Modelling the effects of hypoxia on fish Kenneth



















- Slides: 38

Modelling the effects of hypoxia on fish Kenneth Rose Dept. of Oceanography and Coastal Sciences Louisiana State University plus Many Co-authors

• Aaron Adamack and Shaye Sable - Louisiana State University • Cheryl A. Murphy – LSU, now University of Toronto • Peter Thomas and Saydur Rahman – University of Texas Marine Science Institute • Marius Brouwer and Nancy Brown-Peterson - University of Southern Mississippi • Ann O. Cheek - University of Texas Health Science Center • Carl Cerco - U. S. ACOE • Sandra Diamond, Texas Tech University

Acknowledgements • EPA’s Science to Achieve Results (STAR) Program to University of Texas • STAR Estuarine and Great Lakes (Ea. GLe) Program through funding to the Consortium for Estuarine Ecoindicator Research for the Gulf of Mexico (CEER-GOM), US EPA Agreement (R 82945801) • NOAA Aquatic Research Consortium (ARC, Phase 2) to USM and Texas State University

Introduction • Quantifying and forecasting effects of hypoxia is needed for effective management • Today: four examples – Physiological – Croaker matrix projection – Marsh community – Bay anchovy coupled to water quality

Disclaimer No real data were harmed in the preparation of this presentation

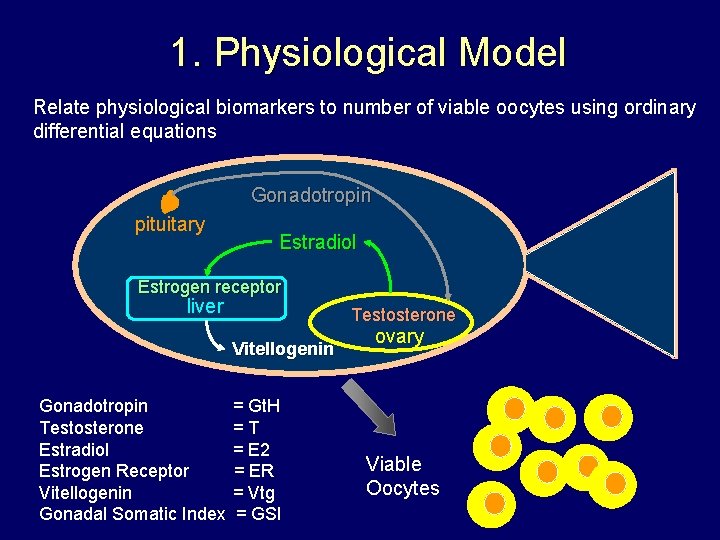
1. Physiological Model Relate physiological biomarkers to number of viable oocytes using ordinary differential equations Gonadotropin pituitary Estradiol Estrogen receptor liver Testosterone Vitellogenin Gonadotropin Testosterone Estradiol Estrogen Receptor Vitellogenin Gonadal Somatic Index = Gt. H =T = E 2 = ER = Vtg = GSI ovary Viable Oocytes

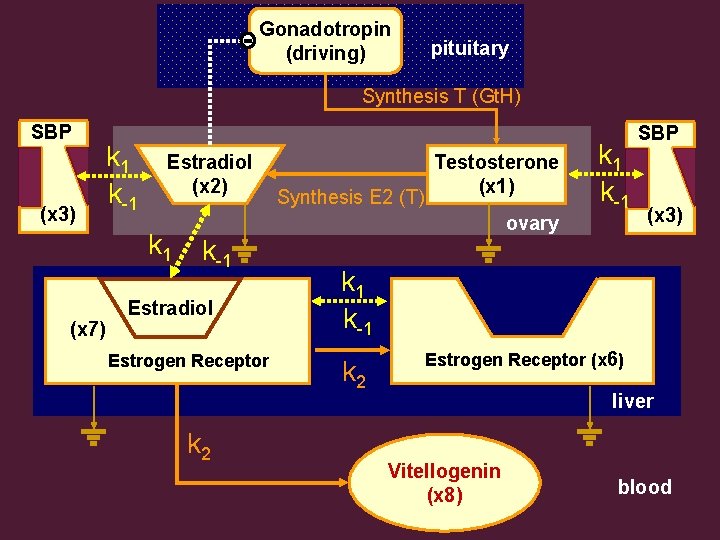
Gonadotropin (driving) pituitary Synthesis T (Gt. H) SBP (x 3) k 1 k-1 Estradiol (x 2) k 1 (x 7) Synthesis E 2 (T) Testosterone (x 1) ovary k-1 Estradiol Estrogen Receptor k 2 k 1 k-1 SBP (x 3) k 1 k-1 k 2 Estrogen Receptor (x 6) liver Vitellogenin (x 8) blood

