ML 6 sonly bonding d 0 d 10
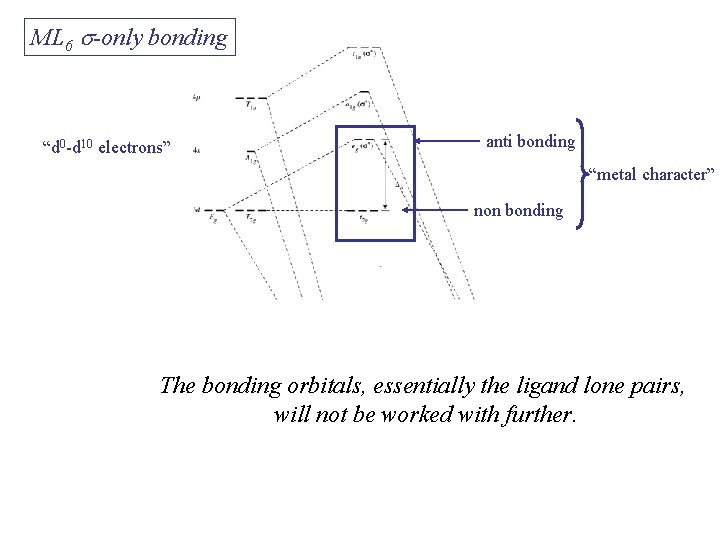
ML 6 s-only bonding “d 0 -d 10 electrons” anti bonding “metal character” non bonding 6 s ligands x 2 e each The bonding orbitals, essentially the ligand lone pairs, 12 s bondingwill e not be worked with further. “ligand character”
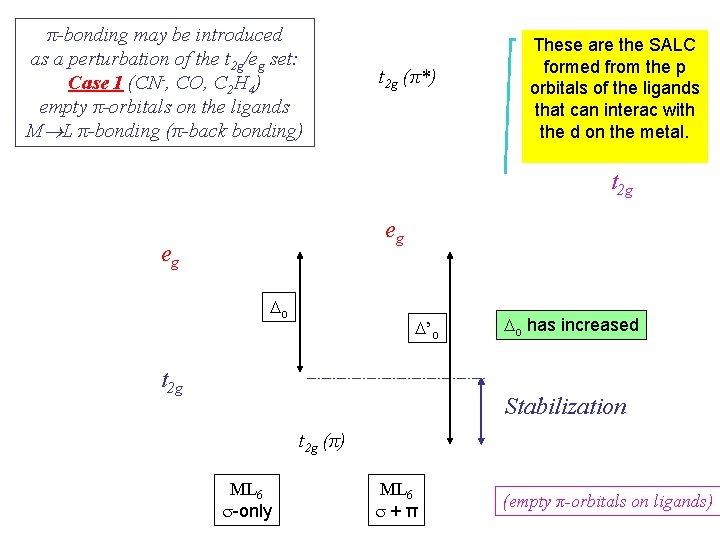
π-bonding may be introduced as a perturbation of the t 2 g/eg set: Case 1 (CN-, CO, C 2 H 4) empty π-orbitals on the ligands M L π-bonding (π-back bonding) t 2 g (π*) These are the SALC formed from the p orbitals of the ligands that can interac with the d on the metal. t 2 g eg eg Do D’o t 2 g Do has increased Stabilization t 2 g (π) ML 6 s-only ML 6 s+π (empty π-orbitals on ligands)
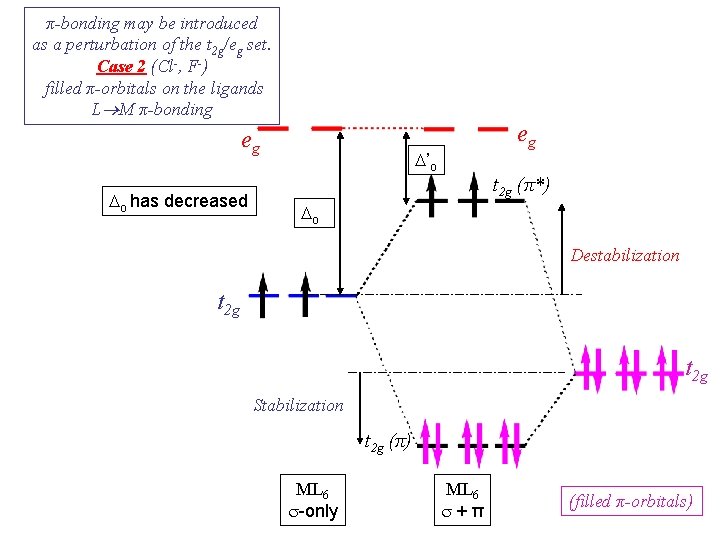
π-bonding may be introduced as a perturbation of the t 2 g/eg set. Case 2 (Cl-, F-) filled π-orbitals on the ligands L M π-bonding eg Do has decreased eg D’o t 2 g (π*) Do Destabilization t 2 g Stabilization t 2 g (π) ML 6 s-only ML 6 s+π (filled π-orbitals)
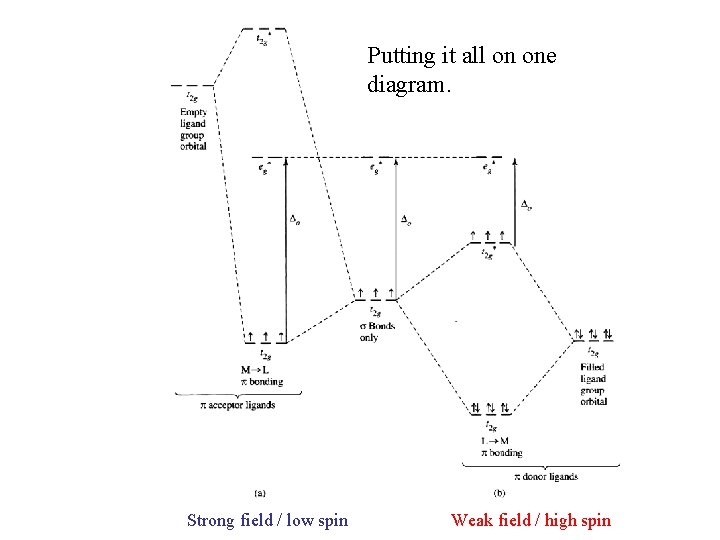
Putting it all on one diagram. Strong field / low spin Weak field / high spin

