Modelbased mechanical ventilation managing pulmonary disease using computation

Model-based mechanical ventilation: managing pulmonary disease using computation J. Geoffrey Chase With major input from: Jennifer Dickson, Sophie Morton, Sarah Howe, KT Kim, Oliver Kanangara and Geoffrey M. Shaw

Mechanical Ventilation n Mechanical ventilation (MV) provides pressure and airflow to the lungs to support the work of breathing, maintain patient condition, and thus allow recovery from underlying condition n No consistent use worldwide; practice depends on local context and clinician experience n MV settings are clinically determined n Based first on experience and then on patient response n Patients are all different n Trial and error can result in injury - want to be able to tailor MV to individual patients to quickly optimise care and outcome
Mechanical Ventilation
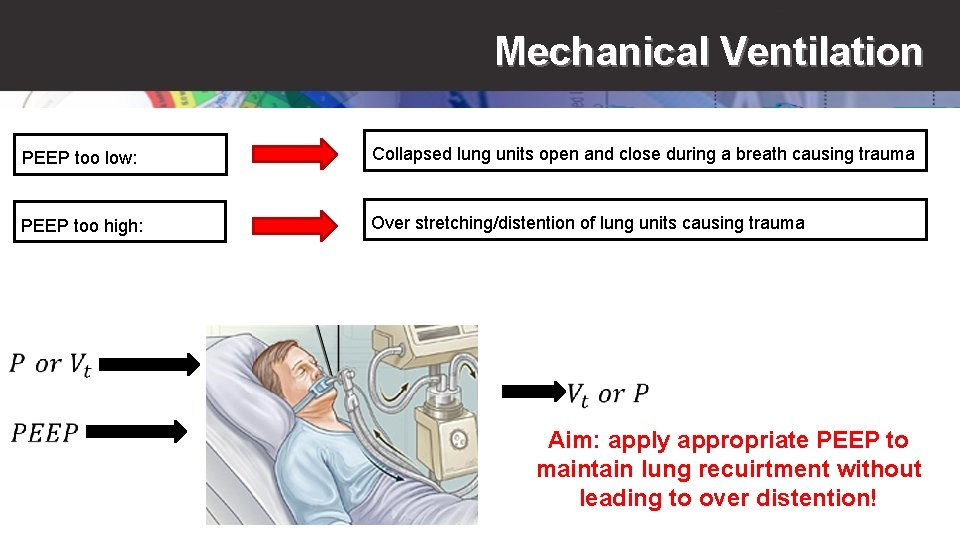
Mechanical Ventilation PEEP too low: Collapsed lung units open and close during a breath causing trauma PEEP too high: Over stretching/distention of lung units causing trauma Aim: apply appropriate PEEP to maintain lung recuirtment without leading to over distention!

The Goal? n Can we use breathing – in a range of settings to identify / find patient-specific lung mecahnics n Underlying elastance (1 / compliance) and/or resistance can tell us a lot about patient condition n There is a great deal of work suggesting minimum elastance is the optimal point to apply MV But, how to find it? ?

This should be easy but… n “Humans are horribly variable” (Dickson et al, 2014)

So, really we need models to… n Manage inter- and intra- patient variability in response to care (and over time)… A lot of variables to consider n Disease and response is patient-specific n Clinicians rely on intuitions and experience to set MV (PEEP) ¨ Relatively little non invasive assessment methods to assess lung condition – so how do we know what is happening?

We have a number of model results (w/ clinical data) n Minimal Model, Gaussian Distribution model (Sundaresan et al. , 2009, 2010, Chiew et al. , 2012, Redmond et al. 2014) n Functional Residual Capacity Estimation (Sundaresan et al. , 2010, Mishra et al. , 2013, van Drunen et al. , 2013) n Pressure Recruitment and Multi Compartment Models (Schranz n et al. , 2011, Docherty et al. , 2013) Patient-Ventilator Interaction, Asynchrony Analysis (Moorhead et al. , 2012, Chiew et al. , 2013) n Expiratory Time Constant Model (van Drunen et al. , 2013) n Time-Varying Elastance Model (van Drunen et al. , 2013, Chiew et al. , 2014) n Airway Branching Model (Damanhuri et al. , 2014) n Nonlinear Auto Regressive Modelling (Langdon et al. , 2015)

