Mixtures Chapter 3 Section 3 Lesson Objectives Differentiate

Mixtures Chapter 3 Section 3

Lesson Objectives Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Differentiate between solutions, suspensions and colloids Calculate solubility using graphs

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Keywords: Mixture • Homogeneous • Heterogeneous Solutions Alloys Solute Solvent Concentration Types of Solutions: Saturated Unsaturated Supersaturated Solubility Suspensions Colloids Calculate solubility using graphs Differentiate between solutions, suspensions and colloids
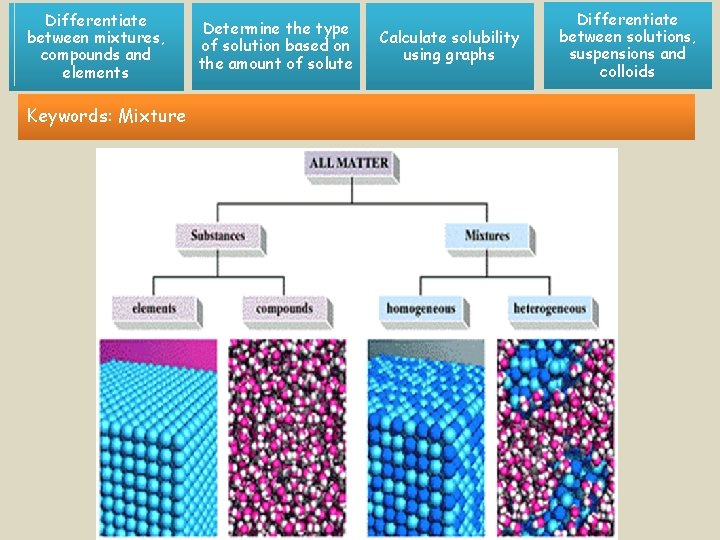
Differentiate between mixtures, compounds and elements Keywords: Mixture Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids
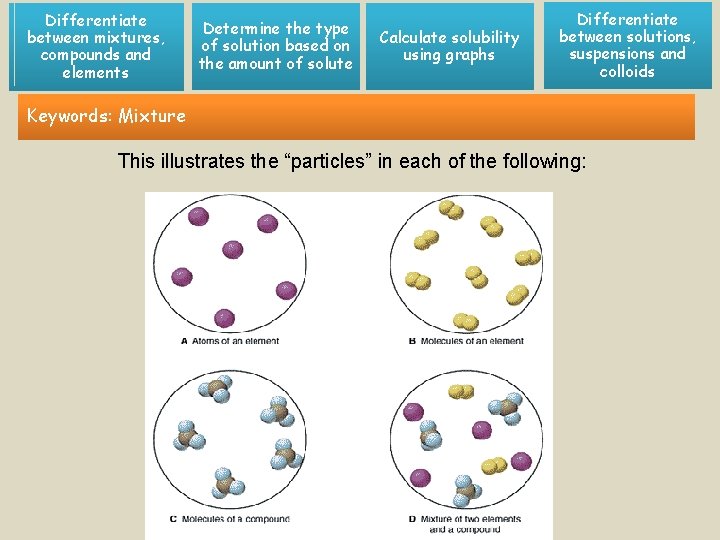
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Mixture This illustrates the “particles” in each of the following:

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Mixtures: Combination of two or more substances that are not chemically combined and they are not in specific ratios. Ex: Cereal and milk, iced tea

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Mixture Substances within a mixture retain their properties because it is not a chemical reaction! Pepperoni Pizza • The cheese is still cheese • The pepperoni is still pepperoni • The sauce is still sauce The identity of the substances that make up the pizza do not change!
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Keywords: Heterogeneous • Does not look the same throughout Differentiate between solutions, suspensions and colloids

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Keywords: Homogeneous • Looks the same throughout Calculate solubility using graphs Differentiate between solutions, suspensions and colloids

Homogeneous or Heterogeneous?

Homogeneous or Heterogeneous?

Homogeneous or Heterogeneous?


Homogeneous or Heterogeneous?

Homogeneous or Heterogeneous?

Homogeneous or Heterogeneous?

Element, Compound, or Mixture?