Spectrochemical Series Purely s ligands: D: en > NH 3 (order of proton basicity) p donating which decreases splitting and causes high spin: D: H 2 O > F > RCO 2 > OH > Cl > Br > I (also proton basicity) p accepting ligands increase splitting and may be low spin D: CO, CN-, > phenanthroline > NO 2 - > NCS-

Merging to get spectrochemical series CO, CN- > phen > NH 3 > NCS- > H 2 O > F- > RCO 2 - > OH- > Cl- > Br- > I- Strong field, p acceptors large D low spin s only Weak field, p donors small D high spin

Turning to Square Planar Complexes Most convenient to use a local coordinate system on each ligand with y pointing in towards the metal. py to be used for s bonding. z being perpendicular to the molecular plane. pz to be used for p bonding perpendicular to the plane, p^. x lying in the molecular plane. px to be used for p bonding in the molecular plane, p|.
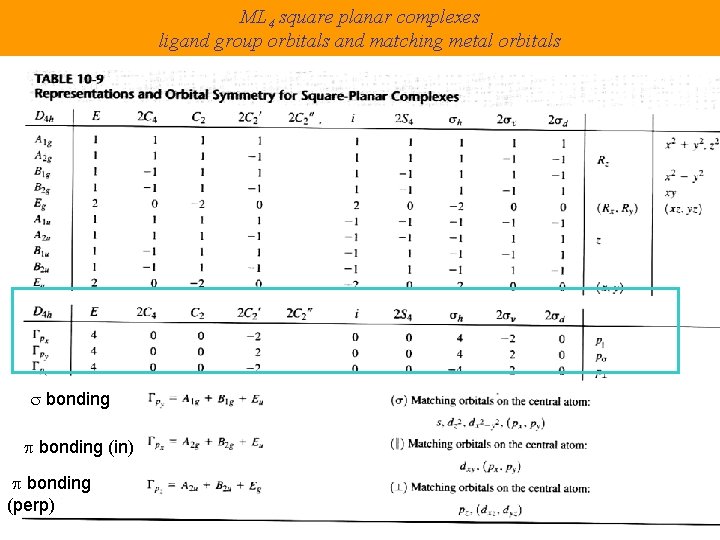
ML 4 square planar complexes ligand group orbitals and matching metal orbitals s bonding p bonding (in) p bonding (perp)
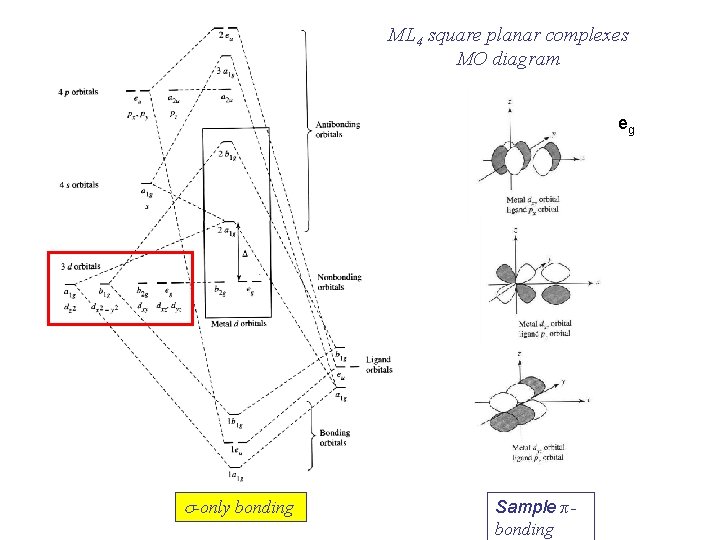
ML 4 square planar complexes MO diagram eg s-only bonding Sample π bonding