Background: Elastance n

Modelling Approach n Utilising real clinical data to better understand the underlying mechanics and variability in real patients n Use basis functions to describe the shapes of physiologically expected results. E. g. exponential for tissue relaxation n Compilation and comparison of inter patient differences

A major Problem Statement n Overall Goal: identify patient-specific PEEP n Supporting Goal: Can we use models to predict how the lung will respond at different PEEP? ¨ ¨ Can I predict what changing the positive pressure support (PEEP) will do to pressure and peak pressure in a breath will do before I do it? I. e. will it hurt the patient or not? Always good to know first!!! Can the prediction use basis functions that “mean something“ and relate to physiological behaviour?

Single-Compartment Lung Model V(t) Rrs Q(t), Paw(t) Ers P(t)

Dynamic Elastance n Elastance follows a pattern over a breath for different ventilation modes n These dynamic patterns can be broken into contributing shapes through clinically relevant basis function methods n Allows study of mechanics of breathing in terms of relevant anatomy and physiology Edrs in Spontaneous Breathing

Ventilation Modes n This Investigation focuses on volume controlled ventilation n n Different modes of ventilation will require different clinically relevant basis functions Volume Controlled n A set target volume is delivered n Pressure allowed to vary (up to a PIP) to deliver that Vt

Predicting the Future n Broken into 2 basis functions + Resistance: n “Recruitment” n “Distension”
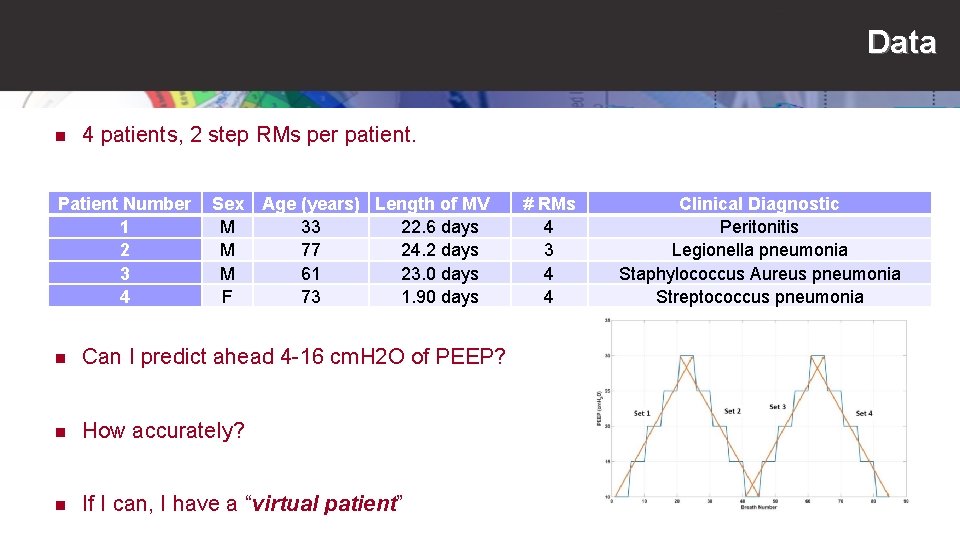
Data n 4 patients, 2 step RMs per patient. Patient Number 1 2 3 4 Sex M M M F Age (years) Length of MV 33 22. 6 days 77 24. 2 days 61 23. 0 days 73 1. 90 days n Can I predict ahead 4 -16 cm. H 2 O of PEEP? n How accurately? n If I can, I have a “virtual patient” # RMs 4 3 4 4 Clinical Diagnostic Peritonitis Legionella pneumonia Staphylococcus Aureus pneumonia Streptococcus pneumonia

Fit to Data n Fit is excellent at all PEEP levels within 2 -5% on pressure n Patients 1 -4 shown (rows) n PEEP rises with column n So far, so good, a good model, but can it predict?

