Mm wave and ab initio studies of the

Mm wave and ab initio studies of the conformational landscape of METHOXYPHENOLS, identified as SOA precursors � Presented by: Atef JABRI Post-doctorant at LPCA Lab, Dunkirk, France International Symposium on Molecular Spectroscopy CONFORMERS AND ISOMERS
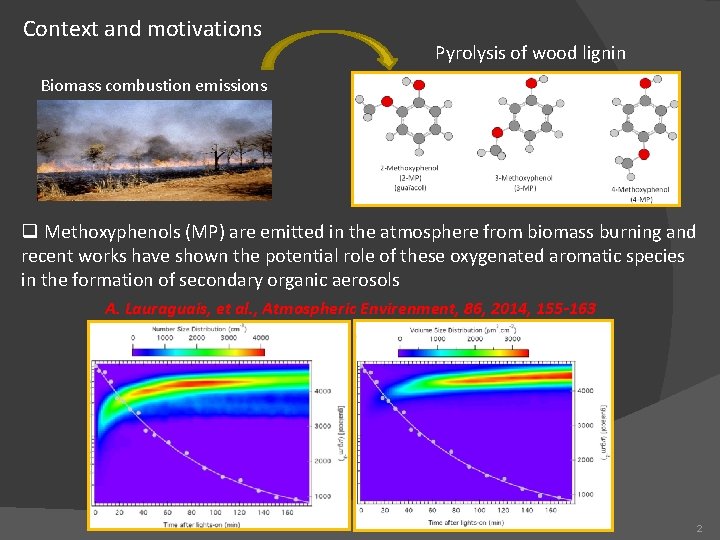
Context and motivations Pyrolysis of wood lignin Biomass combustion emissions q Methoxyphenols (MP) are emitted in the atmosphere from biomass burning and recent works have shown the potential role of these oxygenated aromatic species in the formation of secondary organic aerosols A. Lauraguais, et al. , Atmospheric Envirenment, 86, 2014, 155 -163 2
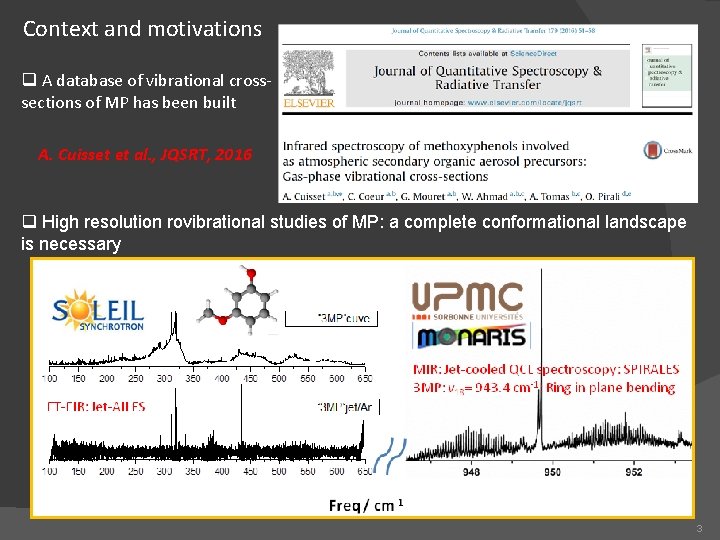
Context and motivations q A database of vibrational crosssections of MP has been built A. Cuisset et al. , JQSRT, 2016 q High resolution rovibrational studies of MP: a complete conformational landscape is necessary 3
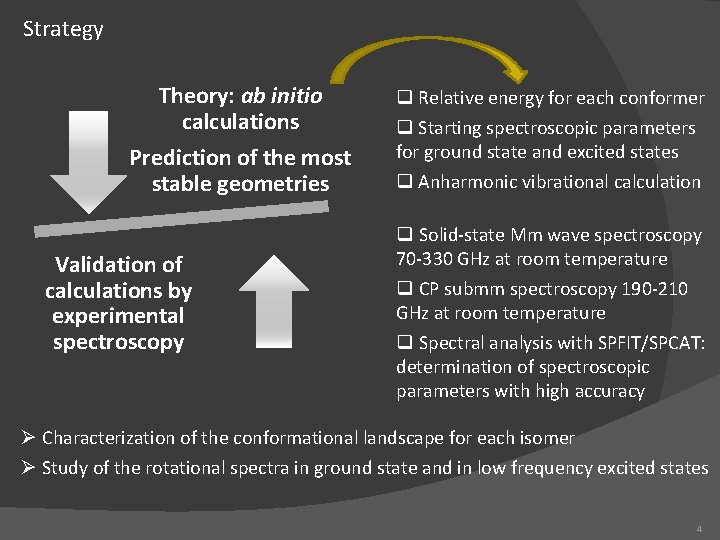
Strategy Theory: ab initio calculations Prediction of the most stable geometries Validation of calculations by experimental spectroscopy q Relative energy for each conformer q Starting spectroscopic parameters for ground state and excited states q Anharmonic vibrational calculation q Solid-state Mm wave spectroscopy 70 -330 GHz at room temperature q CP submm spectroscopy 190 -210 GHz at room temperature q Spectral