Modeling Chemistry Unit 5 Writing and Naming Compounds




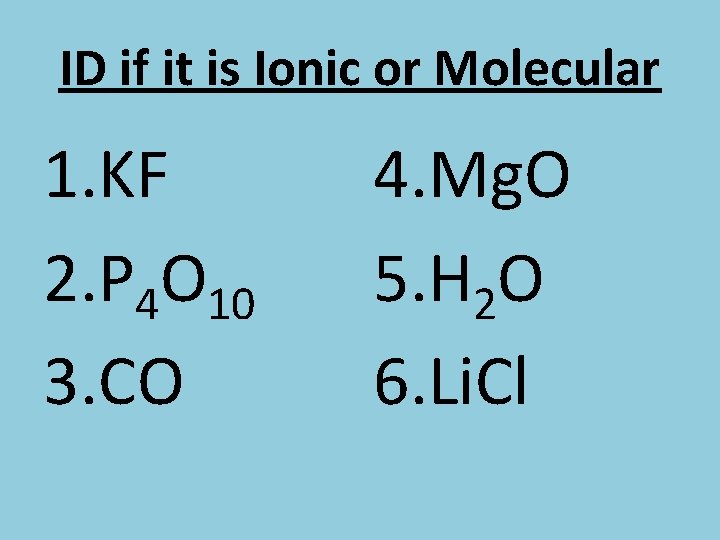














- Slides: 29

Modeling Chemistry Unit 5 Writing and Naming Compounds


Metal vs. Nonmetals • Metals – Left side of the Periodic Table • Nonmetals – Right side of the Periodic Table plus hydrogen

• Number of electrons an atom will gain or lose to become stable • Code: +1, +2, +3, ± 4, -3, -2, -1, 0

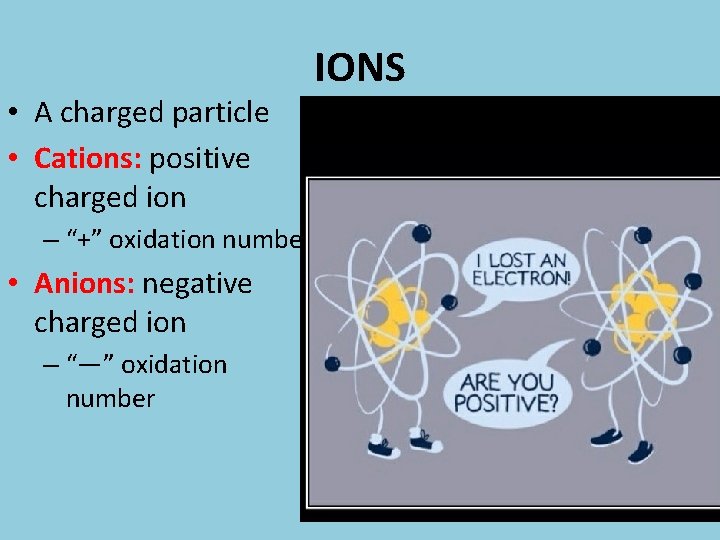
• A charged particle • Cations: positive charged ion – “+” oxidation number • Anions: negative charged ion – “—” oxidation number IONS

Chemical Bond • Is the force that holds atoms or ions together • Three Types – atomic – Molecular – ionic

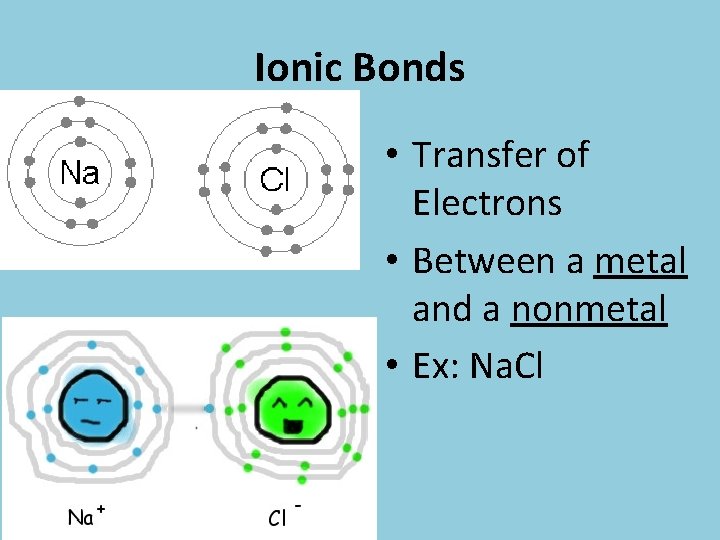
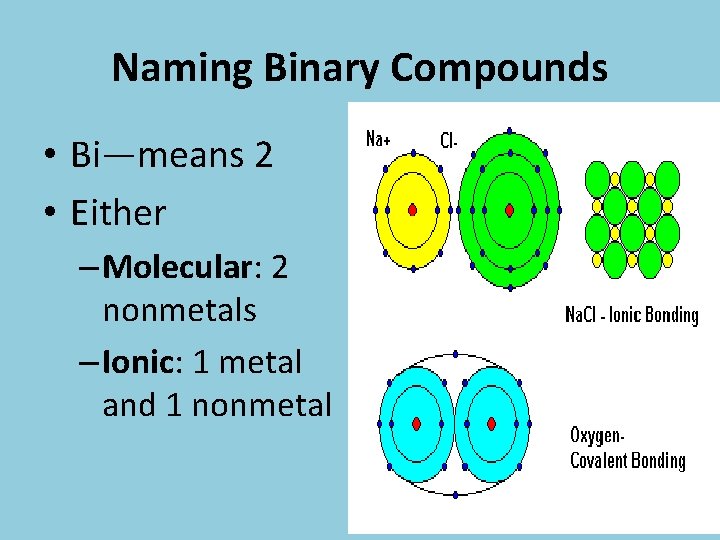
Ionic Bonds • Transfer of Electrons • Between a metal and a nonmetal • Ex: Na. Cl

