MODELING SolutionPhase Nonlinear Dynamics Heterogeneous Dynamics 1 Brief

MODELING • Solution-Phase Nonlinear Dynamics • Heterogeneous Dynamics 1

Brief History of Chemical NLD • Chemical waves and oscillations rejected – Violate Second Law – Difficult to reproduce • Prigogine – No Second Law violation far from equilibrium • BZ reaction – Easily reproduced • Doubts allayed─new theories, experiments 2

Mechanism and Model • Mechanism─elementary steps at molecular level • Model─simplified mechanism – Abstract: Lotka-Volterra – Derived: Oregonator, Brusselator – Empirical: Rate Law 3

Calculating Rate Constants • • Diffusion-controlled limits Marcus theory of electron transfer Eigen theory of proton transfer Eigen theory of metal complex formation 4

Lotka-Volterra Model A + X 2 X, k 1 X + Y 2 Y, k 2 Y , k 3 d. X/dt = k 1 AX - k 2 XY d. Y/dt = k 2 XY - k 3 Y 5
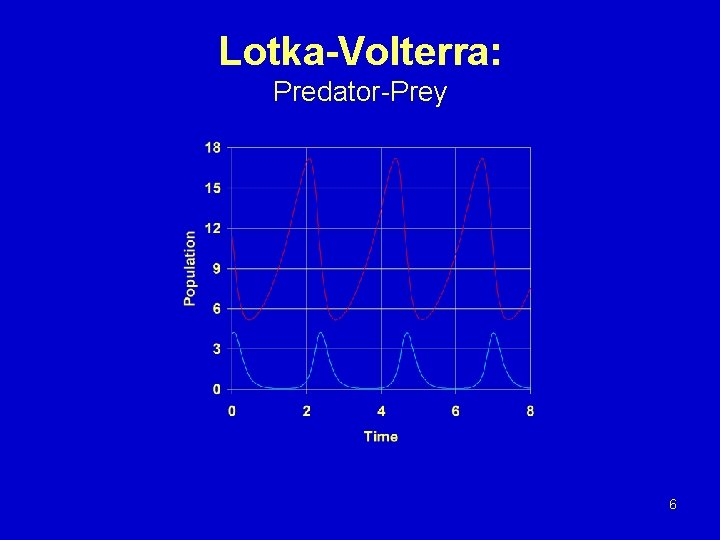
Lotka-Volterra: Predator-Prey 6

Lotka-Volterra: Increase in Predator Efficiency 7

Empirical Model: Iodate-Sulfite-Ferrocyanide A) B) C) D) E) IO 3 - + 8 I- + 6 H+ 3 I 3 - + 3 H 2 O I 3 - + HSO 3 - + H 2 O 3 I- + HSO 4 - + 2 H+ I 3 - + 2 Fe(CN)64 - 3 I- + 2 Fe(CN)63 IO 3 - + 3 HSO 3 - I- + 3 HSO 4 H+ + SO 32 - = HSO 3 - 8