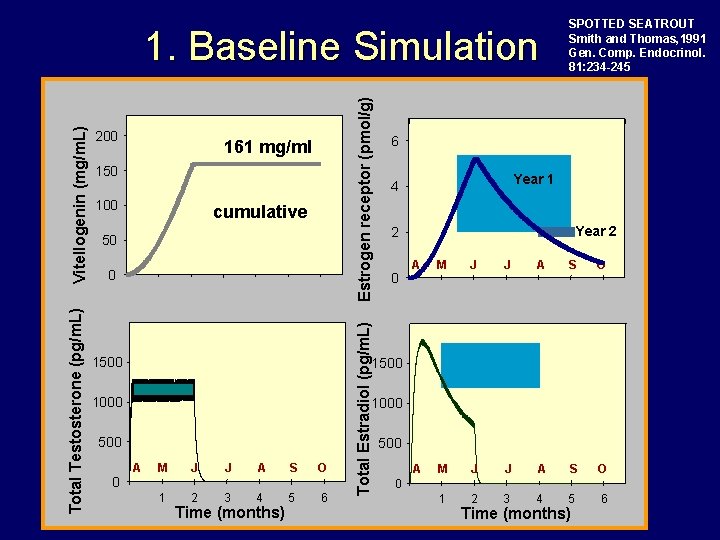
Estrogen receptor (pmol/g) 200 161 mg/ml 150 100 cumulative 50 0 1500 6 Year 1 4 Year 2 2 0 A M J J A S O 1 2 3 4 5 6 1500 1000 500 0 Total Estradiol (pg/m. L) Total Testosterone (pg/m. L) Vitellogenin (mg/m. L) 1. Baseline Simulation SPOTTED SEATROUT Smith and Thomas, 1991 Gen. Comp. Endocrinol. 81: 234 -245 A M J J A S O 1 2 3 4 5 6 Time (months) 500 0 Time (months)

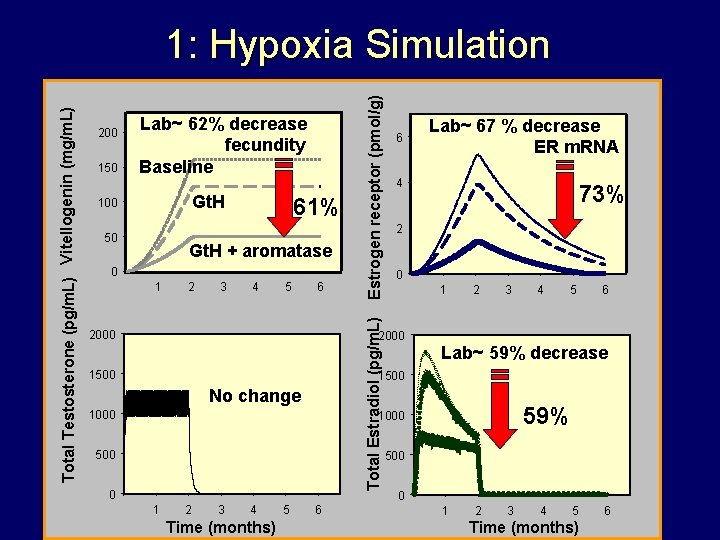
1. Hypoxia Simulation 1. Simulate gonadotropin suppression • Multiply gonadotropin driving variable by 0. 74 2. Simulate aromatase impairment • • • Less Testosterone converted to estradiol Small % of Estradiol sent to a “sink” every timestep Total E 2 is 41% of Control

150 Lab~ 62% decrease fecundity Baseline Gt. H 100 50 61% Gt. H + aromatase 0 1 2 3 4 5 6 Estrogen receptor (pmol/g) 200 Total Estradiol (pg/m. L) Total Testosterone (pg/m. L) Vitellogenin (mg/m. L) 1: Hypoxia Simulation 2000 6 4 73% 2 0 1 2000 1500 Lab~ 67 % decrease ER m. RNA 2 3 4 5 6 Lab~ 59% decrease 1500 No change 1000 59% 1000 500 0 1 2 3 4 Time (months) 5 6 500 0 1 2 3 4 5 Time (months) 6