Angular Overlap Method An attempt to systematize the interactions for all geometries. The various complexes may be fashioned out of the ligands above Linear: 1, 6 Tetrahedral: 7, 8, 9, 10 Trigonal: 2, 11, 12 Square planar: 2, 3, 4, 5 T-shape: 1, 3, 5 Trigonal bipyramid: 1, 2, 6, 11, 12 Square pyramid: 1, 2, 3, 4, 5 Octahedral: 1, 2, 3, 4, 5, 6

Cont’d All s interactions with the ligands are stabilizing to the ligands and destabilizing to the d orbitals. The interaction of a ligand with a d orbital depends on their orientation with respect to each other, estimated by their overlap which can be calculated. The total destabilization of a d orbital comes from all the interactions with the set of ligands. For any particular complex geometry we can obtain the overlaps of a particular d orbital with all the various ligands and thus the destabilization.
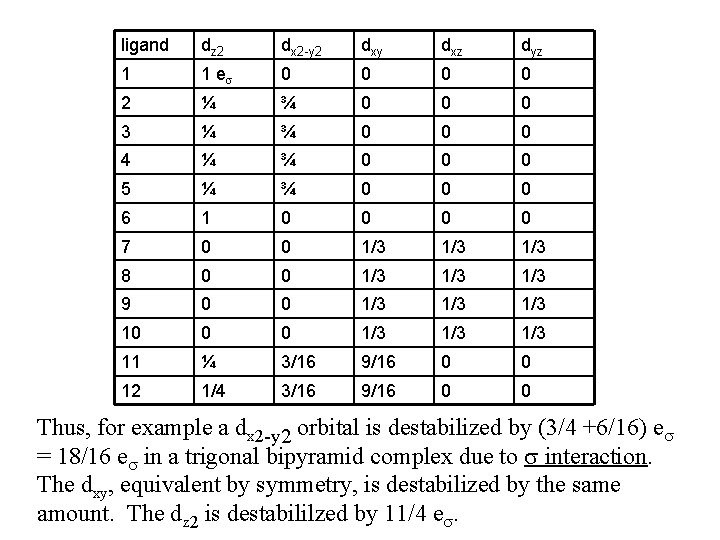
ligand dz 2 dx 2 -y 2 dxy dxz dyz 1 1 es 0 0 2 ¼ ¾ 0 0 0 3 ¼ ¾ 0 0 0 4 ¼ ¾ 0 0 0 5 ¼ ¾ 0 0 0 6 1 0 0 7 0 0 1/3 1/3 8 0 0 1/3 1/3 9 0 0 1/3 1/3 10 0 0 1/3 1/3 11 ¼ 3/16 9/16 0 0 12 1/4 3/16 9/16 0 0 Thus, for example a dx 2 -y 2 orbital is destabilized by (3/4 +6/16) es = 18/16 es in a trigonal bipyramid complex due to s interaction. The dxy, equivalent by symmetry, is destabilized by the same amount. The dz 2 is destabililzed by 11/4 es.

Coordination Chemistry Electronic Spectra of Metal Complexes

Electronic spectra (UV-vis spectroscopy)
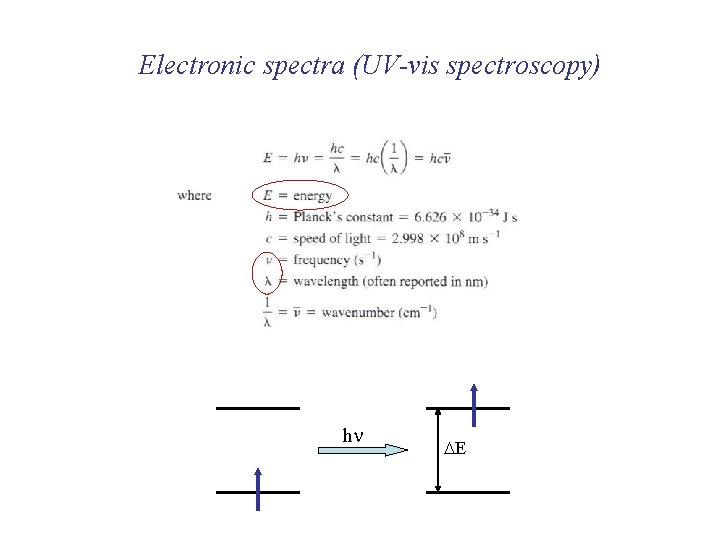
Electronic spectra (UV-vis spectroscopy) hn DE
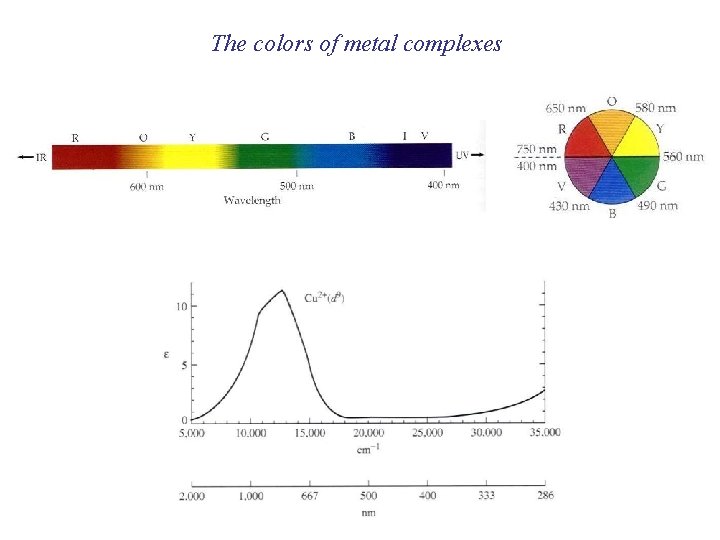
The colors of metal complexes