Derived Model-1: Epstein-Orbán-Edblom (EOE) • Concentrations in large excess: iodate, iodide, ferrocyanide, sulfate – Incorporate into rate equation • Four variables: A = [SO 32 -], X =[HSO 3 -], Y = [H+], Z = [I 2] • Reciprocal residence time, k 0, varied • Inflow variables: Y 0, A 0 9
![Derived EOE Model-2 A = [SO 32 -], X =[HSO 3 -], Y = Derived EOE Model-2 A = [SO 32 -], X =[HSO 3 -], Y =](http://slidetodoc.com/presentation_image/5e5a3810484237a4b396b3f38e3e430a/image-10.jpg)
Derived EOE Model-2 A = [SO 32 -], X =[HSO 3 -], Y = [H+], Z = [I 2] 10
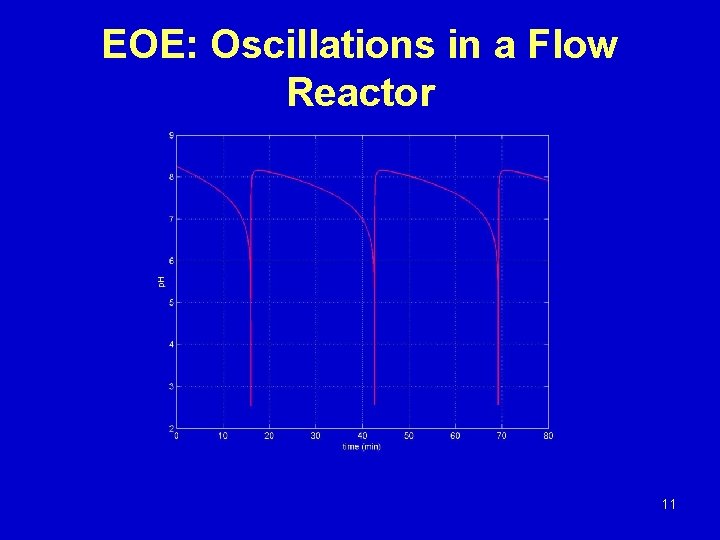
EOE: Oscillations in a Flow Reactor 11
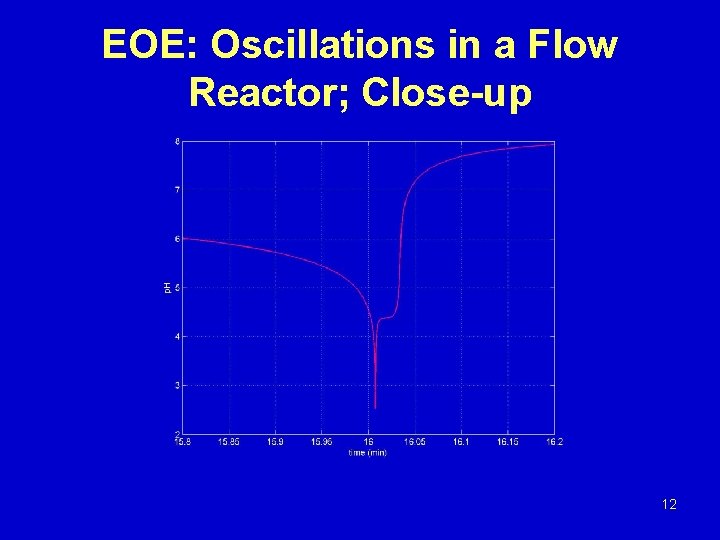
EOE: Oscillations in a Flow Reactor; Close-up 12
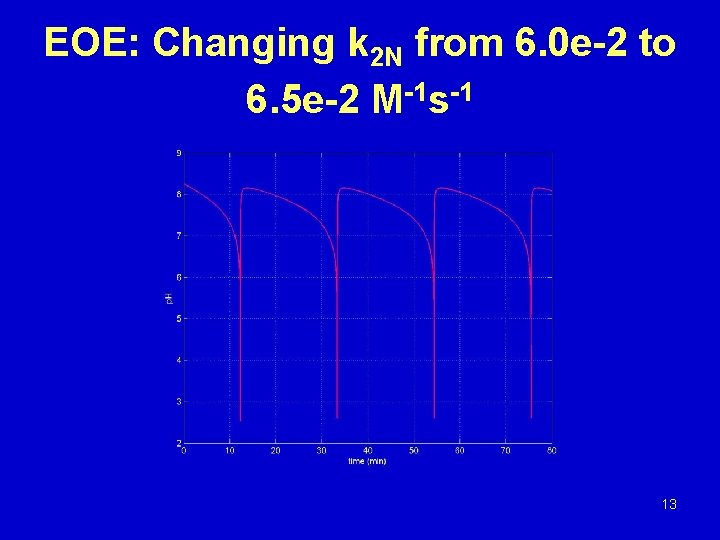
EOE: Changing k 2 N from 6. 0 e-2 to 6. 5 e-2 M-1 s-1 13

