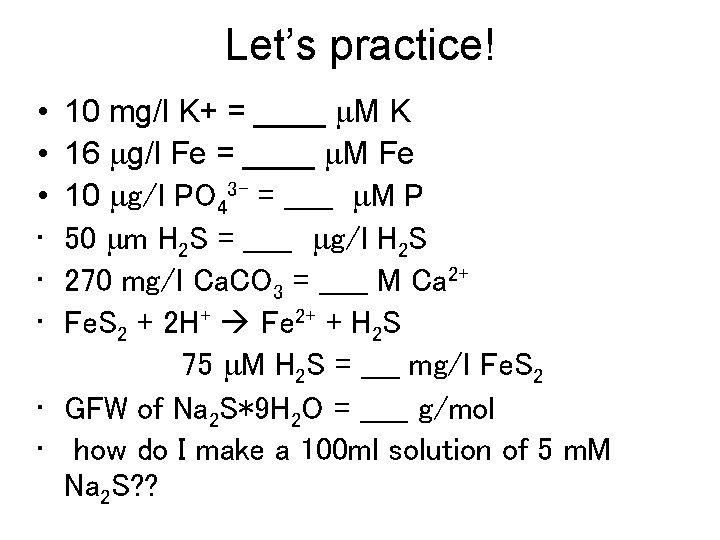
Let’s practice! 10 mg/l K+ = ____ m. M K 16 mg/l Fe = ____ m. M Fe 10 mg/l PO 43 - = _____ m. M P 50 mm H 2 S = _____ mg/l H 2 S 270 mg/l Ca. CO 3 = _____ M Ca 2+ Fe. S 2 + 2 H+ Fe 2+ + H 2 S 75 m. M H 2 S = ____ mg/l Fe. S 2 • GFW of Na 2 S*9 H 2 O = _____ g/mol • how do I make a 100 ml solution of 5 m. M Na 2 S? ? • • •

Scientific Notation • 4. 517 E-06 = 4. 517 x 10 -6 = 0. 000004517 • Another way to represent this: take the log = 105. 345 M k 1 E+6 1000 1 d c m m n p 0. 1 0. 01 1 E-3 1 E-6 1 E-9 1 E-12

Significant Figures • Precision vs. Accuracy • Significant figures – number of digits believed to be precise LAST digit is always assumed to be an estimate • Using numbers from 2 sources of differing precision must use lowest # of digits – Mass = 2. 05546 g, volume= 100. 0 ml = 0. 2055 g/l

Logarithm review • 103 = 1000 • ln = 2. 303 log x • p. H = -log [H+] 0. 015 M H+ is what p. H? • Antilogarithms: 10 x or ex (anti-natural log) • p. H = -log [H+] how much H+ for p. H 2?
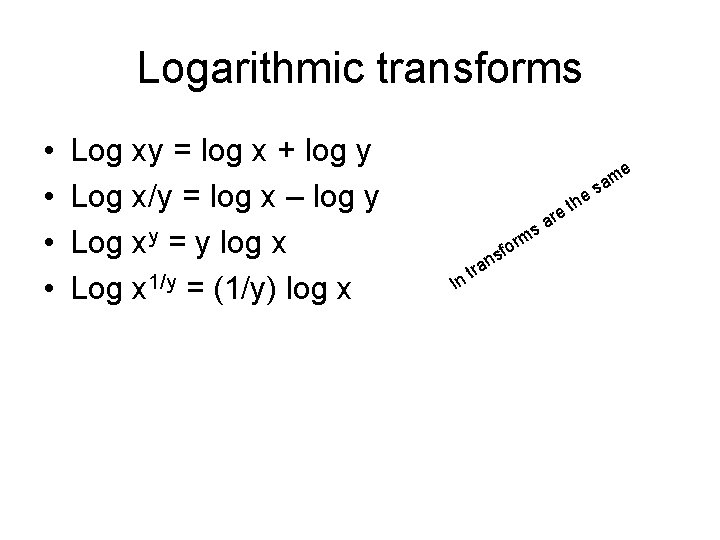
Logarithmic transforms • • Log xy = log x + log y Log x/y = log x – log y Log xy = y log x Log x 1/y = (1/y) log x m re a s ln t for s ran th e m a es

Line Fitting • Line fitting is key to investigating experimental data and calibrating instruments for analysis • Common assessment of how well a line ‘fits’ is the R 2 value – 1 is perfect, 0 is no correlation
- Slides: 19