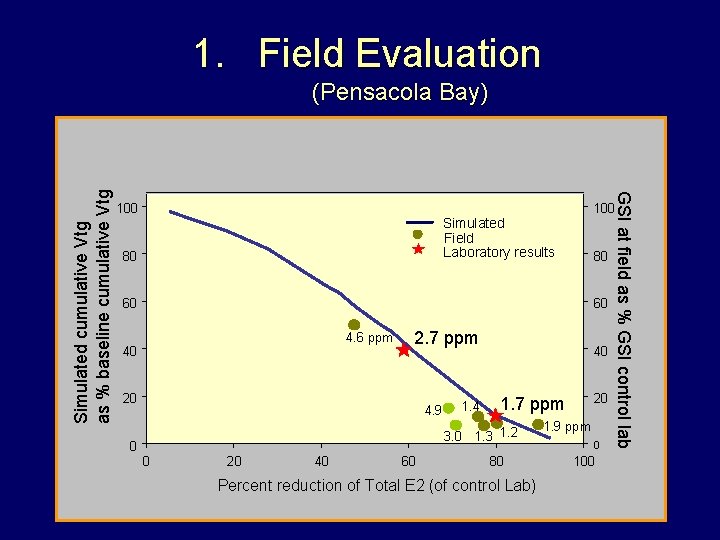
1. Field Evaluation 100 Simulated Field Laboratory results 80 80 60 60 4. 6 ppm 40 2. 7 ppm 20 4. 9 1. 4 40 0 0 20 40 60 20 1. 7 ppm 3. 0 1. 3 1. 2 80 Percent reduction of Total E 2 (of control Lab) GSI at field as % GSI control lab Simulated cumulative Vtg as % baseline cumulative Vtg (Pensacola Bay) 1. 9 ppm 0 100

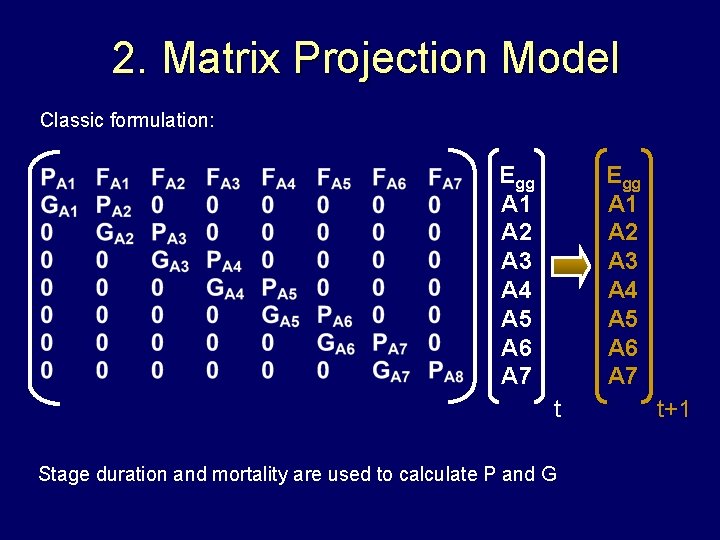
2. Matrix Projection Model Classic formulation: Egg A 1 A 2 A 3 A 4 A 5 A 6 A 7 t Stage duration and mortality are used to calculate P and G t+1

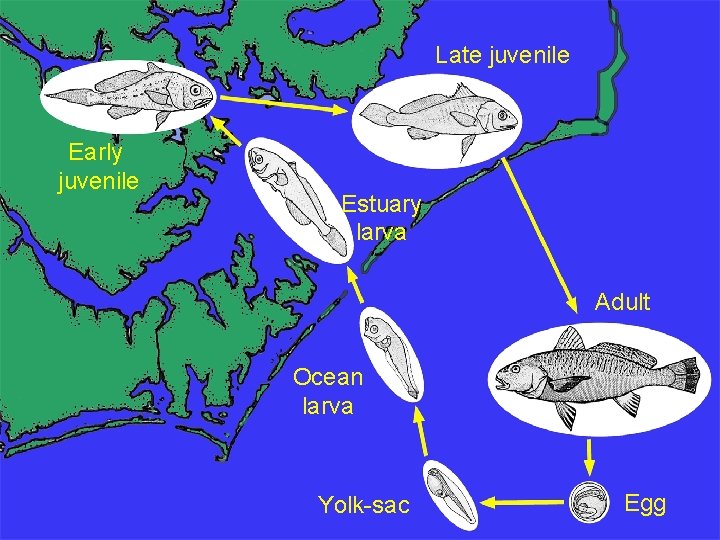
Late juvenile Early juvenile Estuary larva Adult Ocean larva Yolk-sac Egg