Prediction n Results generally within 10% error on PIP and pressure and much lower for smaller PEEP steps n No one else can do this!
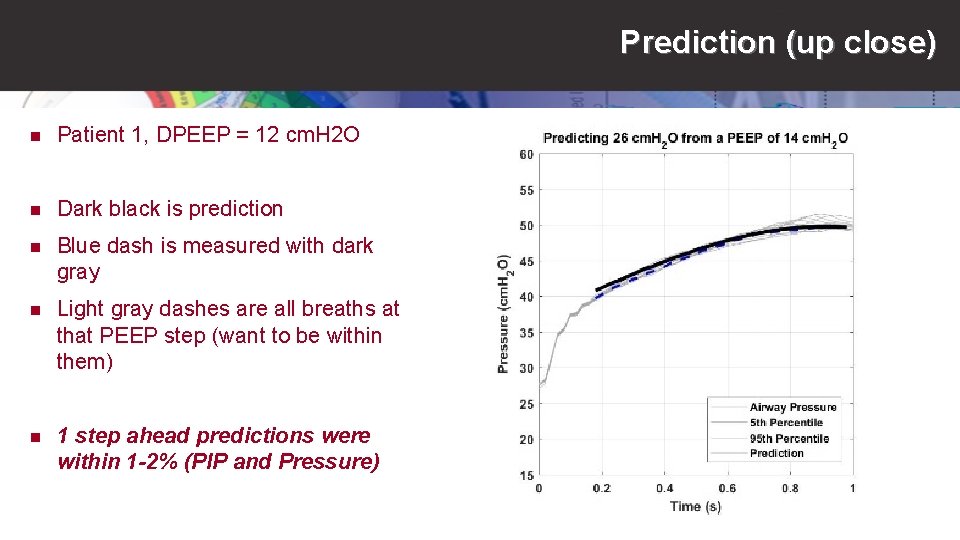
Prediction (up close) n Patient 1, DPEEP = 12 cm. H 2 O n Dark black is prediction n Blue dash is measured with dark gray n Light gray dashes are all breaths at that PEEP step (want to be within them) n 1 step ahead predictions were within 1 -2% (PIP and Pressure)

Clinical Implications n We can now predict whether a higher PEEP will give better care without having the risk of physically increasing the PEEP to check. n Being able to predict behaviours without risking damage is the centre point of many computational methods (particularly in application to clinical medicine). Answer: A method to forward predict has been created and validated allowing better safety in changing PEEP in volume controlled MV

CURE and Virtual Patients n CURE = Clinical Use of Respiratory Elastance ¨ n Virtual patients = Predictive Capability (what will happen when I change my inputs? ) ¨ ¨ ¨ n Major RCT commencing in Christchurch Hospital ICU Demonstrated and well-validated in glycemic (blood glucose) control Now validated here Used to make models patient specific and thus to guide (patient specific) care We now have this capability in mechanical ventilation and thus for pulmonary mechanics. ¨ Where else can we use this model? ?
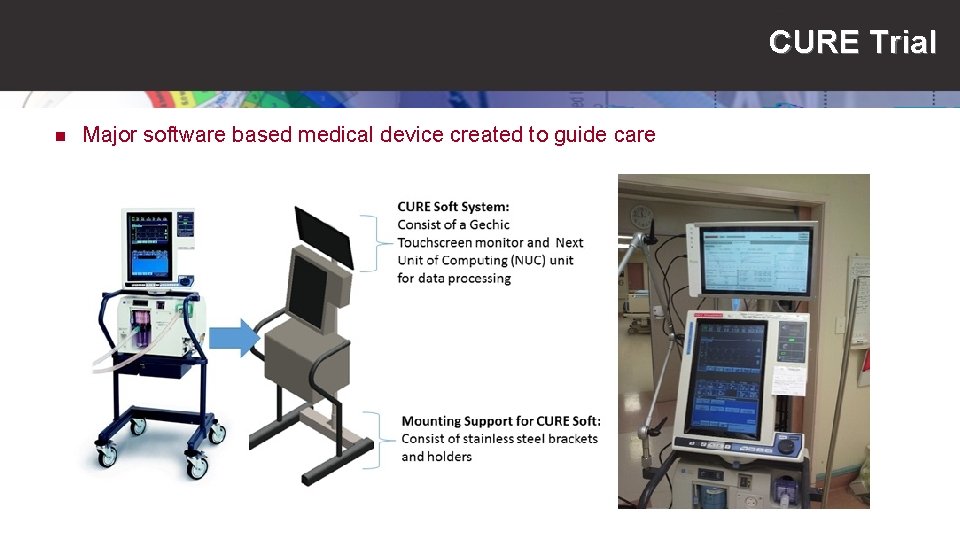
CURE Trial n Major software based medical device created to guide care