Photoresponse of the Chlorine Dioxide-Iodine-Malonic Acid Reaction in a Flow Reactor • First experimental demonstration of Turing structures • Inflow solution A – 0. 04 M H 2 SO 4 with 0. 0022 M I 2 • Inflow solution B – varying amounts of – malonic acid (w/wo) starch with chlorine dioxide 14

CDIMA: No Light (1) I 2 + MA H+ + I- + IMA (2) Cl. O 2 + I- ½I 2 + Cl. O 2(3) Cl. O 2 - + 4 I- + 4 H+ Cl- + 2 I 2 + 2 H 2 O (4) I- + I 2 + Starch. I 3(5) Starch. I 3 - I- + I 2 + Starch (6) I 2 + I- I 3(7) I 3 - I 2 + I(h 1) I 2 + H 2 O I- + H+ + IOH (h 2) I- + H+ + IOH I 2 + H 2 O 15
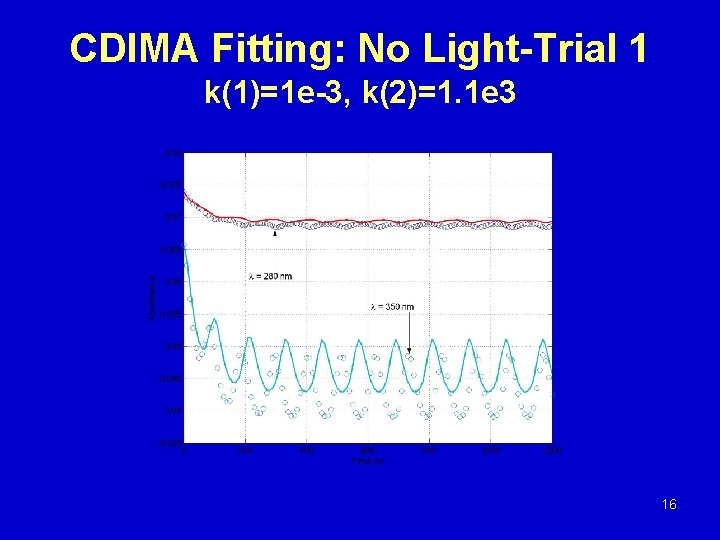
CDIMA Fitting: No Light-Trial 1 k(1)=1 e-3, k(2)=1. 1 e 3 16
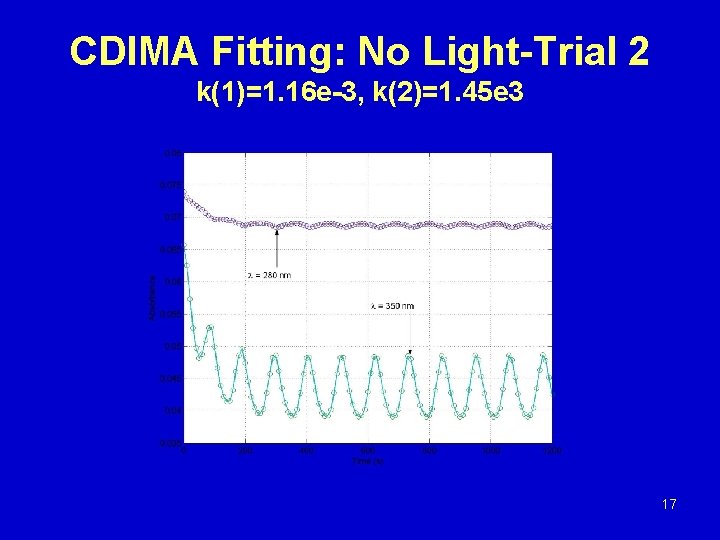
CDIMA Fitting: No Light-Trial 2 k(1)=1. 16 e-3, k(2)=1. 45 e 3 17