Electronic configurations of multi-electron atoms What is a 2 p 2 configuration? n = 2; l = 1; ml = -1, 0, +1; ms = ± 1/2 Many configurations fit that description These configurations are called microstates and they have different energies because of inter-electronic repulsions

Electronic configurations of multi-electron atoms Russell-Saunders (or LS) coupling For each 2 p electron n = 1; l = 1 ml = -1, 0, +1 ms = ± 1/2 For the multi-electron atom L = total orbital angular momentum quantum number S = total spin angular momentum quantum number Spin multiplicity = 2 S+1 ML = ∑ml (-L, … 0, …+L) MS = ∑ms (S, S-1, …, 0, …-S) ML/MS define microstates and L/S define states (collections of microstates) Groups of microstates with the same energy are called terms
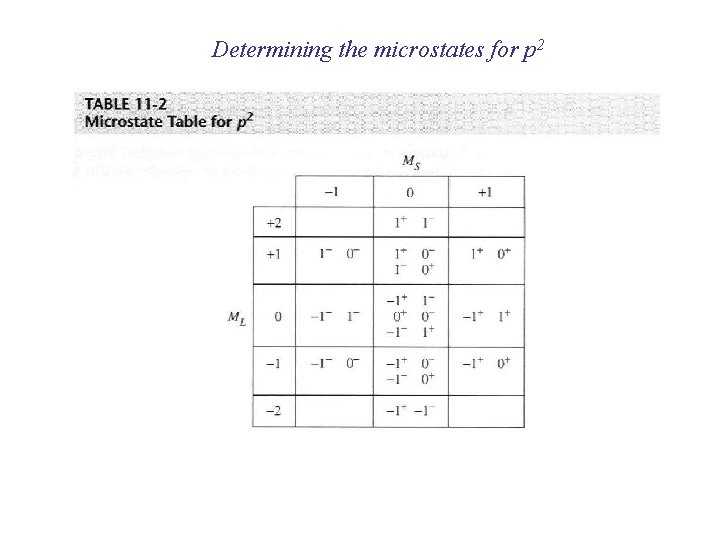
Determining the microstates for p 2

Spin multiplicity 2 S + 1
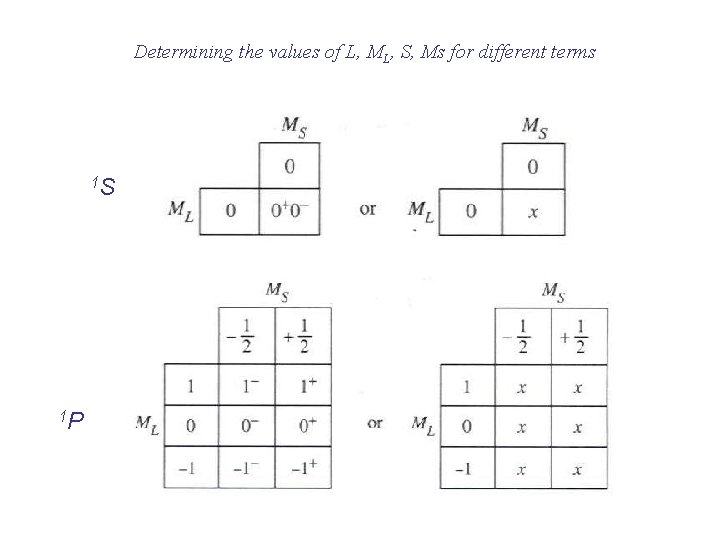
Determining the values of L, ML, S, Ms for different terms 1 S 1 P