Neonatal MV n Mechanical ventilation for neonates is much less studied than for adults n Thus, neonates are often treated like small adults § If this doesn’t work High Frequency Oscillatory Ventilation is tried, which is even less studied n As young as 24 weeks gestational age n Can weigh 600 -2000 g n Underdeveloped lungs and physiology n Probably neonates ≠ Small Adults

Specific NICU Differences and Challenges n Smaller, less developed lungs and tissues n Surfactant deficiency n Currently, little is known about neonatal lung mechanics, or the mechanics of specific lung tissues (Neonates = Small Adults? ) § Mechanics likely different for different tissue development

Well, the model works! n Fitting model to ventilator driven inspiration n Fits well with the neonatal data Model captures all apparent measured data in NICU MV No need to optimise model or change dynamics Neonates “may be” small adults?

First Thought Adults ≠ Infants Neonates are not small adults!
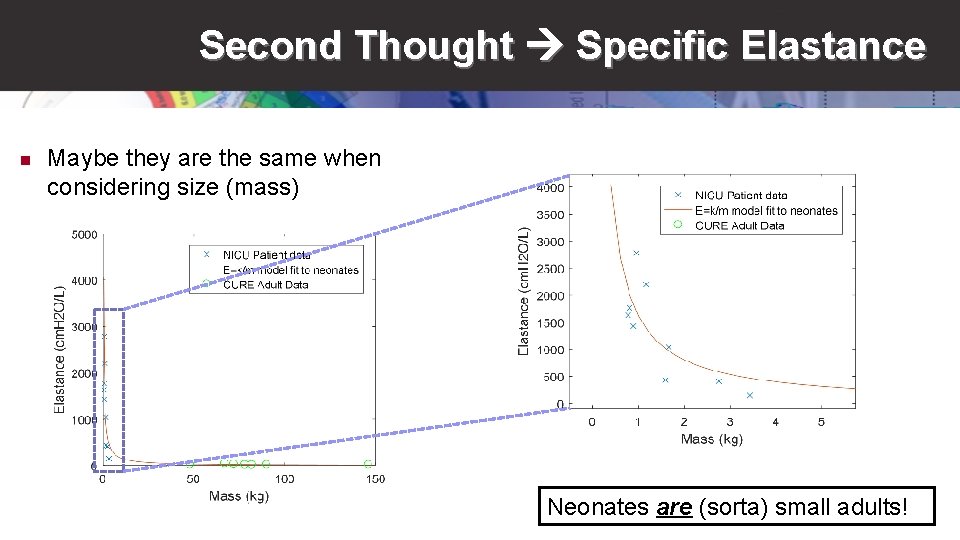
Second Thought Specific Elastance n Maybe they are the same when considering size (mass) Neonates are (sorta) small adults!

