MO Diagrams for More Complex Molecules Chapter 5


- Slides: 11

MO Diagrams for More Complex Molecules Chapter 5 Wednesday, October 14, 2015

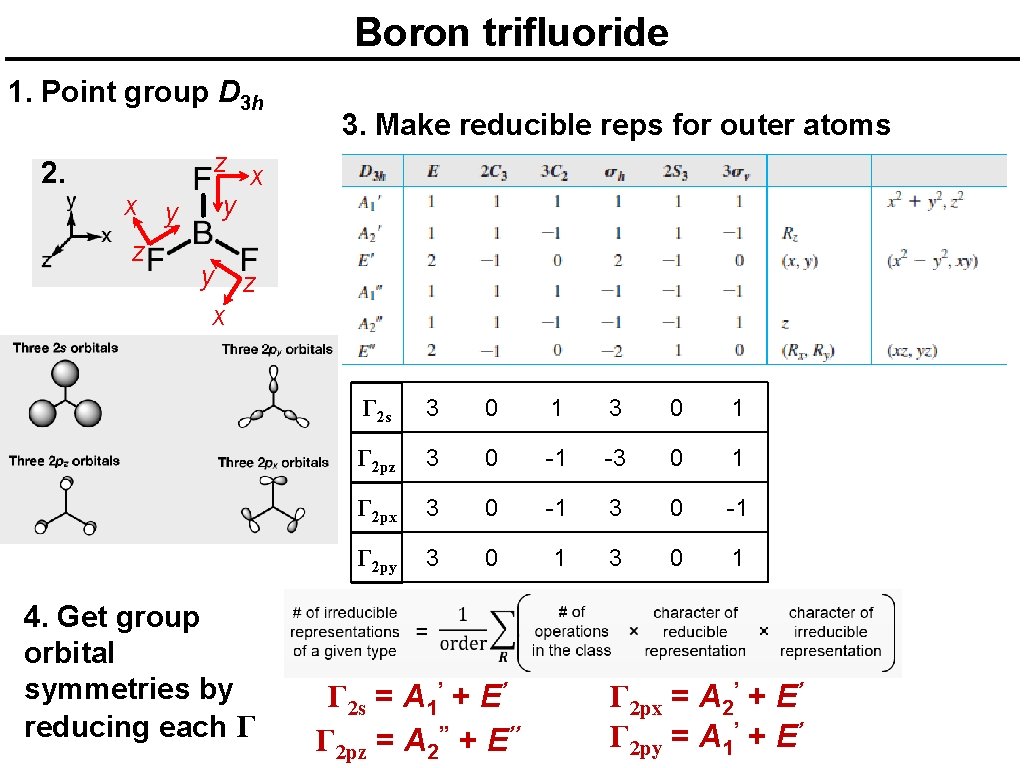
Boron trifluoride 1. Point group D 3 h 2. x y z 3. Make reducible reps for outer atoms z x y y z x 4. Get group orbital symmetries by