analysis with SPFIT/SPCAT: determination of spectroscopic parameters with high accuracy Ø Characterization of the conformational landscape for each isomer Ø Study of the rotational spectra in ground state and in low frequency excited states 4
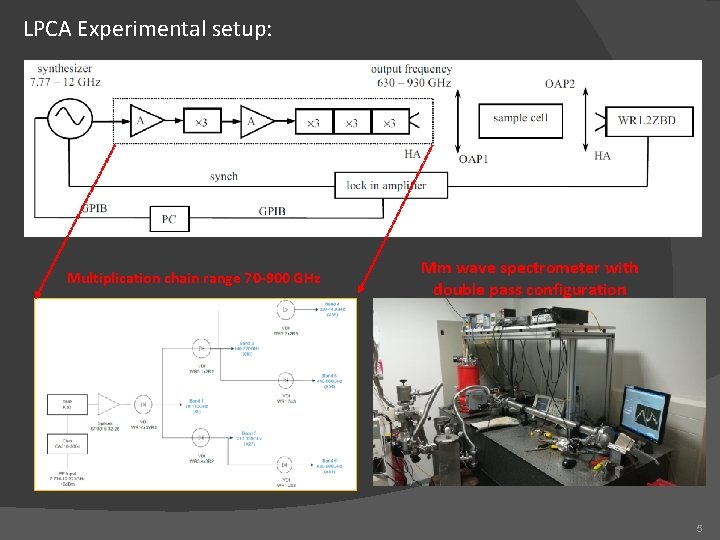
LPCA Experimental setup: Multiplication chain range 70 -900 GHz Mm wave spectrometer with double pass configuration 5
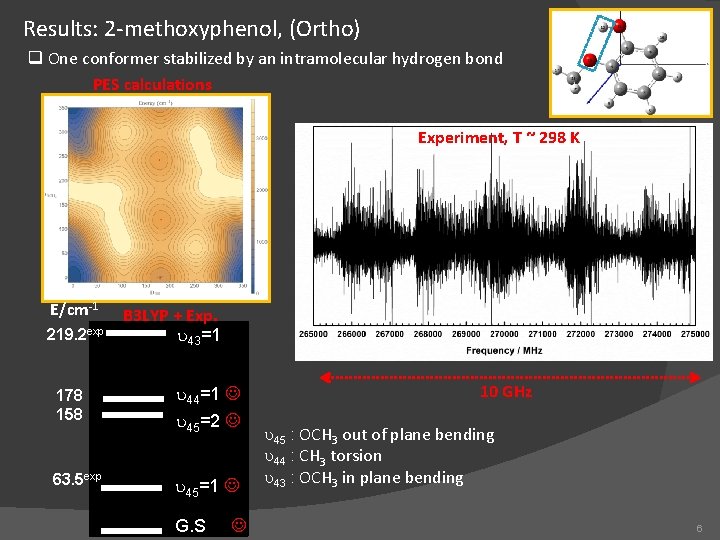
Results: 2 -methoxyphenol, (Ortho) q One conformer stabilized by an intramolecular hydrogen bond PES calculations Experiment, T ~ 298 K E/cm-1 219. 2 exp B 3 LYP + Exp. u 43=1 178 158 u 44=1 63. 5 exp u 45=1 u 45=2 G. S 10 GHz u 45 : OCH 3 out of plane bending u 44 : CH 3 torsion u 43 : OCH 3 in plane bending 6
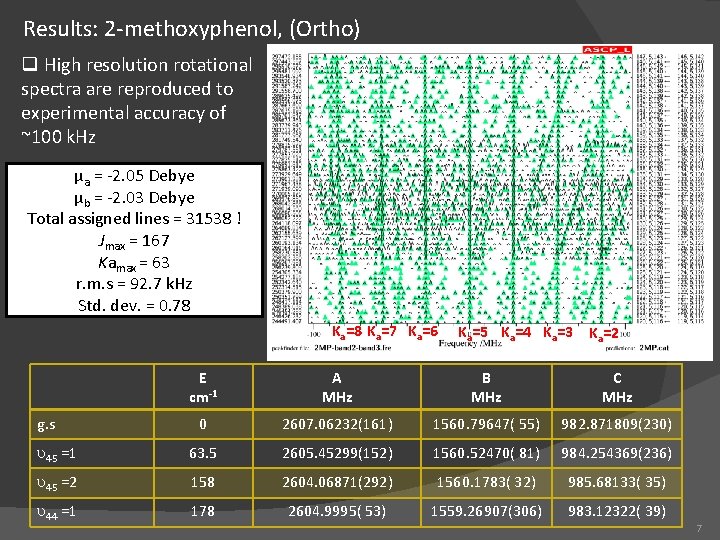
Results: 2 -methoxyphenol, (Ortho) q High resolution rotational spectra are reproduced to experimental accuracy of ~100 k. Hz µa = -2. 05 Debye µb = -2. 03 Debye Total assigned lines = 31538 ! Jmax = 167 Kamax = 63 r. m. s = 92. 7 k. Hz Std. dev. = 0. 78 Ka=7 Ka=6 Ka=5 Ka=4 Ka=3 Ka=2 E cm-1 A MHz B MHz C MHz 0 2607. 06232(161) 1560. 79647( 55) 982. 871809(230) u 45 =1 63. 5 2605. 45299(152) 1560. 52470( 81) 984. 254369(236) u 45 =2 158 2604. 06871(292) 1560. 1783( 32) 985. 68133( 35) u 44 =1 178 2604. 9995( 53) 1559. 26907(306) 983. 12322( 39) g. s 7
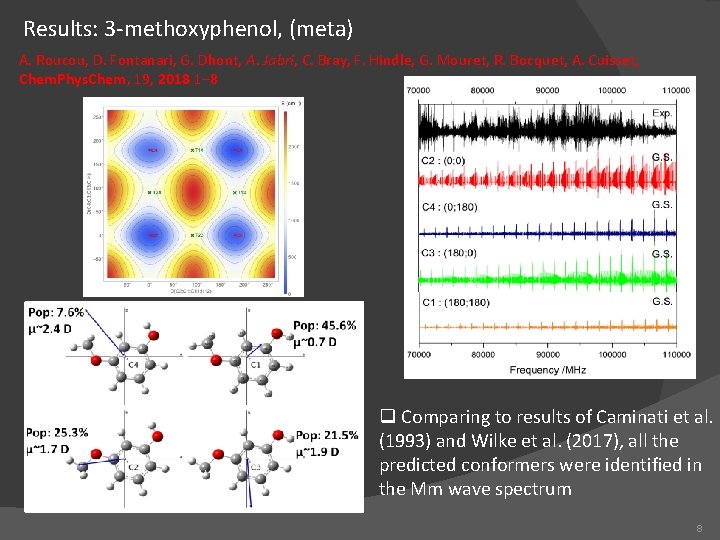