Classifying the microstates for p 2 Largest ML is +2, so L = 2 (a D term) and MS = 0 for ML = +2, 2 S +1 = 1 (S = 0) 1 D Next largest ML is +1, so L = 1 (a P term) and MS = 0, ± 1/2 for ML = +1, 2 S +1 = 3 3 P Spin multiplicity = # columns of microstates One remaining microstate ML is 0, L = 0 (an S term) and MS = 0 for ML = 0, 2 S +1 = 1 1 S
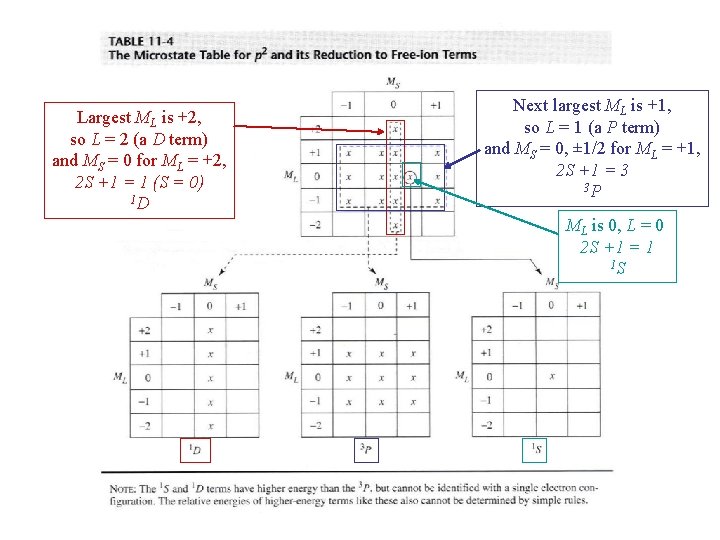
Largest ML is +2, so L = 2 (a D term) and MS = 0 for ML = +2, 2 S +1 = 1 (S = 0) 1 D Next largest ML is +1, so L = 1 (a P term) and MS = 0, ± 1/2 for ML = +1, 2 S +1 = 3 3 P ML is 0, L = 0 2 S +1 = 1 1 S

Energy of terms (Hund’s rules) Lowest energy (ground term) Highest spin multiplicity 3 P term for p 2 case 3 P has S = 1, L = 1 If two states have the same maximum spin multiplicity Ground term is that of highest L
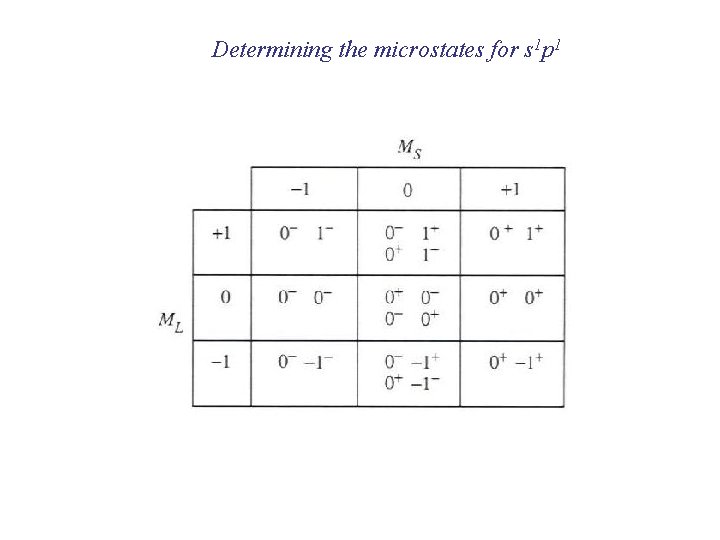
Determining the microstates for s 1 p 1

Determining the terms for s 1 p 1 Ground-state term

Coordination Chemistry Electronic Spectra of Metal Complexes cont.

Electronic configurations of multi-electron atoms Russell-Saunders (or LS) coupling For each 2 p electron n = 1; l = 1 ml = -1, 0, +1 ms = ± 1/2 For the multi-electron atom L = total orbital angular momentum quantum number S = total spin angular momentum quantum number Spin multiplicity = 2 S+1 ML = ∑ml (-L, … 0, …+L) MS = ∑ms (S, S-1, …, 0, …-S) ML/MS define microstates and L/S define states (collections of microstates) Groups of microstates with the same energy are called terms
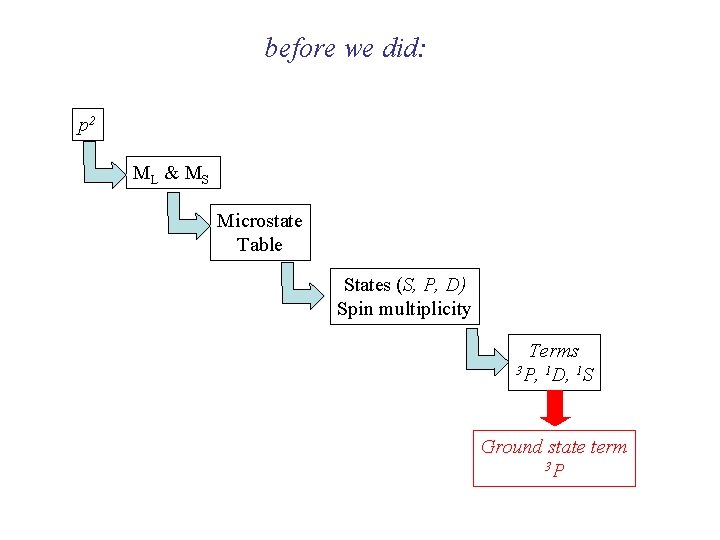
before we did: p 2 ML & M S Microstate Table States (S, P, D) Spin multiplicity Terms 3 P, 1 D, 1 S Ground state term 3 P