reducing each Γ Γ 2 s 3 0 1 3 0 1 Γ 2 pz 3 0 -1 -3 0 1 Γ 2 px 3 0 -1 3 0 -1 Γ 2 py 3 0 1 3 0 1 Γ 2 s = A 1’ + E’ Γ 2 pz = A 2’’ + E’’ Γ 2 px = A 2’ + E’ Γ 2 py = A 1’ + E’

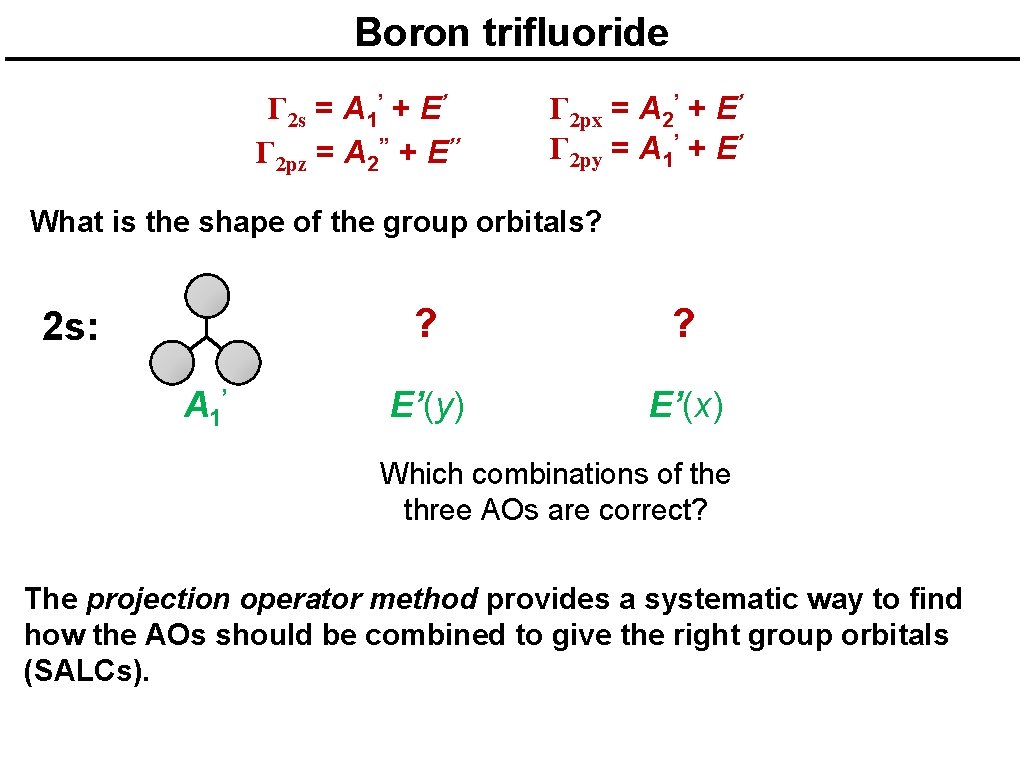
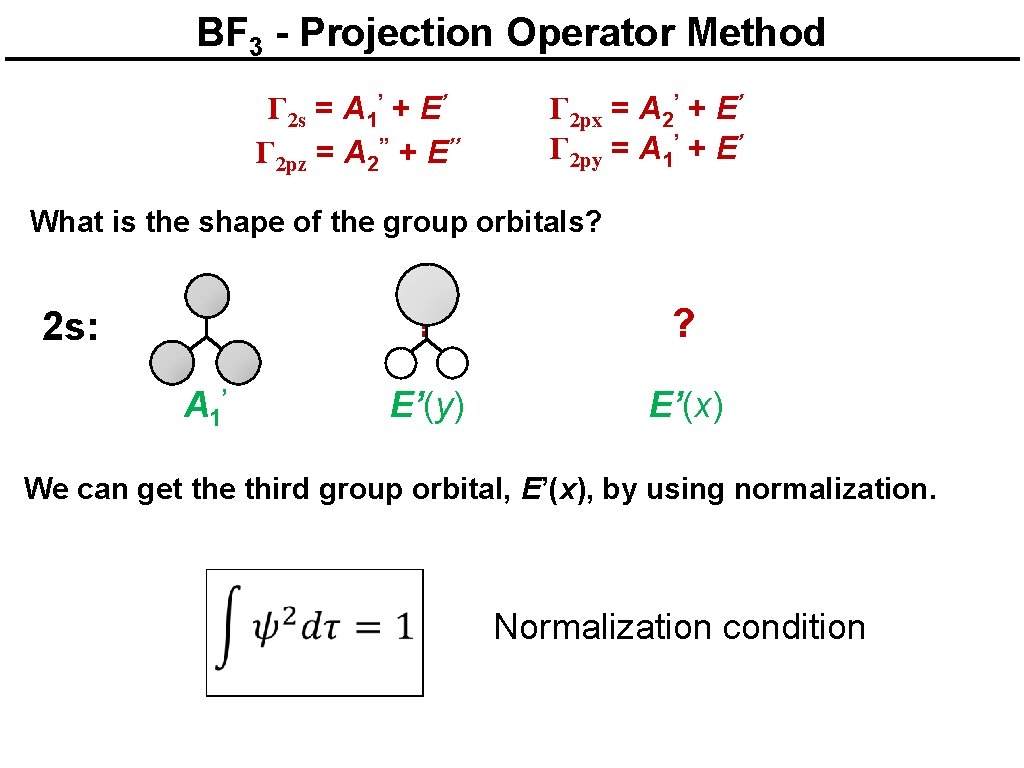
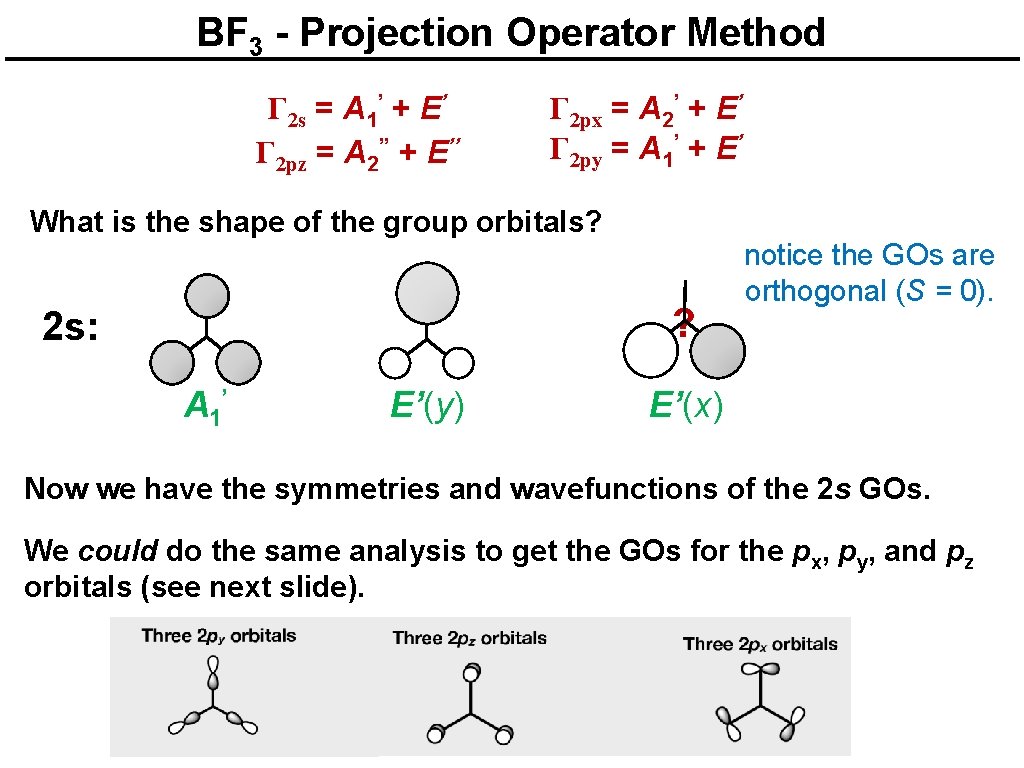
Boron trifluoride Γ 2 s = A 1’ + E’ Γ 2 pz = A 2’’ + E’’ Γ 2 px = A 2’ + E’ Γ 2 py = A 1’ + E’ What is the shape of the group orbitals? 2 s: A 1’ ? ? E’(y) E’(x) Which combinations of the three AOs are correct? The projection operator method provides a systematic way to find how the AOs should be combined to give the right group orbitals (SALCs).

