Mixtures Solutions What are Substances Substances can be

Mixtures & Solutions

What are “Substances”? �Substances - can be identified as either an element, compound, or a mixture. �A substance cannot be further broken down by physical means.
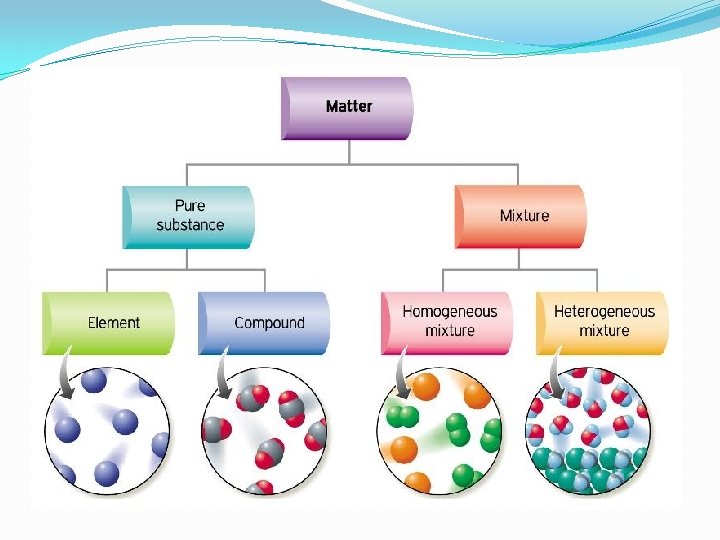

What is a “Mixture”? �A mixture is made when two or more substances are combined, but they are NOT combined chemically. �Mixtures can be liquids, gases, and solids.

Homogenous Mixture �In a homogenous mixture all the substances are evenly distributed throughout the mixture. � Examples: salt water, air, blood, yogurt, & brass

Heterogeneous Mixture �In a heterogeneous mixture the substances are not evenly distributed and they are easily separated. �Examples: chocolate chip cookies, pizza, fruit salad soil, oil & water

Solutions �A solution is a homogeneous mixture, thus it has the same appearance and properties throughout the mixture. �Examples: sugar water, salt water

Soluble vs. Insoluble • A substance that dissolves in a solvent is said to be soluble. �Insoluble – a substance that does not dissolve in a solvent.

Parts of a Solution �Solute- is the substance being dissolved. o present in the smaller amount �Solvent- is the substance that dissolves the solute. o present in the larger amount

Examples: Solution Solvent Solute 1. ) Chocolate Milk: Choc. Syrup + Milk Choc. Syrup 2. ) Kool-Aid: Water + Drink Powder Water Drink powder 3. ) Alka-seltzer: CO 2 + Water CO 2

Universal Solvent v. Water is called the universal solvent because many substances can dissolve in it. ØWater is a polar molecule. • Oil is not polar. • Oil will not dissolve in water.

Solutions �An aqueous solution - is one in which WATER is the solvent.

�Solutions song �http: //matthews. watchknowlearn. org/Video. aspx? Vid eo. ID=34238&Category. ID=6741

Miscible �Two liquids that are soluble in each other are said to be miscible. �Ex 1: Water and vinegar �Ex 2: Acetone & water

Immiscible �Two liquids that are not soluble in each other are said to be immiscible. �Ex 1: Oil & water �Ex 2: Oil & vinegar
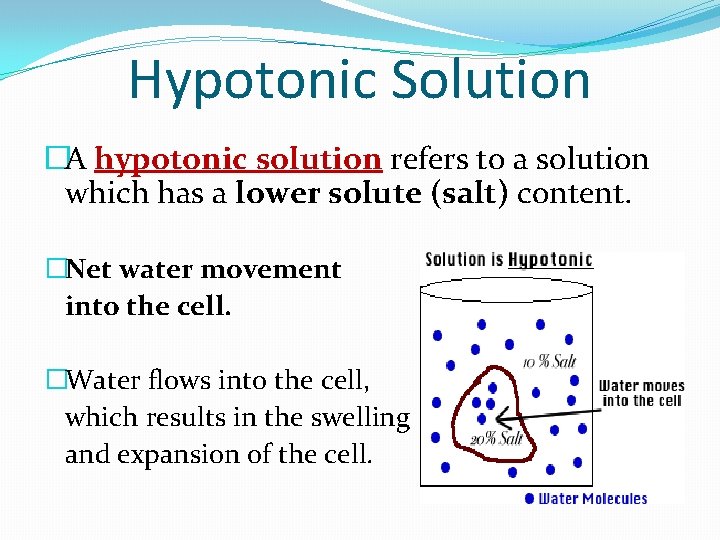
Hypotonic Solution �A hypotonic solution refers to a solution which has a lower solute (salt) content. �Net water movement into the cell. �Water flows into the cell, which results in the swelling and expansion of the cell.