Element, Compound, or Mixture?
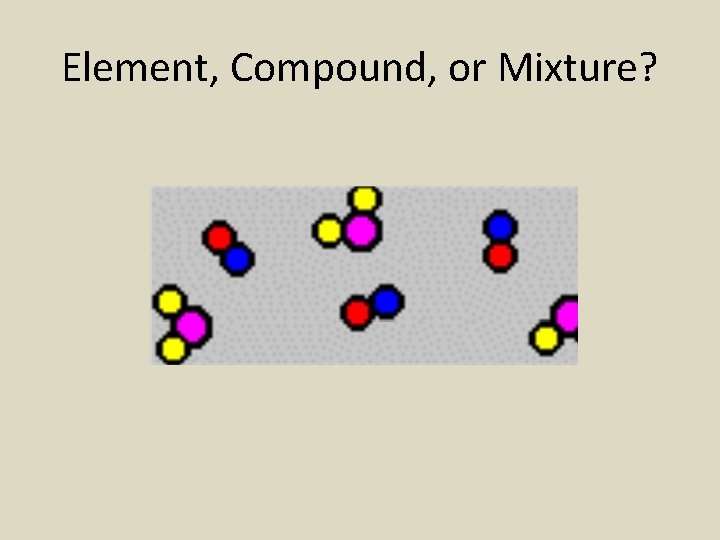
Element, Compound, or Mixture?
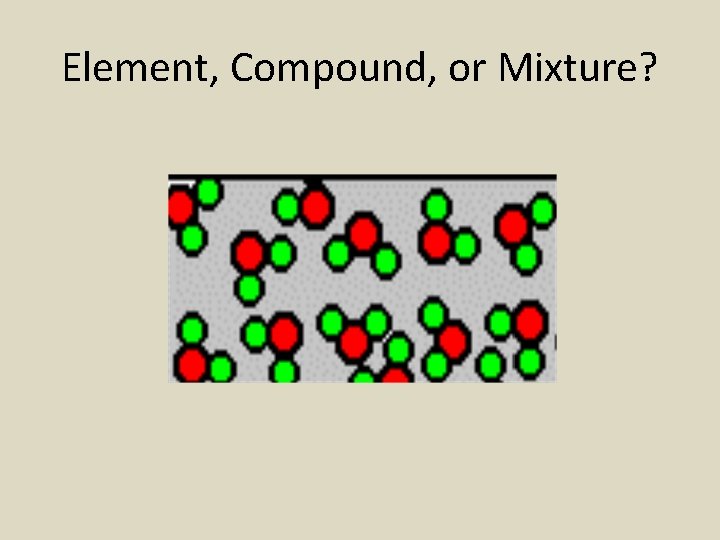
Element, Compound, or Mixture?
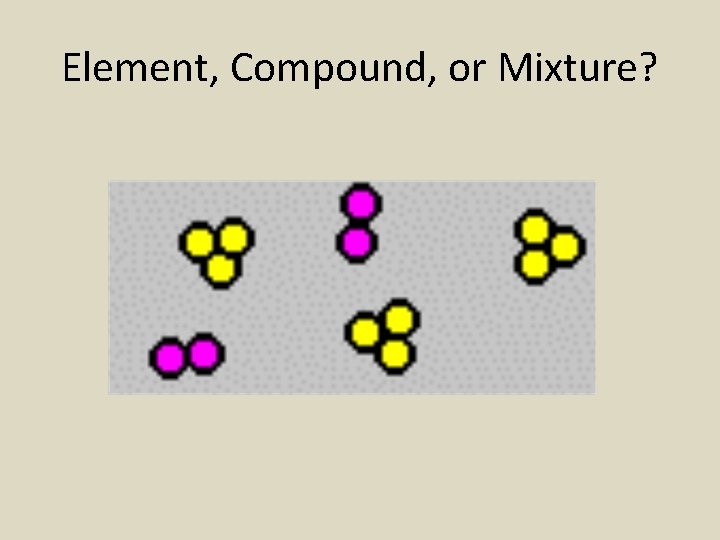
Element, Compound, or Mixture?
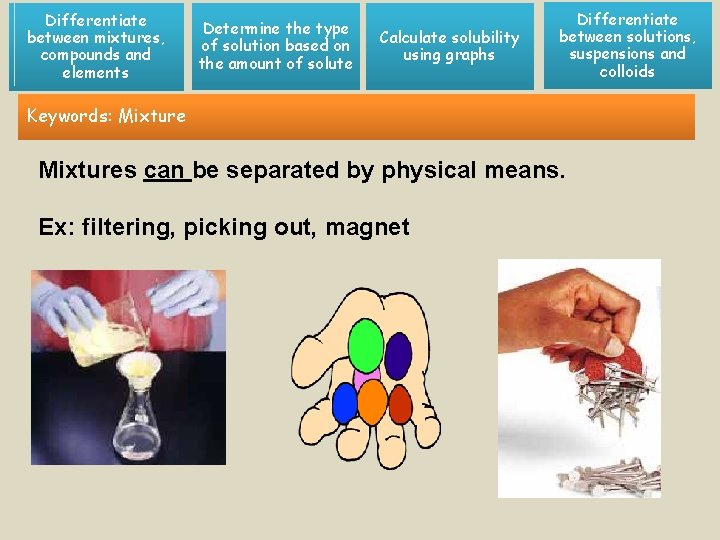
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Mixtures can be separated by physical means. Ex: filtering, picking out, magnet
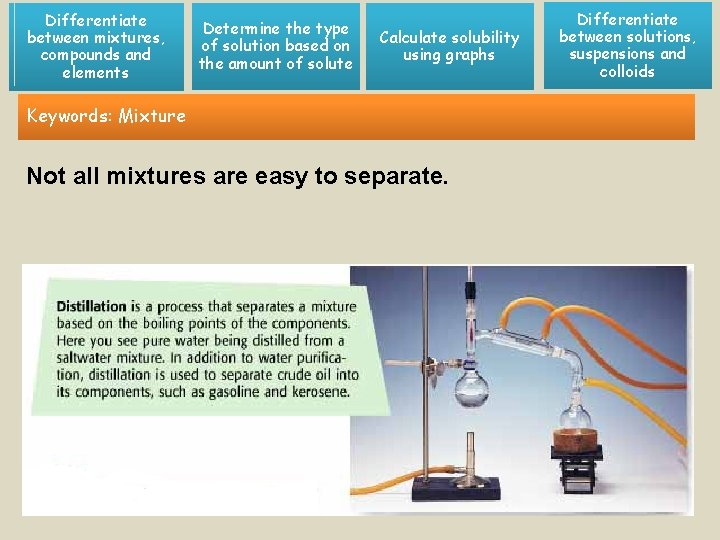
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Keywords: Mixture Not all mixtures are easy to separate. Differentiate between solutions, suspensions and colloids