For metal complexes we need to consider d 1 -d 10 d 2 3 F, 3 P, 1 G, 1 D, 1 S For 3 or more electrons, this is a long tedious process But luckily this has been tabulated before…

Transitions between electronic terms will give rise to spectra

Selection rules (determine intensities) Laporte rule g g forbidden (that is, d-d forbidden) but g u allowed (that is, d-p allowed) Spin rule Transitions between states of different multiplicities forbidden Transitions between states of same multiplicities allowed These rules are relaxed by molecular vibrations, and spin-orbit coupling

Group theory analysis of term splitting

An e electron superimposed on a spherical distribution energies reversed because tetrahedral High Spin Ground States Holes in d 5 and d 10, reversing energies relative to d 1 dn Free ion GS Oct. complex Tet complex d 0 1 S t 2 g 0 eg 0 e 0 t 20 d 1 2 D t 2 g 1 eg 0 e 1 t 20 d 2 3 F t 2 g 2 eg 0 e 2 t 20 d 3 4 F t 2 g 3 eg 0 e 2 t 21 d 4 5 D t 2 g 3 eg 1 e 2 t 22 d 5 6 S t 2 g 3 eg 2 e 2 t 23 d 6 5 D t 2 g 4 eg 2 e 3 t 23 d 7 4 F t 2 g 5 eg 2 e 4 t 23 d 8 3 F t 2 g 6 eg 2 e 4 t 24 d 9 2 D t 2 g 6 eg 3 e 4 t 25 d 10 1 S t 2 g 6 eg 4 e 4 t 26 Holes: dn = d 10 -n and neglecting spin dn = d 5+n; same splitting but reversed energies because positive. Expect oct d 1 and d 6 to behave same as tet d 4 and d 9 Expect oct d 4 and d 9 (holes), tet d 1 and d 6 to be reverse of oct d 1 A t 2 hole in d 5, reversed energies, reversed again relative to octahedral since tet.
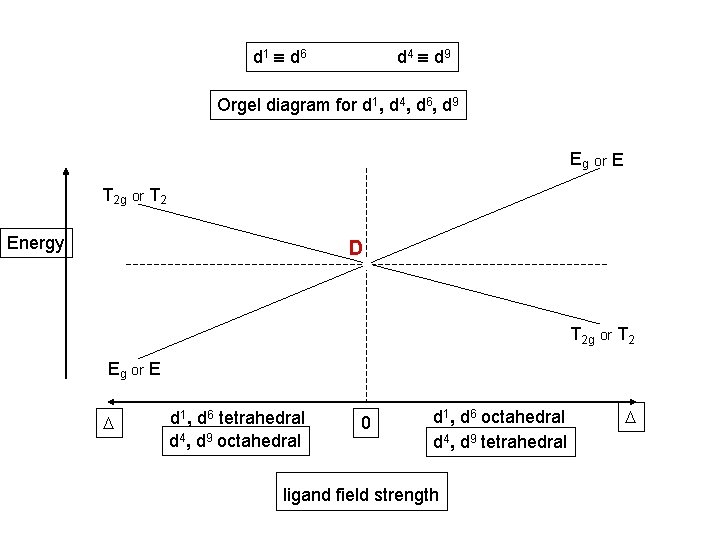
d 1 d 6 d 4 d 9 Orgel diagram for d 1, d 4, d 6, d 9 Eg or E T 2 g or T 2 Energy D T 2 g or T 2 Eg or E D d 1, d 6 tetrahedral d 4, d 9 octahedral 0 d 1, d 6 octahedral d 4, d 9 tetrahedral ligand field strength D
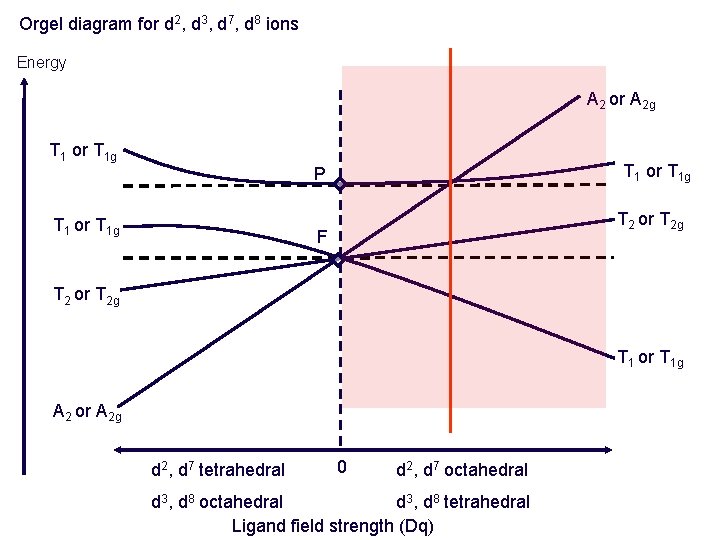
Orgel diagram for d 2, d 3, d 7, d 8 ions Energy A 2 or A 2 g T 1 or T 1 g P T 1 or T 1 g T 2 or T 2 g F T 2 or T 2 g T 1 or T 1 g A 2 or A 2 g d 2, d 7 tetrahedral 0 d 2, d 7 octahedral d 3, d 8 tetrahedral Ligand field strength (Dq)
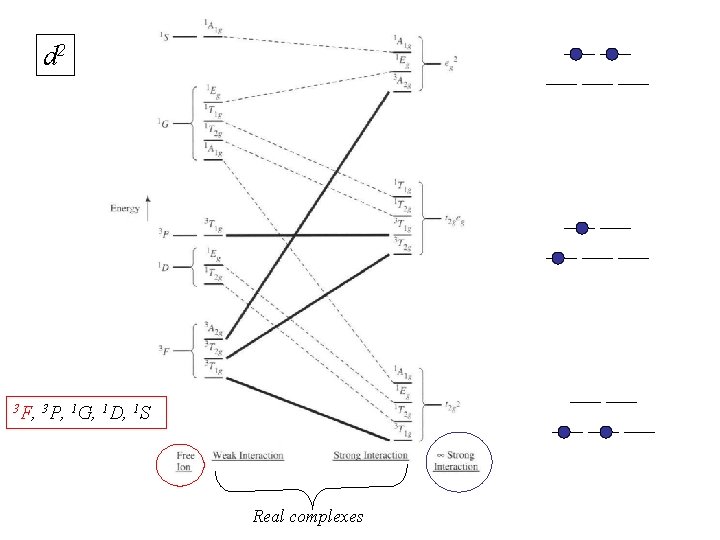
d 2 3 F, 3 P, 1 G, 1 D, 1 S Real complexes