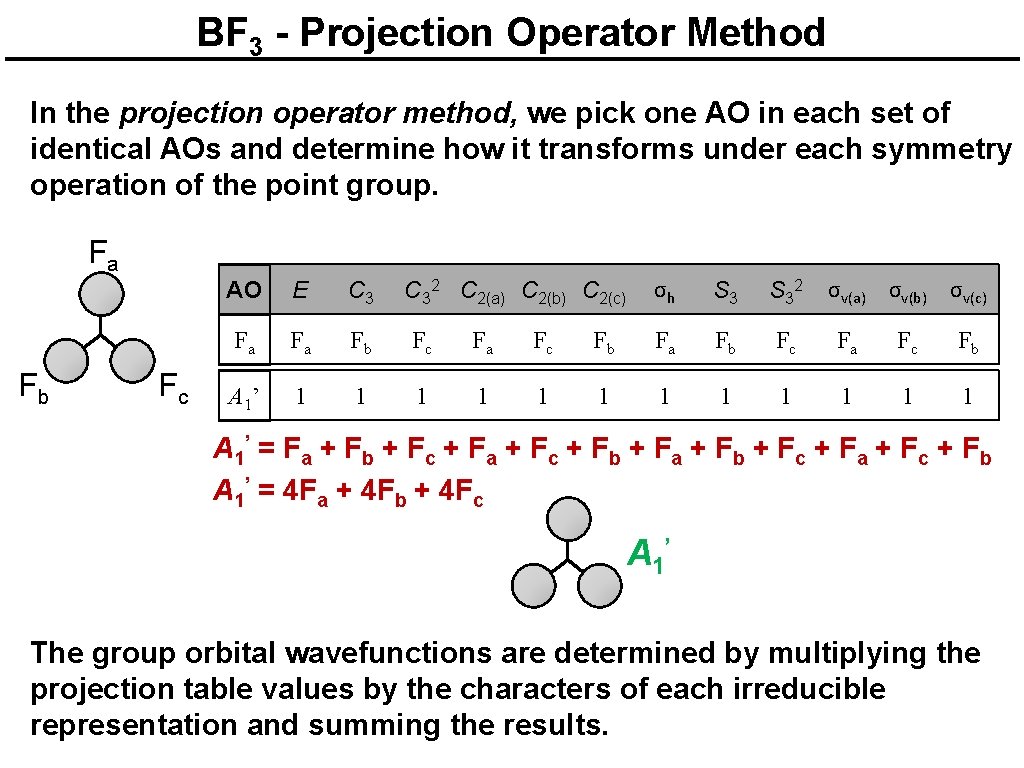
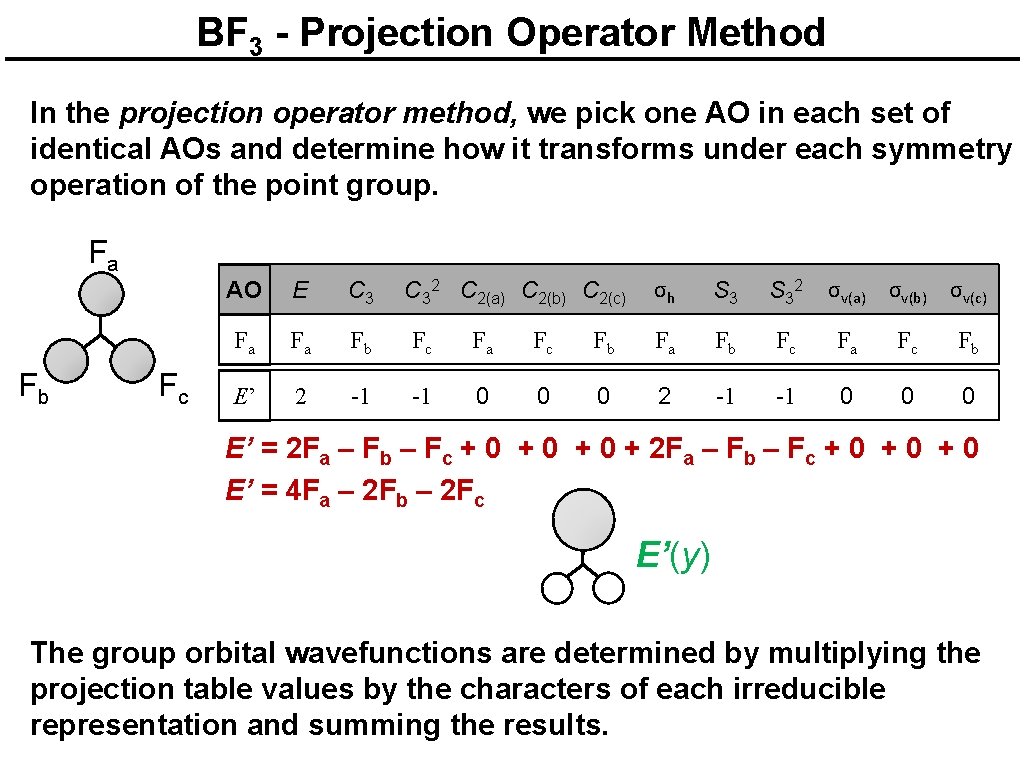
BF 3 - Projection Operator Method In the projection operator method, we pick one AO in each set of identical AOs and determine how it transforms under each symmetry operation of the point group. F a F b Fc AO E C 32 C 2(a) C 2(b) C 2(c) σh S 32 σv(a) σv(b) σv(c) Fa Fa Fb Fc Fa Fc Fb A 1’ 1 1 1 A 1’ = F a + F b + F c + F a + F c + F b + F a + F b + F c + F a + F c + F b A 1’ = 4 Fa + 4 Fb + 4 Fc A 1’ The group orbital wavefunctions are determined by multiplying the projection table values by the characters of each irreducible representation and summing the results.