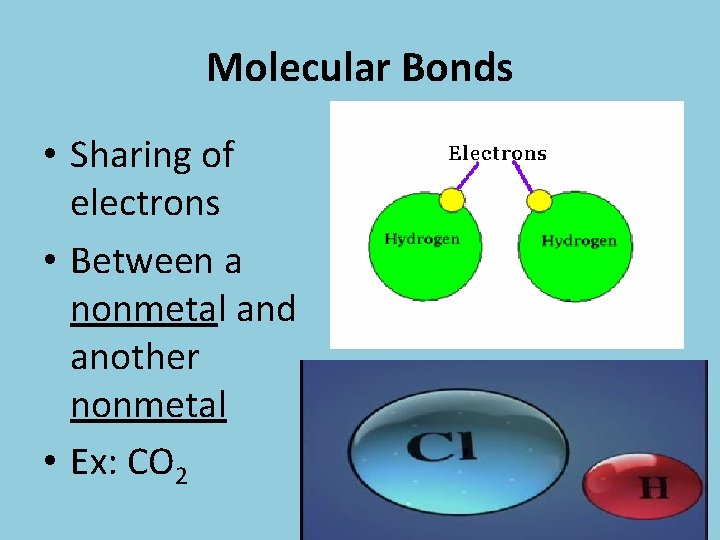
Molecular Bonds • Sharing of electrons • Between a nonmetal and another nonmetal • Ex: CO 2
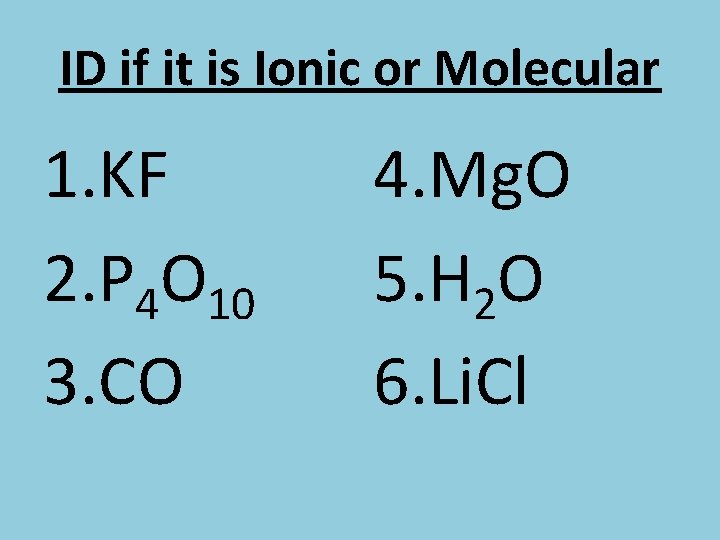
ID if it is Ionic or Molecular 1. KF 2. P 4 O 10 3. CO 4. Mg. O 5. H 2 O 6. Li. Cl

Diatomic Atoms/Molecules • Atoms of the same element bonding together • AKA: Super Seven • All are Molecular Bonds • • F 2 Cl 2 Br 2 I 2 H 2 O 2 N 2

Naming Binary Compounds • Bi—means 2 • Either – Molecular: 2 nonmetals – Ionic: 1 metal and 1 nonmetal

Naming Ionic Binary Compounds • 2 elements present – 1 metal then 1 nonmetal Steps 1. Write the name of the first element 2. Write the name of the second element 3. Change the ending of the second element to “—ide” 4. Combine together