Tanabe-Sugano diagrams
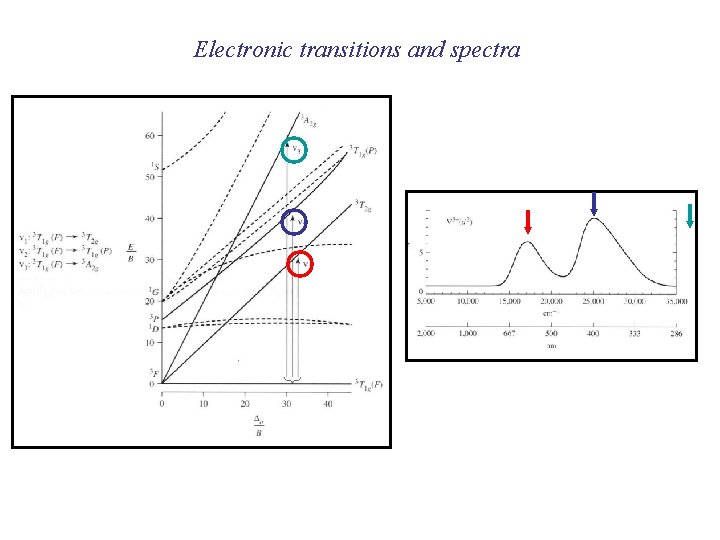
Electronic transitions and spectra

Other configurations d 3 d 9 d 1 d 2 d 8
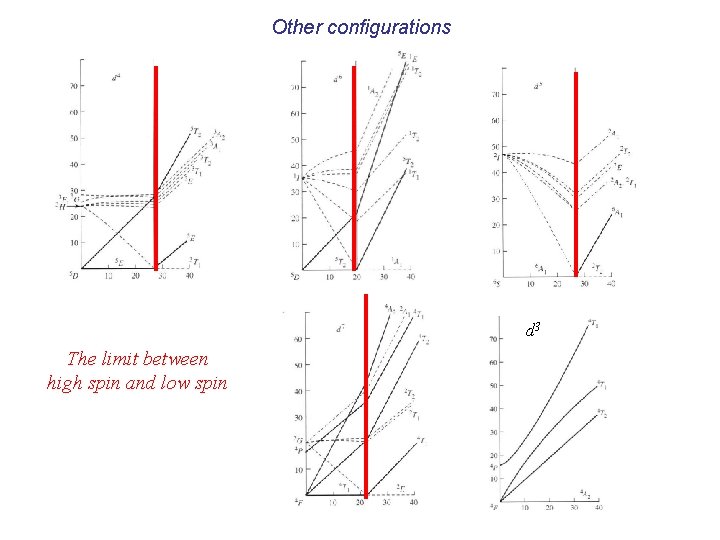
Other configurations d 3 The limit between high spin and low spin
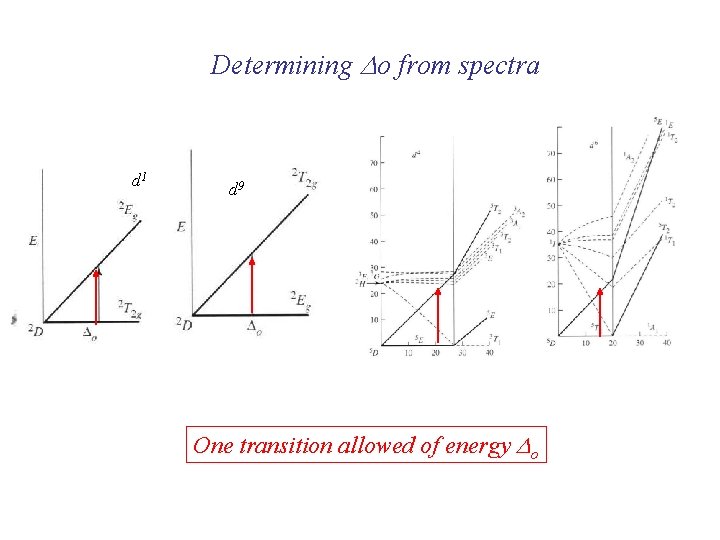
Determining Do from spectra d 1 d 9 One transition allowed of energy Do