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Mixture Some mixtures take more than one step to separate

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Keywords: mixtures versus compounds Calculate solubility using graphs Differentiate between solutions, suspensions and colloids

Are you now able to…. Differentiate between mixtures, compounds and elements I still feel unsure. I need some more help to understand. I feel ok. I need to do some more work on my own to check my understanding. I am happy and feel I understand can explain the main points.

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Solutions • Appears to be a single substance but really two or more substances dissolved in a solvent and evenly distributed • Very small particles that never settle out • Homogeneous Examples of Solutions: Iced tea, salt water, soda, gasoline, alloys
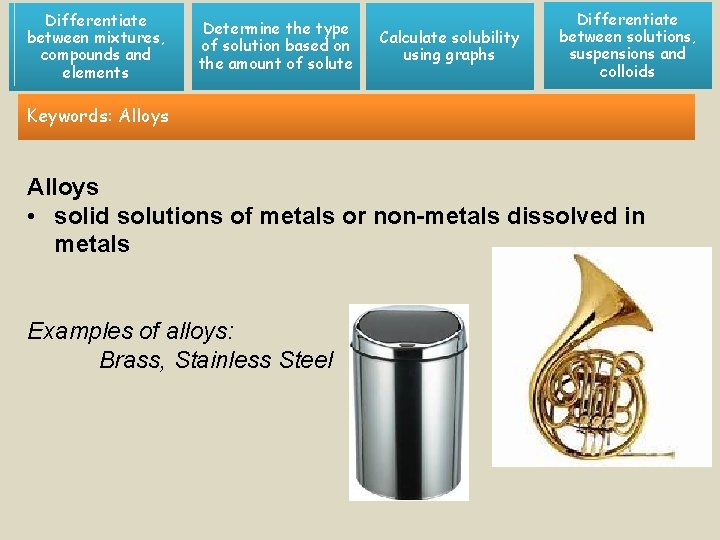
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Alloys • solid solutions of metals or non-metals dissolved in metals Examples of alloys: Brass, Stainless Steel

