Mixtures Solutions Ms Rosendo Mixtures a combination of

Mixtures (Solutions) Ms. Rosendo

Mixtures • a combination of two or more substances that do not combine chemically, but remain the same individual substances; can be separated by physical means • Two types: – Heterogeneous – Homogeneous Based on the prefixes “hetero” and “homo, ” what do you think are characteristics of these two types of mixtures?

Heterogeneous Mixture • “Hetero” means different • consists of visibly different substances or phases (solid, liquid, gas) • a suspension is a special type of heterogeneous mixture of larger particles that eventually settle • Example: Trail Mix Notice the visibly different substances

Homogeneous Mixture • “Homo” means the same • has the same uniform appearance and composition throughout; maintain one phase (solid, liquid, gas) • Commonly referred to as solutions • Example: Salt Water Notice the uniform appearance
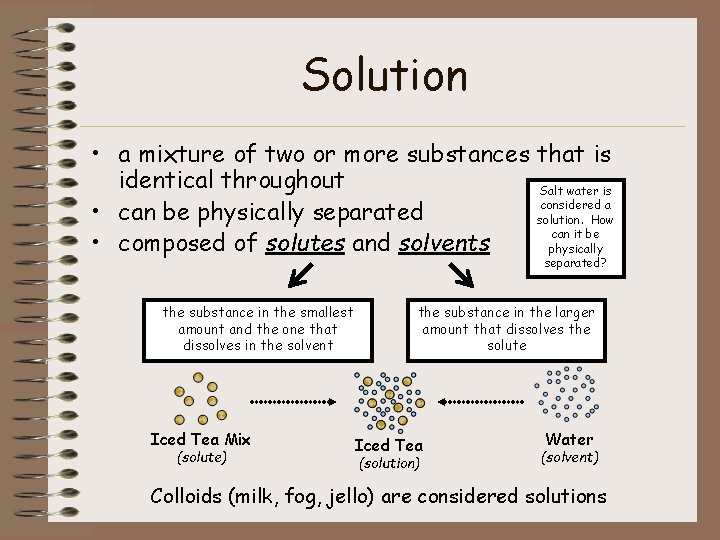
Solution • a mixture of two or more substances that is identical throughout Salt water is considered a • can be physically separated solution. How can it be • composed of solutes and solvents physically separated? the substance in the smallest amount and the one that dissolves in the solvent Iced Tea Mix (solute) the substance in the larger amount that dissolves the solute Iced Tea (solution) Water (solvent) Colloids (milk, fog, jello) are considered solutions

Concentration • the amount of solute dissolved in a solvent at a given temperature • described as dilute if it has a low concentration of solute • described as saturated if it has a high concentration of solute • described as supersaturated if contains more dissolved solute than normally possible

Solubility • the amount of solute that dissolves in a certain amount of a solvent at a given temperature and pressure to produce a saturated solution What do we call things that are not soluble? • influenced by: Temperature Pressure Solids increased temperature causes them to be more soluble and vice versa Solids increased pressure has no effect on solubility Gases increased temperature causes them to be less soluble and vice versa Gases increased pressure causes them to be more soluble and vice versa Ex. Iced Coffee Ex. Soda, “The Bends”

Concentration of Solute The amount of solute in a solution is given by its concentration. Molarity (M) = moles solute liters of solution

PROBLEM: Dissolve 5. 00 g of Ni. Cl 2 in enough water to make 250 m. L of solution. Calculate the Molarity. Step 1: Calculate moles of Ni. Cl 2 Step 2: Calculate Molarity [Ni. Cl 2] = 0. 154 M

USING MOLARITY What mass of oxalic acid, H 2 C 2 O 4, is required to make 250. m. L of a 0. 0500 M solution? moles = M • V Step 1: Change m. L to L. 250 m. L * 1 L/1000 m. L = 0. 250 L Step 2: Calculate. Moles = (0. 0500 mol/L) (0. 250 L) = 0. 0125 moles Step 3: Convert moles to grams. (0. 0125 mol)(90. 00 g/mol) = 1. 13 g

Learning Check How many grams of Na. OH are required to prepare 400. m. L of 3. 0 M Na. OH solution? 1) 12 g 2) 48 g 3) 300 g

Two Other Concentration Units MOLALITY, m mol solute m of solution = kilograms solvent % by mass = grams solute grams solution

Calculating Concentrations Dissolve 62. 1 g (1. 00 mol) of ethylene glycol in 250. g of H 2 O. Calculate molality and % by mass of ethylene glycol.

p. H Scale Na. OH (sodium hydroxide) Tums (antacid tablets) Pure Water (distilled) Soda HCl (hydrochloric acid) • ranges from 0 -14 – Acids found between a number close to 0 & 7 – Bases found between 7 & 14 – Neutral 7 • measures the acidity or basicity of a solution by focusing on the concentration of hydrogen ions (H+) in a solution • equals the negative log of the concentration of H+
- Slides: 14