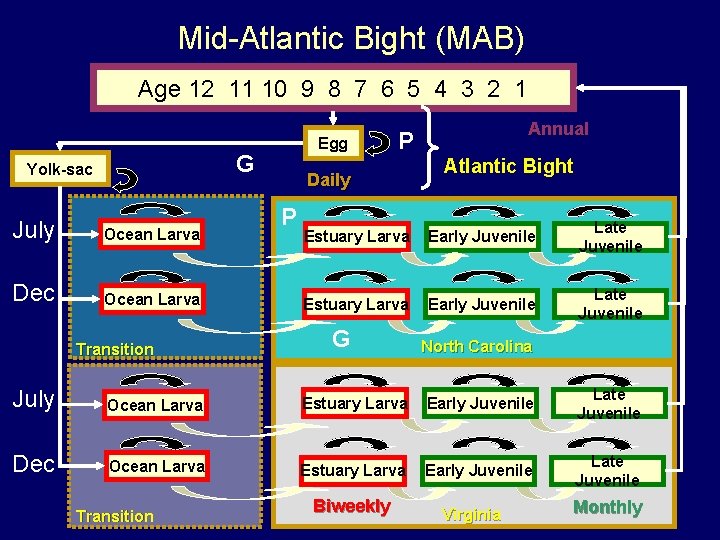
Mid-Atlantic Bight (MAB) Age 12 11 10 9 8 7 6 5 4 3 2 1 Egg G Yolk-sac July Ocean Larva Dec Ocean Larva Transition P Daily P Annual Atlantic Bight Estuary Larva Early Juvenile Late Juvenile G North Carolina July Ocean Larva Estuary Larva Early Juvenile Late Juvenile Dec Ocean Larva Estuary Larva Early Juvenile Late Juvenile Transition Biweekly Virginia Monthly

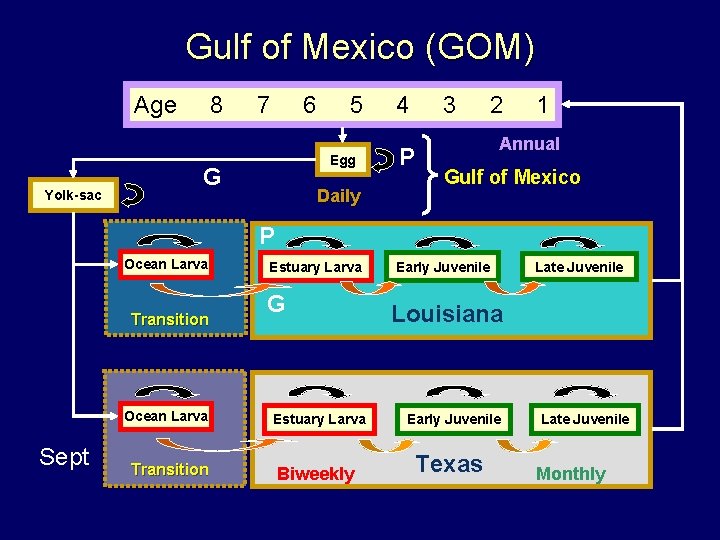
Gulf of Mexico (GOM) Age Yolk-sac 8 7 6 5 Egg G Daily 4 3 P 2 1 Annual Gulf of Mexico P Ocean Larva Transition Ocean Larva Sept Transition Estuary Larva G Estuary Larva Biweekly Early Juvenile Late Juvenile Louisiana Early Juvenile Texas Late Juvenile Monthly

