Mixtures Solutions Ch 14 Mixtures A combination of

Mixtures & Solutions Ch 14

Mixtures • A combination of 2 or more pure substances in which each pure substance retains its individual chemical properties. • Solutions are homogeneous mixtures.

Heterogeneous Mixture • Suspensions-a mixture containing particles that settle out if left undisturbed. Particle size is large. Ex. Muddy water • Suspensions can be separated with filtration • Colloids-intermediate size particles that stay suspended in solvent and do not settle out. Cannot be separated by filtration. Ex-Milk

Tyndell Effect • The scattering of light by colloidal particles. Pg 478 Figure 14 -3

Homogeneous Mixtures • Solution – homogenous mixture of two or more substances – Solute – substance that dissolves – Solvent – dissolving medium • Soluble-describes a substance that can be dissolved in a given solvent. • Miscible-describes two liquids that are soluble in each other. • Insoluble-describes a substance that can be dissolved in a given solvent. • Immiscible-describes two liquids that can not be mixed together but separate shortly after you cease mixing them.

Checkpoint • 1. Use the properties of seawater to describe the characteristics of mixtures. • 2. Distinguish between suspensions and colloids. • 3. Identify the various types of solutions. Describe the characteristics of each type of solution. • 4. Explain using the Tyndall Effect why it is more difficult to drive through fog using high beams than using low beams.

Checkpoint con’t • 5. Describe the different tyoes of colloids. • 6. Explain why dispersed particles stay dispersed? • 7. What causes Brownian motion? • 8. Compare and contrast –make a table that compares the properties of suspensions, colloids, and solutions.

Answers • 1. Seawater is heterogeneous mixture with dirt and mud particles and dissolved minerals. • 2. Suspension particles are larger than colloidal particles. Suspension particles settle out of the mixture and colloidal do not. Suspension particles can be separated by filtration and colloidal cannot. • 3. All solutions are homogeneous mixtures containing 2 or more substances. Solutions can be liquids, solids, or gases. •

Answers con’t • 4. High beams are aimed farther down the road than low beams. Because the fog scatters light, there is less light from high beams to illuminate the road than from the low beams. Also, because the high beams are aimed more directly into the fog, more of their light is reflected back toward the driver, making it more difficult to see. • 5. See table 14 -1 p 477. • 6. The particles do not settle out because they have polar or charged layers surrounding them. These layers repel each other and prevent the particles from settling or separating.

• 7. Collisions of particles of the dispersion medium with the dispersed particles rsults in Brownian Motion. • 8. Table should include particle size, if the particles settle out, and if the particles display Tyndall Effect.

Types of Solutions

Video of Solvation • http: //www. glencoe. com/sites/co mmon_assets/science/cmc/cim/an imations/ch 14_1. swf
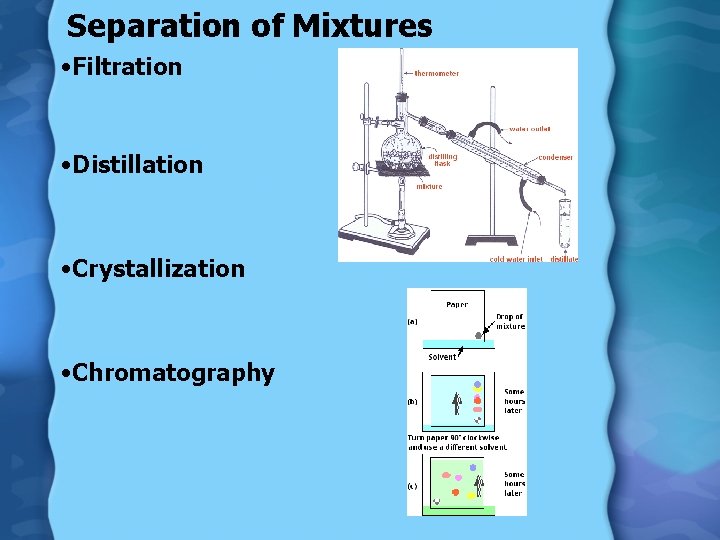
Separation of Mixtures • Filtration • Distillation • Crystallization • Chromatography

Mixtures



Web. Link Online quizzes, flashcards. And more FUN http: //glencoe. mcgrawhill. com/sites/007874637 x/student_view 0/chapter 14/

Mixture Pure substance • A combination of 2 or more pure substances in which each pure substance retains its individual chemical properties. • Elements • Compounds • Solutions are homogeneous mixtures. • Blood is a heterogeneous
- Slides: 19