CDIMA: Light on/off Light on: (8) I 2 + h I· + I· (9) I· + I· I 2 (10) I· + I- I 2(11) I 2 - I· + I(12) I· +Cl. O 2 ICl. O 2 Recovery-Light off: (13) ICl. O 2 + H 2 O IO 3 - + Cl- + 2 H+ (14) 5 I- + IO 3 - + 6 H+ 3 I 2 + 3 H 2 O (16) Cl. O 2 + I 2 - Cl. O 2 - + I 2 18
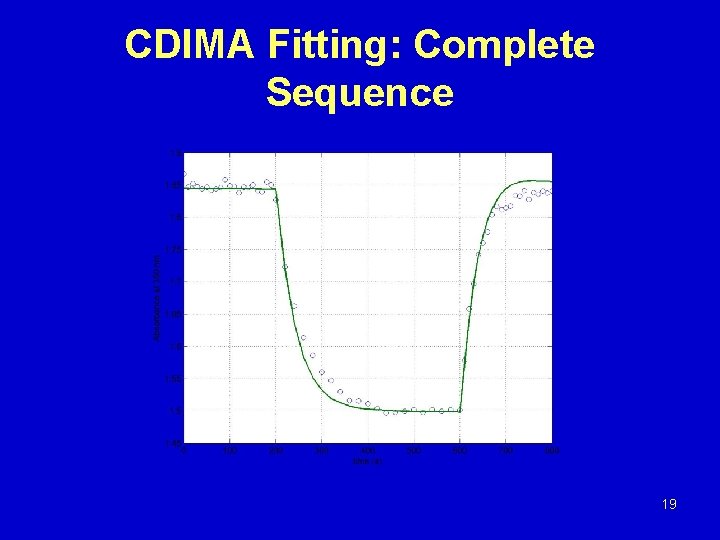
CDIMA Fitting: Complete Sequence 19

Detection of eaq- in Reductive Processes • Electrochemical reductions • Sodium amalgams • Solid-phase reductants • Heterogeneous reactions where yields may be decreased by scavenging precursors such as eaq- and H· 20

Elementary Steps in the Generation of H 2 from the Reduction of Water by Magnesium • Mg(s) 2 eaq- + Mg 2+ • eaq- + H+ H· • eaq- + eaq- H 2 + 2 OH • eaq- + H· H 2 + OH • H· + H · H 2 21

Benzoate vs. Ethanol with Trichloroacetate Mg + 2 H 2 O Mg(OH)2 + 1. 24 H· + 0. 25 eaq- 2. 18 e-3 s-1 eaq- + Cl 3 CCOO- Cl 2 C·COO + Cl- 8. 5 e 10 M-1 s-1 H· + C 6 H 5 COO- ·C 6 H 6 COO- 9. 2 e 8 M-1 s-1 H· + C 2 H 5 OH CH 3 C·HOH + H 2 1. 7 e 7 M-1 s-1 22
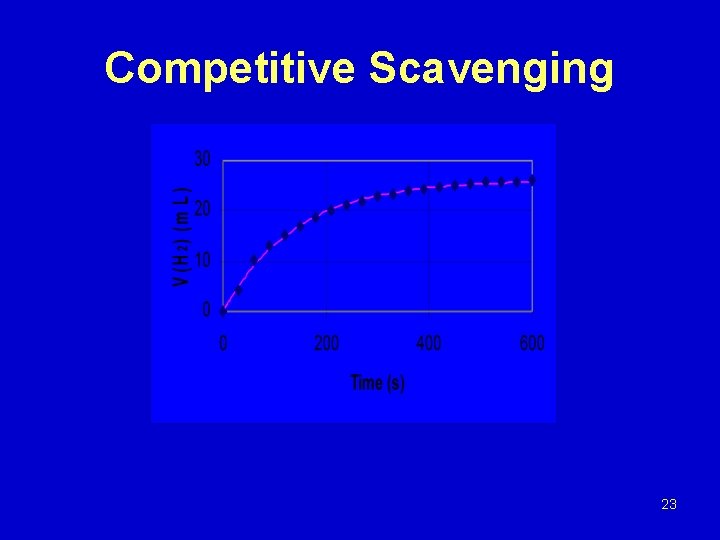
Competitive Scavenging 23
- Slides: 23