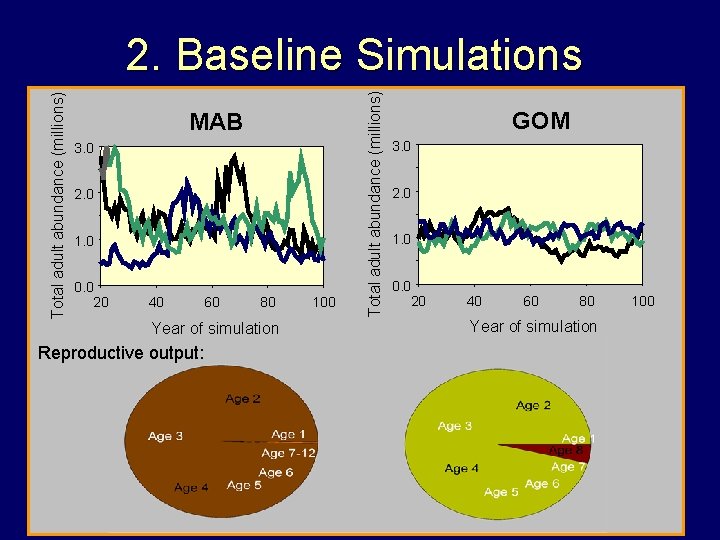
MAB 3. 0 2. 0 1. 0 0. 0 20 40 60 80 Year of simulation Reproductive output: 100 Total adult abundance (millions) 2. Baseline Simulations GOM 3. 0 2. 0 1. 0 0. 0 20 40 60 80 Year of simulation 100

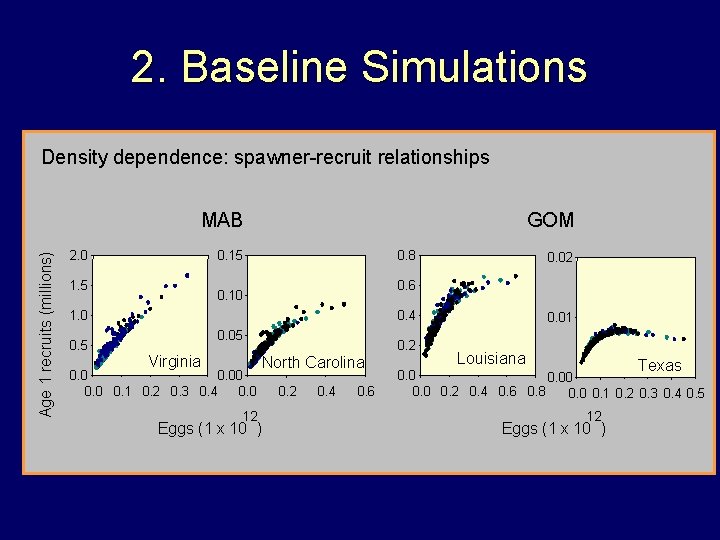
2. Baseline Simulations Density dependence: spawner-recruit relationships Age 1 recruits (millions) MAB 2. 0 GOM 0. 15 1. 5 0. 8 0. 6 0. 10 1. 0 0. 5 0. 02 0. 4 0. 05 Virginia 0. 00 0. 1 0. 2 0. 3 0. 4 0. 0 12 0. 2 North Carolina Eggs (1 x 10 ) 0. 2 0. 4 0. 6 0. 01 Louisiana Texas 0. 00 0. 2 0. 4 0. 6 0. 8 0. 0 0. 1 0. 2 0. 3 0. 4 0. 5 12 Eggs (1 x 10 )

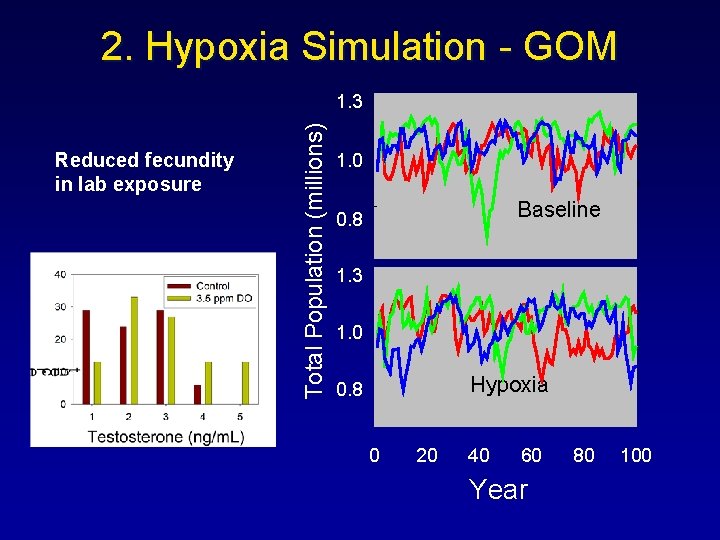
2. Hypoxia Simulation - GOM Reduced fecundity in lab exposure Total Population (millions) 1. 3 1. 0 Baseline 0. 8 1. 3 1. 0 Hypoxia 0. 8 0 20 40 60 Year 80 100