Determining Do from spectra mixing Lowest energy transition = Do

Ground state is mixing E (T 1 g A 2 g) - E (T 1 g T 2 g) = Do
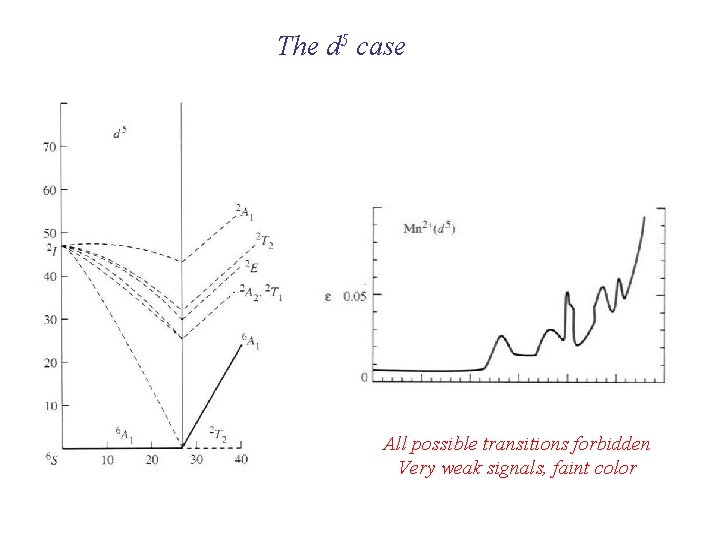
The d 5 case All possible transitions forbidden Very weak signals, faint color
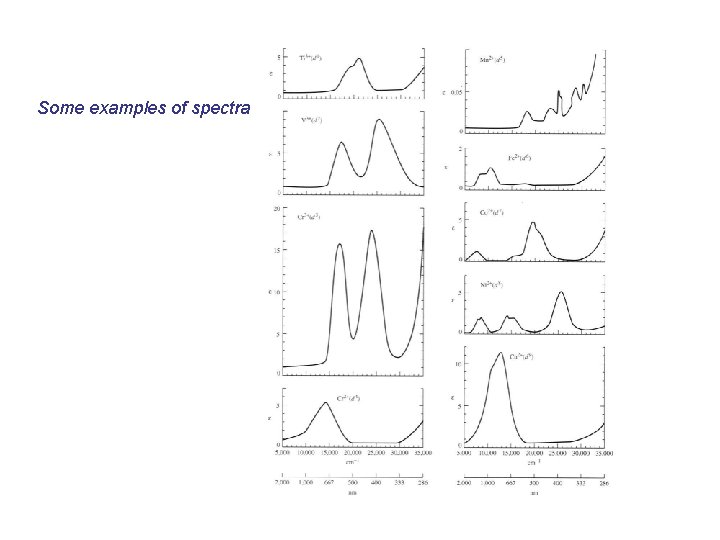
Some examples of spectra
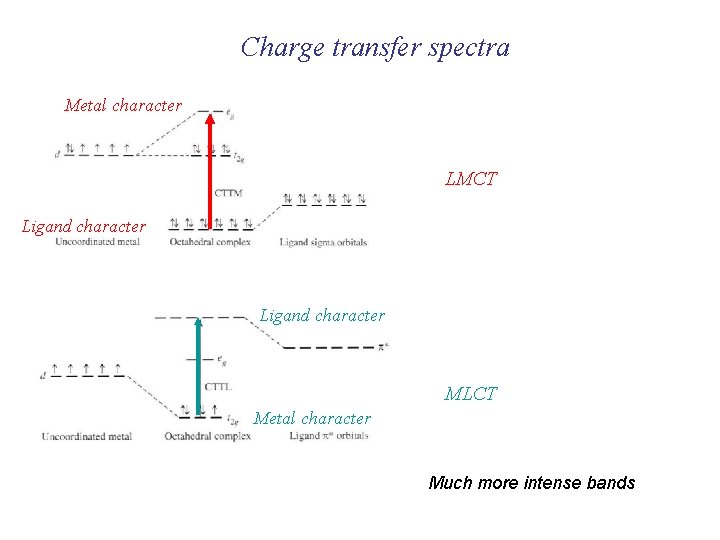
Charge transfer spectra Metal character LMCT Ligand character MLCT Metal character Much more intense bands
- Slides: 48