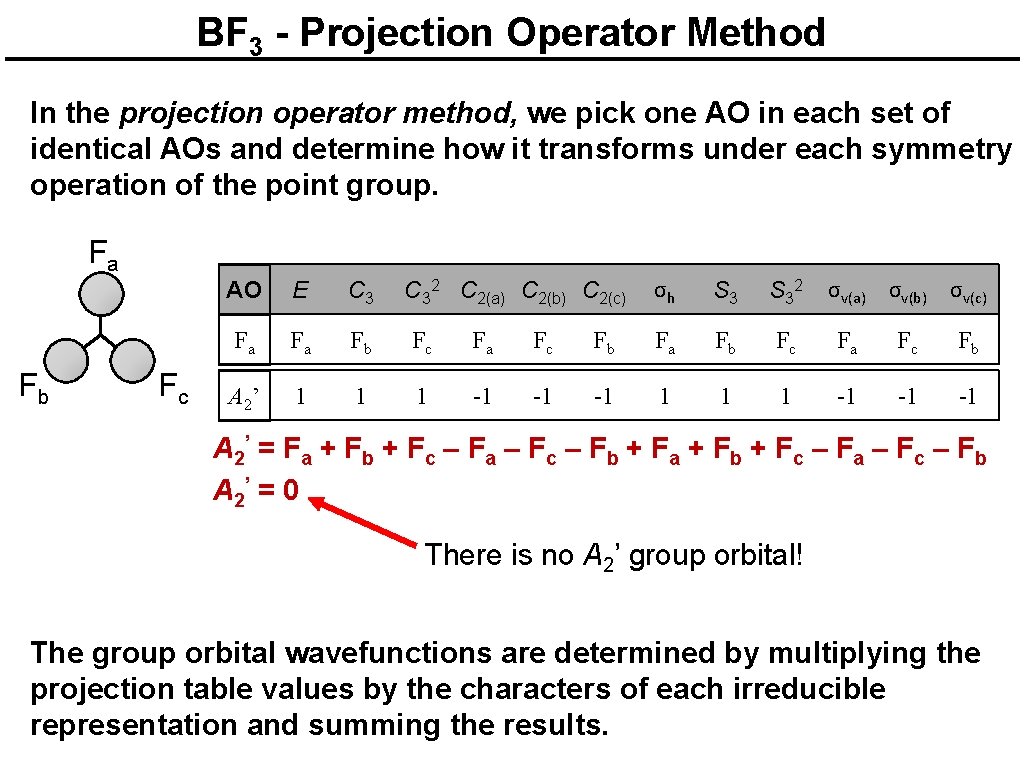
BF 3 - Projection Operator Method In the projection operator method, we pick one AO in each set of identical AOs and determine how it transforms under each symmetry operation of the point group. F a F b Fc AO E C 32 C 2(a) C 2(b) C 2(c) σh S 32 σv(a) σv(b) σv(c) Fa Fa Fb Fc Fa Fc Fb A 2’ 1 1 1 -1 -1 -1 A 2’ = F a + F b + F c – F a – F c – F b + F a + F b + F c – F a – F c – F b A 2’ = 0 There is no A 2’ group orbital! The group orbital wavefunctions are determined by multiplying the projection table values by the characters of each irreducible representation and summing the results.