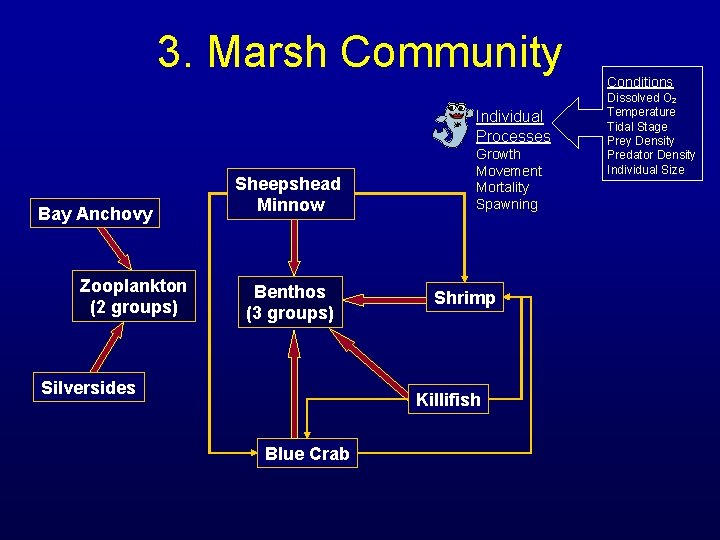
3. Marsh Community Individual Processes Bay Anchovy Zooplankton (2 groups) Sheepshead Minnow Benthos (3 groups) Silversides Growth Movement Mortality Spawning Shrimp Killifish Blue Crab Conditions Dissolved O 2 Temperature Tidal Stage Prey Density Predator Density Individual Size

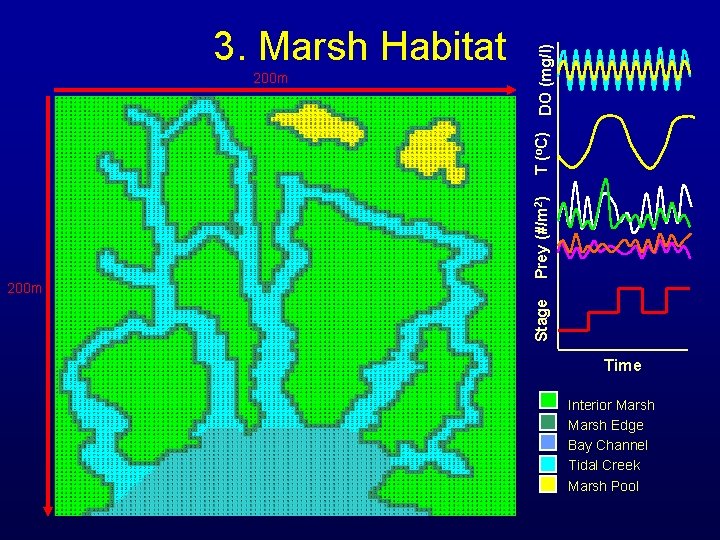
Prey (#/m 2) T (o. C) 200 m DO (mg/l) 3. Marsh Habitat Stage 200 m Time Interior Marsh Edge Bay Channel Tidal Creek Marsh Pool

3. Individual Processes • Growth: – bioenergetics – consumption based on prey and predator sizes; prey densities • Movement: – neighborhood depends on tidal stage and individual motility – move to cell with highest fitness score in neighborhood • Mortality: – predation; starvation; stranding; natural • Spawning: – temperature and weight-dependent fecundity – fractional spawning with brood intervals

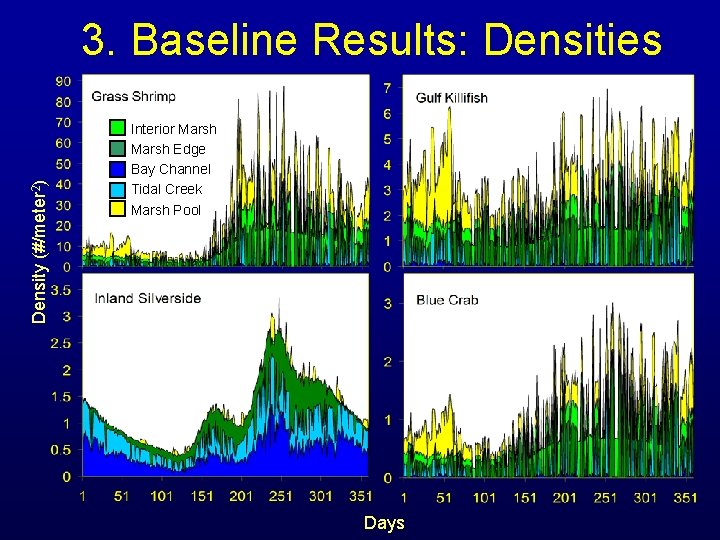
Density (#/meter 2) 3. Baseline Results: Densities Interior Marsh Edge Bay Channel Tidal Creek Marsh Pool Days