Results: 3 -methoxyphenol, (meta) A. Roucou, D. Fontanari, G. Dhont, A. Jabri, C. Bray, F. Hindle, G. Mouret, R. Bocquet, A. Cuisset, Chem. Phys. Chem, 19, 2018 1– 8 q Comparing to results of Caminati et al. (1993) and Wilke et al. (2017), all the predicted conformers were identified in the Mm wave spectrum 8
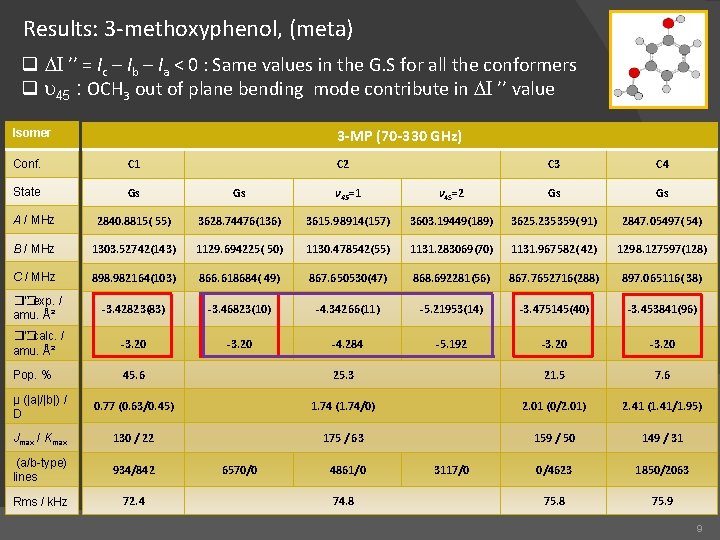
Results: 3 -methoxyphenol, (meta) q DI ’’ = Ic – Ib – Ia < 0 : Same values in the G. S for all the conformers q u 45 : OCH 3 out of plane bending mode contribute in DI ’’ value 3 -MP (70 -330 GHz) Isomer Conf. C 1 C 2 C 3 C 4 State Gs Gs v 45=1 v 45=2 Gs Gs A / MHz 2840. 8815( 55) 3628. 74476(136) 3615. 98914(157) 3603. 19449(189) 3625. 235359( 91) 2847. 05497( 54) B / MHz 1303. 52742(143) 1129. 694225( 50) 1130. 478542(55) 1131. 283069(70) 1131. 967582( 42) 1298. 127597(128) C / MHz 898. 982164(103) 866. 618684( 49) 867. 650530(47) 868. 692281(56) 867. 7652716(288) 897. 065116( 38) �� I’’ exp. / amu. Ų -3. 42823(83) -3. 46823(10) -4. 34266(11) -5. 21953(14) -3. 475145(40) -3. 453841(96) �� I’’ calc. / amu. Ų -3. 20 -4. 284 -5. 192 -3. 20 Pop. % 45. 6 25. 3 21. 5 7. 6 µ (|a|/|b|) / D 0. 77 (0. 63/0. 45) 1. 74 (1. 74/0) 2. 01 (0/2. 01) 2. 41 (1. 41/1. 95) Jmax / Kmax 130 / 22 175 / 63 159 / 50 149 / 31 (a/b-type) lines 934/842 0/4623 1850/2063 Rms / k. Hz 72. 4 75. 8 75. 9 6570/0 4861/0 74. 8 3117/0 9
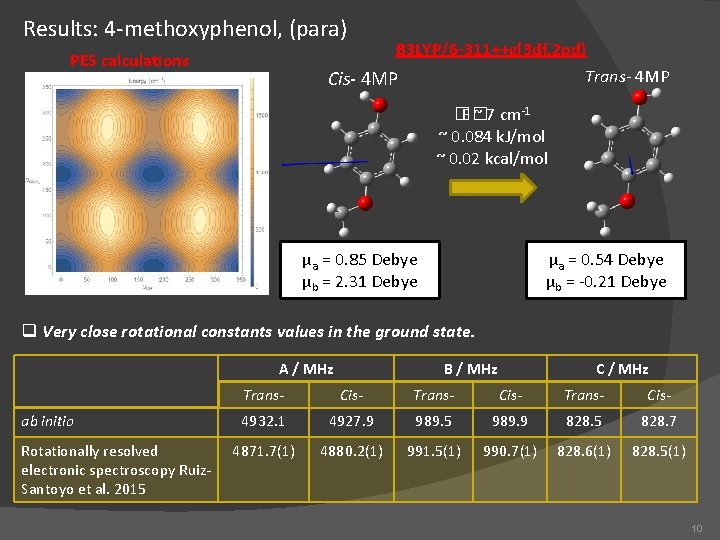