BF 3 - Projection Operator Method In the projection operator method, we pick one AO in each set of identical AOs and determine how it transforms under each symmetry operation of the point group. F a F b Fc AO E C 32 C 2(a) C 2(b) C 2(c) σh S 32 σv(a) σv(b) σv(c) Fa Fa Fb Fc Fa Fc Fb E’ 2 -1 -1 0 0 0 E’ = 2 Fa – Fb – Fc + 0 + 0 + 2 Fa – Fb – Fc + 0 + 0 E’ = 4 Fa – 2 Fb – 2 Fc E’(y) The group orbital wavefunctions are determined by multiplying the projection table values by the characters of each irreducible representation and summing the results.

BF 3 - Projection Operator Method Γ 2 s = A 1’ + E’ Γ 2 pz = A 2’’ + E’’ Γ 2 px = A 2’ + E’ Γ 2 py = A 1’ + E’ What is the shape of the group orbitals? 2 s: A 1’ ? ? E’(y) E’(x) We can get the third group orbital, E’(x), by using normalization. Normalization condition

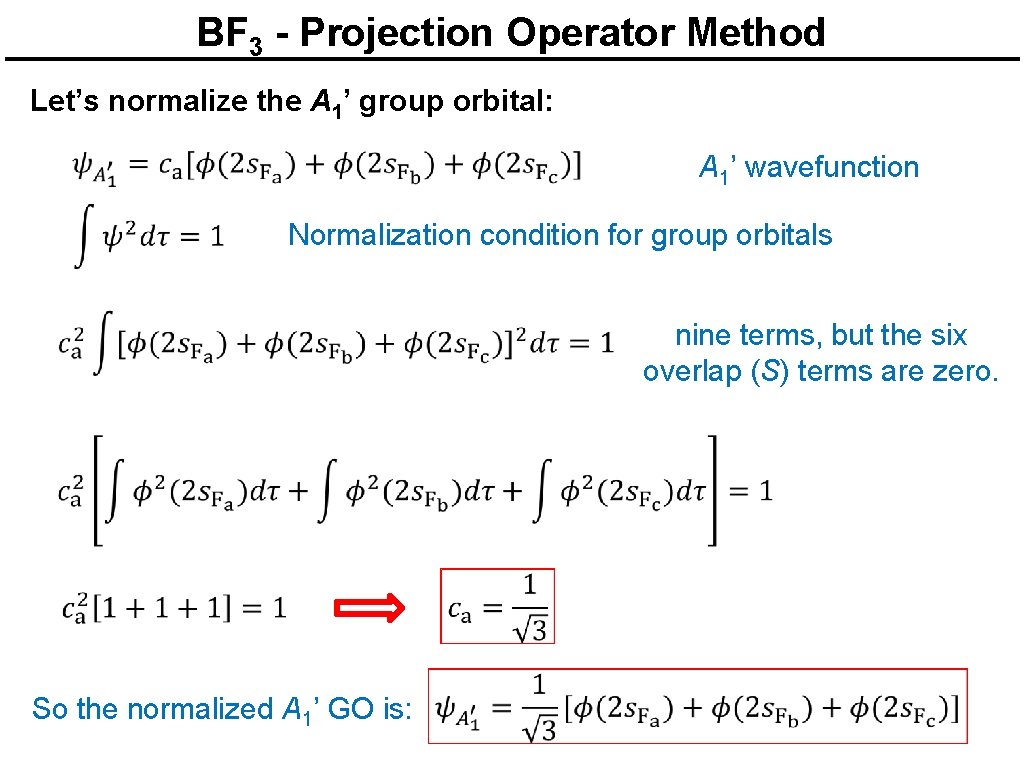