Hypertonic Solution �A hypertonic solution has a higher or has more solutes (salt) than another solution. �The word "HYPER" means more. �Water flows out of the cell so this will cause the cell to shrink.
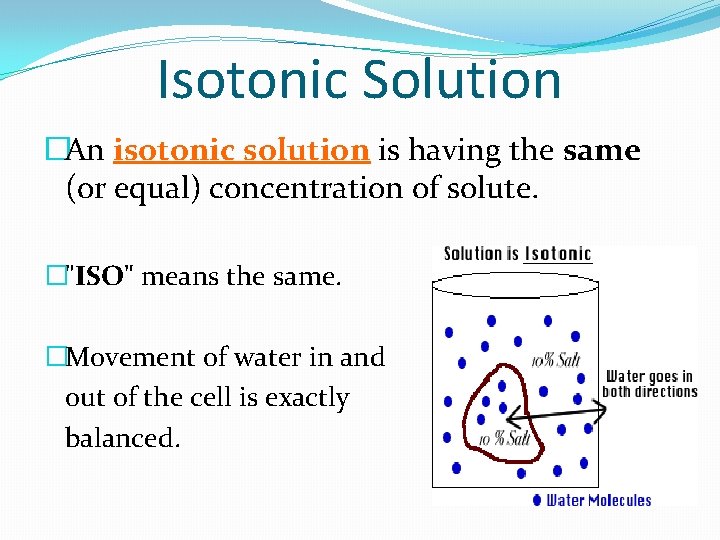
Isotonic Solution �An isotonic solution is having the same (or equal) concentration of solute. �"ISO" means the same. �Movement of water in and out of the cell is exactly balanced.

�http: //www. tvdsb. ca/webpages/brownt 12/files/osmosis. htm
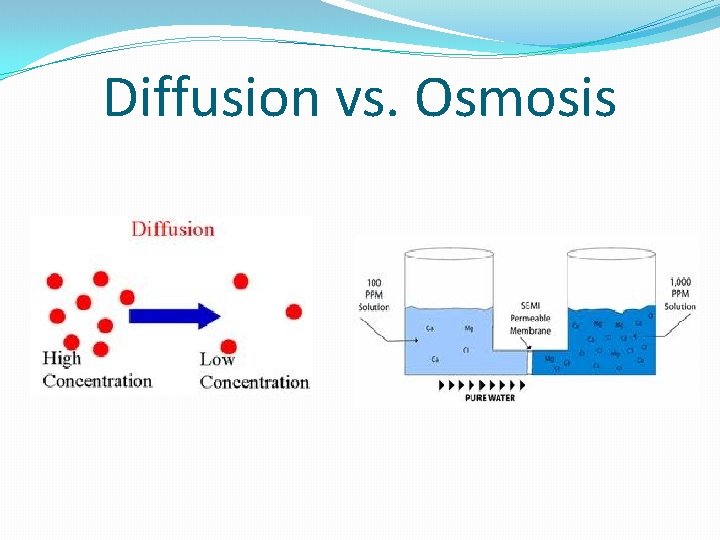
Diffusion vs. Osmosis

Diffusion �Diffusion is the process by which substances move from areas of higher concentration to areas of lower concentration.

Osmosis �Osmosis is the movement of water through a semi-permeable membrane from an area of high-water concentration to an area of low-water concentration.
- Slides: 22