Conclusions and Clinical Implications n Clearly, infant lungs do not behave entirely in the same way as adult lungs, but the model still works well without changes n n Neonates will however likely have to be treated differently than adults in using MV More work is needed to asses relative contribution of smaller tidal volumes to the higher elastances, and contribution of tissue stiffness and alveolar immaturity. n This study introduces the possibility of using model-based methods to develop patient-specific MV treatment in the NICU where “one size fits all”

What about out-patients? COPD et al n COPD and asthma cost ~0. 5 -1. 0% GDP in western countries n Mainly assessed by “ability to breathe out” Peak flow tests = maximum power ¨ ¨ These test maximum power but not average “walking around” power which is critical to quality of life Peak power is not a good representation of avg power or endurance in any sport, why should it be here? n How to test this “average power”? n Can the model help? n Initial results are developing a mask technology worn by out-patients which restricts expiration flow temporarily to allow the model to capture expiratory elastance or the ability to breathe out ¨ Measure over time to monitor out-patient therapy and interventions
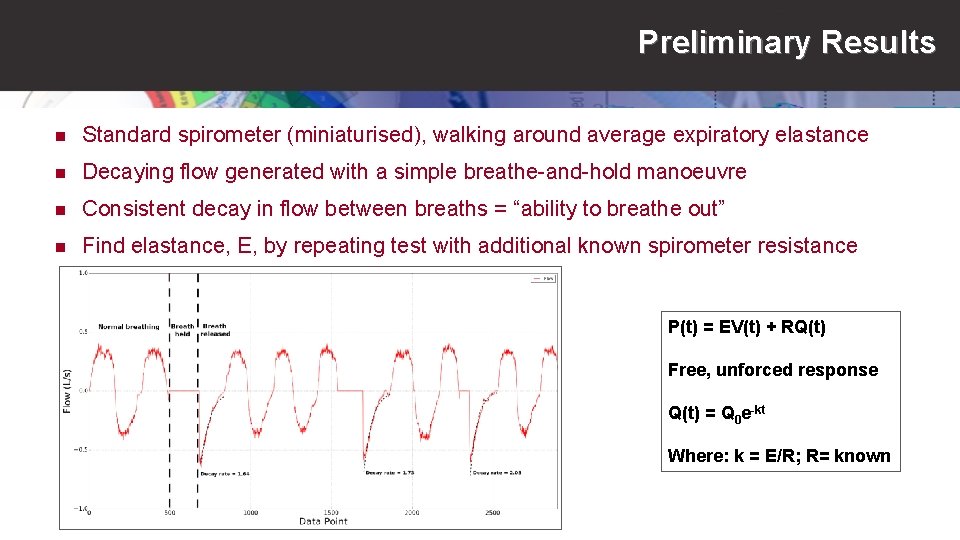
Preliminary Results n Standard spirometer (miniaturised), walking around average expiratory elastance n Decaying flow generated with a simple breathe-and-hold manoeuvre n Consistent decay in flow between breaths = “ability to breathe out” n Find elastance, E, by repeating test with additional known spirometer resistance P(t) = EV(t) + RQ(t) Free, unforced response Q(t) = Q 0 e-kt Where: k = E/R; R= known

Clinical Implications n No one ever considers expiration to be of real value n Goal is a more natural measurement that can be done at home and analysed independently Reducing cost and clinical time n Could obtain everyday monitoring in just 1 -2 mins per day of mask (or less) minimum invasiveness on life ¨ n Could likely dose inhalers of some sort simultaneously (just a technology question) Can we improve monitoring, make the metrics more representative of real life demands (peak power is not a good representation of avg power or endurance in any sport), and thus better allocate care and resources?

A brief pause reflection… A brief pause forforreflection…

Overall (Clinical) Implications n Very simple models with data measured in real-time can offer significant physiological insight into patient condition and the impact (over time) of interventions n Virtual patients are possible and the first such in-silico models have been generated ¨ Potential to guide care and directly manage variability as in glycemic control (see ongoing work in ICU and type 2 diabetes with NZ National Science Challenges) n Patient-specific or Personalised care is eminently possible n So is automated care, which would increase productivity and equity of access ¨ n I. e. better outcomes at reduced cost The future is not biology, but silicon + biology?

Acknowledgements n Centre of Bioengineering (University of Canterbury, New Zealand) n GIGA Cardiovascular Science (University of Liege, Belgium) n Swiss-Belgium NAVA ventilation Research Group (Belgium, Switzerland) n Institute of Technical Medicine (Furtwangen University, Germany) (i. e. it’s all me!!)
- Slides: 34