End Changing Reference • • Chlorine Chloride Oxygen Oxide Nitrogen Nitride Sulfur Sulfide • Phosphorus Phosphide • Bromine Bromide • Iodine Iodide • Fluorine Fluoride

Practice Problem #1 #2 Ca. O Al. F 3

Naming Molecular Binary Compounds • 2 elements both being nonmetals Steps 3. Change the ending 1. Name the first of the second elements element to “—ide” 2. Name the second 4. Add prefixes based element on the subscribe of each element 5. Place together

Prefixes • • • 1 = mono— 2 = di— 3 = tri— 4 = tetra— 5 = penta— • • • 6 = hexa— 7 = hepta— 8 = octa— 9 = nona— 10 = deca— Note: “Mono—” is not used on the first element

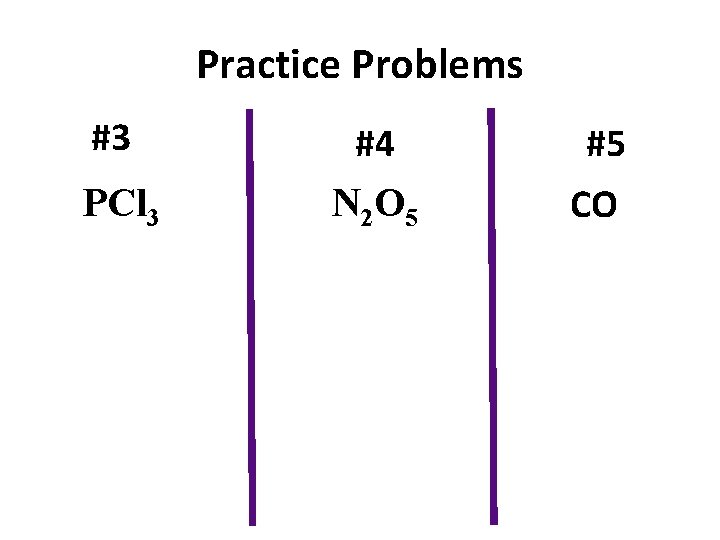
Practice Problems #3 PCl 3 #4 N 2 O 5 #5 CO

Polyatomic Ions • A group of • Ex: atoms that acts — 3 as a single PO 4 charged unit • They are Molecular Bonds

Naming Polyatomic Compounds • 3 or more elements together Steps 3. Write the name 1. Identify the of the polyatomic Polyatomic Ion ions 2. Write the name 4. Place together of the first element Note: Never change the ending to Polyatomic Ions

Practice Problems #1 #2 KNO 3 Li 3 PO 4

Practice Problems #3 #4 NH 4 OH (NH 4)2 O


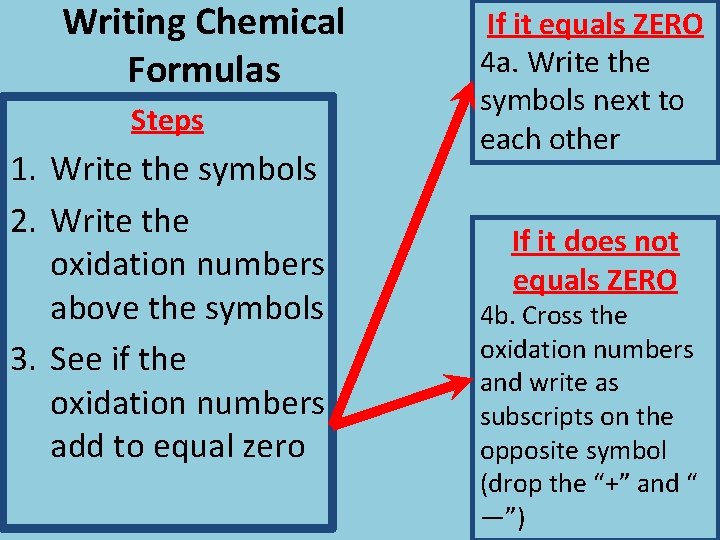
Writing Chemical Formulas Steps 1. Write the symbols 2. Write the oxidation numbers above the symbols 3. See if the oxidation numbers add to equal zero If it equals ZERO 4 a. Write the symbols next to each other If it does not equals ZERO 4 b. Cross the oxidation numbers and write as subscripts on the opposite symbol (drop the “+” and “ —”)

Special Notes • If a two or three is being applied to a polyatomic ion, parenthesis goes around the polyatomic ion and the subscript outside of it

Practice Problem #1 #2 Mg & Cl Li & F

Practice Problem #3 #4 Potassium Oxide Ca & SO 4

Practice Problem #5 #6 Na & PO 4 Mg & Cl. O 3

Practice Problem #7 #8 NH 4 & NO 3 Lithium Acetate