Results: 4 -methoxyphenol, (para) PES calculations B 3 LYP/6 -311++g(3 df, 2 pd) Trans- 4 MP Cis- 4 MP �� E ~ 7 cm-1 ~ 0. 084 k. J/mol ~ 0. 02 kcal/mol µa = 0. 85 Debye µb = 2. 31 Debye µa = 0. 54 Debye µb = -0. 21 Debye q Very close rotational constants values in the ground state. A / MHz ab initio Rotationally resolved electronic spectroscopy Ruiz. Santoyo et al. 2015 B / MHz C / MHz Trans- Cis- 4932. 1 4927. 9 989. 5 989. 9 828. 5 828. 7 4871. 7(1) 4880. 2(1) 991. 5(1) 990. 7(1) 828. 6(1) 828. 5(1) 10

Results: 4 -methoxyphenol, (para) q High resolution analysis of the rotational spectrum of cis - 4 MP Ground state + u 45=1 Total assigned lines = 8683 Jmax = 195 Kamax = 41 r. m. s = 80. 6 k. Hz Std. dev. = 0. 81 E/cm-1 Excited states 164 153 82 u 45=2 u 44=1 Boltz. pop. = 0. 45 + Boltz. pop. = 0. 48 u 45=1 Boltz. Pop. = 0. 67 G. S Boltz. Pop. = 1 Trans- conformer of 4 MP µa = 0. 54 Debye, µb = -0. 21 Debye Structure 1 Structure 2 A = 4872. 3599730(32) MHz B = 990. 7651386(43) MHz C = 828. 84446100(52) MHz A = 4843. 134345(34) MHz B = 991. 3820658(40) MHz C = 830. 39654625( 32) MHz 11
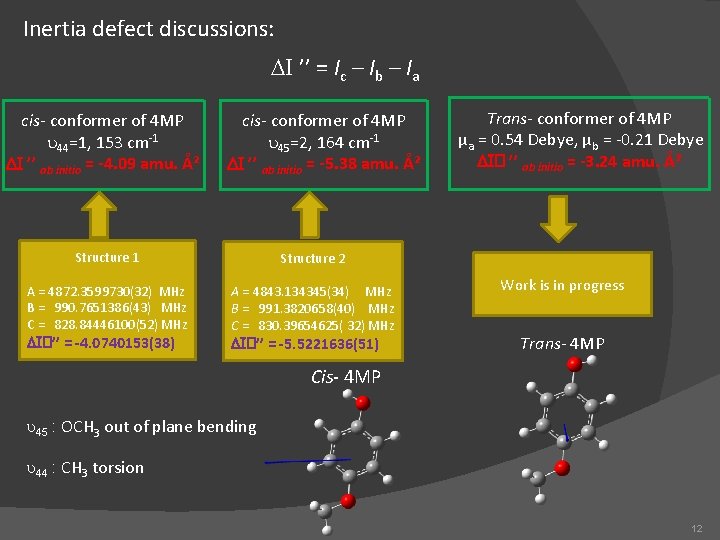
Inertia defect discussions: DI ’’ = Ic – Ib – Ia cis- conformer of 4 MP u 44=1, 153 cm-1 DI ’’ ab initio = -4. 09 amu. Ų cis- conformer of 4 MP u 45=2, 164 cm-1 DI ’’ ab initio = -5. 38 amu. Ų Structure 1 Structure 2 A = 4872. 3599730(32) MHz B = 990. 