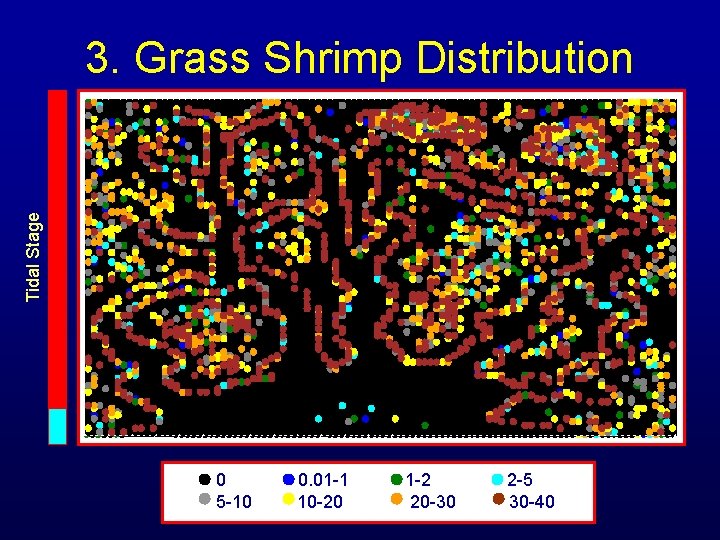
Tidal Stage 3. Grass Shrimp Distribution 0 5 -10 0. 01 -1 10 -20 1 -2 20 -30 2 -5 30 -40

3. Cyclic Dissolved Oxygen Stress * Grass shrimp: Larval z (hour-1) = 5. 5 E-4*(DO) + 0. 0007 * Grass shrimp: Brood interval (days) = 350. 4 + 255. 1/ln(DO) All species: Metabolism multiplier = 1. 79*(DO)-0. 362 when DO < 5 mg/l * Grass shrimp functions fit to laboratory data provided by Brouwer & Brown-Peterson

3. DO Stress Gulf Killifish Blue Crab

Frequency (x 105) 3. DO Stress Length (mm)

3. Gene Chips • Episodes and fluctuations complicate exposure • Data – Grass shrimp and sheepshead minnow – Lab: DO, growth and fecundity, up/down regulation – Field: gene responses • Idea is to add damage-repair (Mancini; Breck) or vitality (Anderson) sub-model to individuals – Calibrate to lab – Apply to field exposures

4. Chesapeake Bay WQM Developed by the Army Corps • 3 D hydrodynamic model (CH 3 D), eutrophication model (CE-QUAL-ICM), and sediment diagenesis model • Simulates 24 constituents – – Forms of N, P, and Si Spring and winter algae Micro- and meso-zooplankton DO and temperature • Bay is divided into 4073 cells – 729 surface cells – Minimum 2 layers thick – Maximum 15 layers thick

4. Bay Anchovy Model • Spatially-explicit, individual-based • Simulates growth, death, and movement • Dynamically coupled to the WQM – Temperature and DO affect anchovy growth and mortality – Micro- and meso-zooplankton affect anchovy growth – Consumption by anchovy is mortality on zooplankton • Movement depends on: – Horizontal: zooplankton density, temperature – Vertical: temperature, dissolved oxygen • Fixed recruitment each year