BF 3 - Projection Operator Method Let’s normalize the A 1’ group orbital: A 1’ wavefunction Normalization condition for group orbitals nine terms, but the six overlap (S) terms are zero. So the normalized A 1’ GO is:

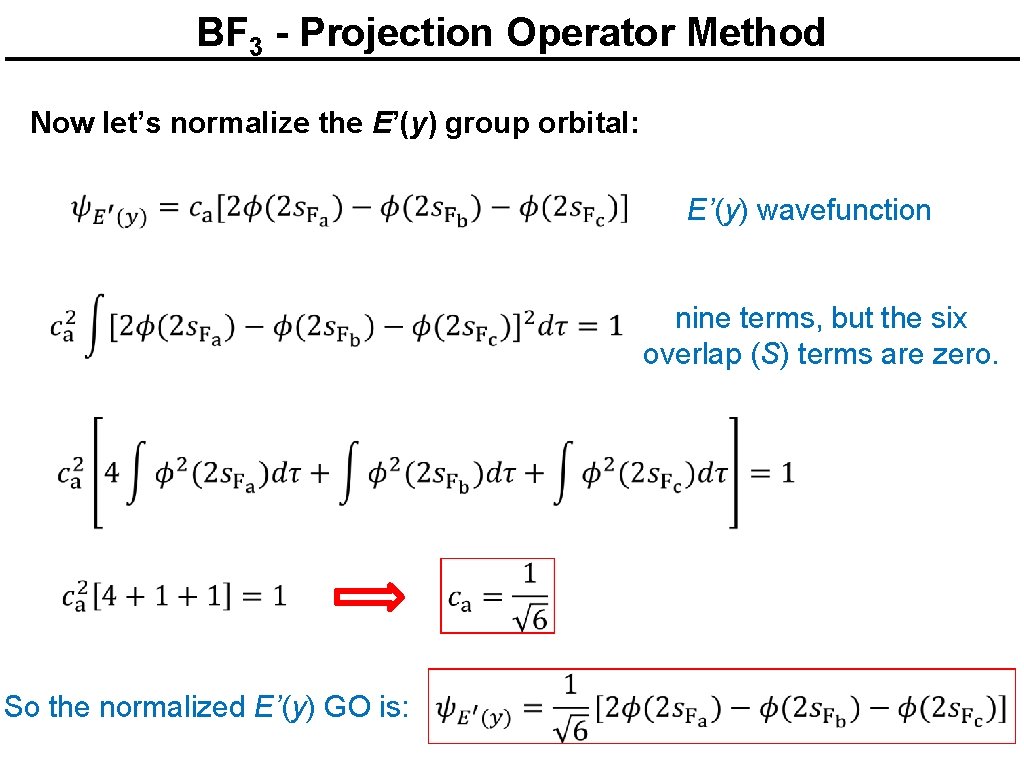
BF 3 - Projection Operator Method Now let’s normalize the E’(y) group orbital: E’(y) wavefunction nine terms, but the six overlap (S) terms are zero. So the normalized E’(y) GO is:

BF 3 - Projection Operator Method So the normalized E’(x) GO is:

BF 3 - Projection Operator Method Γ 2 s = A 1’ + E’ Γ 2 pz = A 2’’ + E’’ Γ 2 px = A 2’ + E’ Γ 2 py = A 1’ + E’ What is the shape of the group orbitals? ? 2 s: A 1’ E’(y) notice the GOs are orthogonal (S = 0). E’(x) Now we have the symmetries and wavefunctions of the 2 s GOs. We could do the same analysis to get the GOs for the px, py, and pz orbitals (see next slide).