7651386(43) MHz C = 828. 84446100(52) MHz A = 4843. 134345(34) MHz B = 991. 3820658(40) MHz C = 830. 39654625( 32) MHz DI�’’ = -4. 0740153(38) DI�’’ = -5. 5221636(51) Trans- conformer of 4 MP µa = 0. 54 Debye, µb = -0. 21 Debye DI� ’’ ab initio = -3. 24 amu. Ų Work is in progress Trans- 4 MP Cis- 4 MP u 45 : OCH 3 out of plane bending u 44 : CH 3 torsion 12

Conclusions q The complete conformational landscape of Methoxyphenols are studied by a combination of ab initio calculations and high resolution Mm wave spectroscopy. q Analysis of the rotation structures at low frequency excited modes: Inertia defect values were used as sensors to straightforwardly assign rovibrational structures Determination of rotational constants for these modes will be very helpful for the analysis of FIR and MID-IR spectra. q The next step would be to exploit these results in the rovibrationel analysis of infrared spectra recorded with Jet-AILES experimental setups (FT-FIR, SOLEIL) and SPIRALES (low-temperature QCL laser spectroscopy in the mid-infrared range, MONARIS Laboratory , UPMC). 13

The LPCA THz team Tha nks for you r at ten tion ! D. Fontanari (postdoc) From left to right: • Francis Hindle (associate Pr) • Cédric Bray (postdoc) • Robin Bocquet (Pr) • Arnaud Cuisset (Pr) • Gaël Mouret (Pr) • Anthony Roucou (Ph. D student) looking for a postdoc position Financial support: Labex Ca. PPA 14
- Slides: 14