4. DO Effects DO effect on growth DO mortality

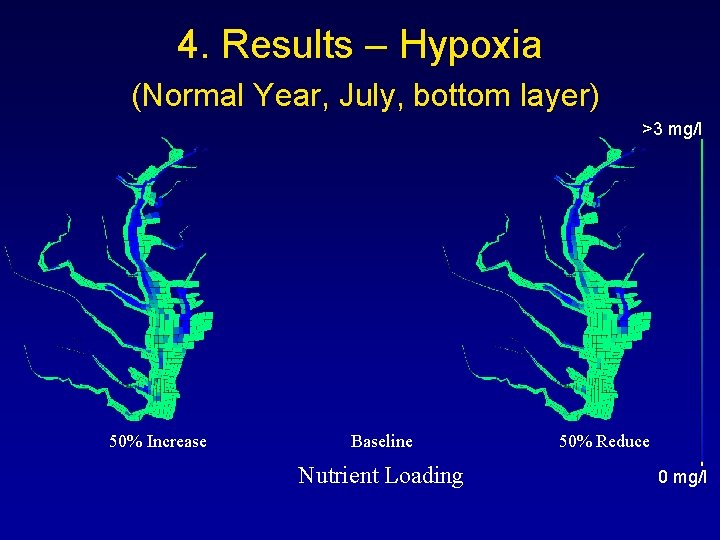
4. Results – Hypoxia (Normal Year, July, bottom layer) >3 mg/l 50% Increase Baseline Nutrient Loading 50% Reduce 0 mg/l

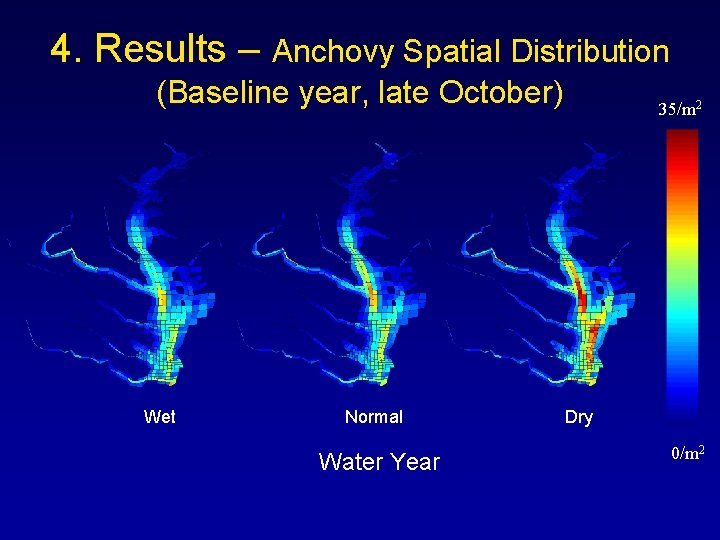
4. Results – Anchovy Spatial Distribution (Baseline year, late October) Wet Normal Water Year 35/m 2 Dry 0/m 2

4. Results – Anchovy Biomass

4. Results – Zooplankton (Station C 5. 2 – mid-bay, with anchovy)

4. Results – YOY Anchovy Length (Late October)

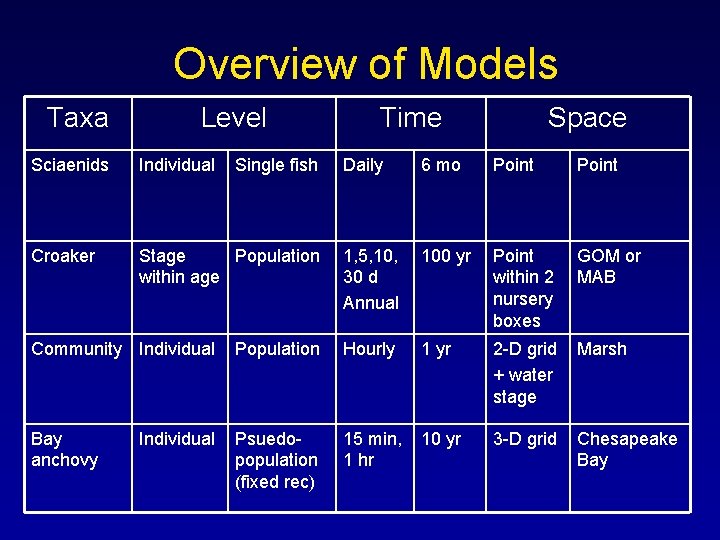
Overview of Models Taxa Level Sciaenids Individual Single fish Croaker Stage Population within age Time Space Daily 6 mo Point 1, 5, 10, 30 d Annual 100 yr Point within 2 nursery boxes GOM or MAB Community Individual Population Hourly 1 yr 2 -D grid + water stage Marsh Bay anchovy Psuedopopulation (fixed rec) 15 min, 1 hr 10 yr 3 -D grid Chesapeake Bay Individual

Concluding Remarks • Modeling techniques, measurements, and understanding are rapidly improving • Advances: – Scaled models – Spatial data – Exposure – Multiple stressors

Concluding Remarks • Optimism for quantifying indirect effects? • Key will be movement