Differentiate between mixtures, compounds and elements Keywords: Solutions Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Solute and Solvent Solute versus Solvent • A solute is the substance in a solution that gets dissolved • A solvent is the substance in a solution that does the dissolving + Solute Solvent Solution
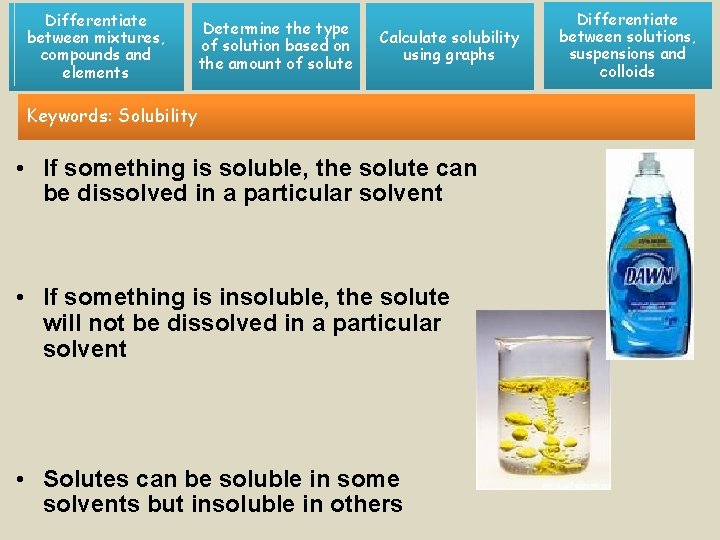
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Keywords: Solubility • If something is soluble, the solute can be dissolved in a particular solvent • If something is insoluble, the solute will not be dissolved in a particular solvent • Solutes can be soluble in some solvents but insoluble in others Differentiate between solutions, suspensions and colloids

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Concentration • The comparison of solute to solvent • When there is not a lot of solute compared to solvent, the solution is dilute • When there is a lot of solute compared to solvent, the solution is concentrated

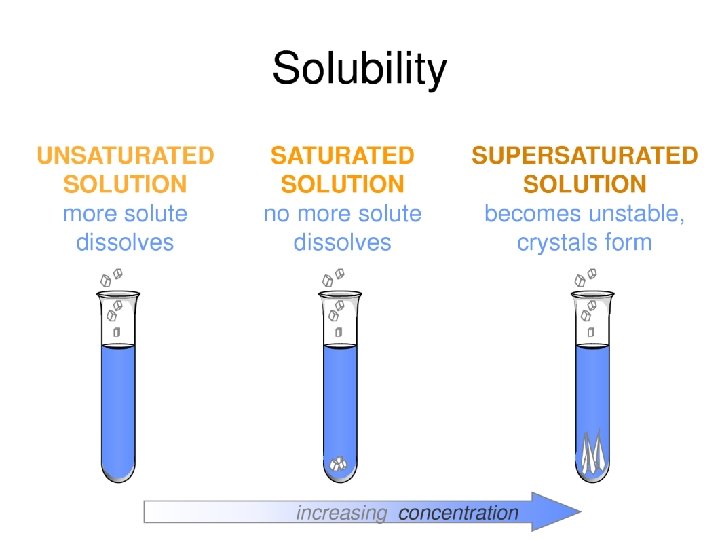
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Saturated Solution: • contains all of the solute it can hold at a given temperature. • If you add even one more molecule of the solute, it will fall out of solution and rest on the bottom

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Keywords: Unsaturated Solution: • Contains less solute than it can hold at a given temperature • Can add more solute and it wont fall out of the solution Differentiate between solutions, suspensions and colloids

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Supersaturated Solution • Solution that holds more than it usually would at a given temperature
Differentiate between mixtures, compounds and elements Keywords: Solutions Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids

Are you now able to…. Determine the type of solution based on the amount of solute I still feel unsure. I need some more help to understand. I feel ok. I need to do some more work on my own to check my understanding. I am happy and feel I understand can explain the main points.

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Solubility: • The amount of solute needed to make a saturated solution in a given solvent at a given temperature • For solid solute in liquid solvent – solubility rises as temperature rises. • For gas solute in a liquid solvent – solubility lowers as temperature rises ex – soda goes flat at warm temperatures

Differentiate between mixtures, compounds and elements Keywords: Solutions Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids
Differentiate between mixtures, compounds and elements Keywords: Solutions Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids

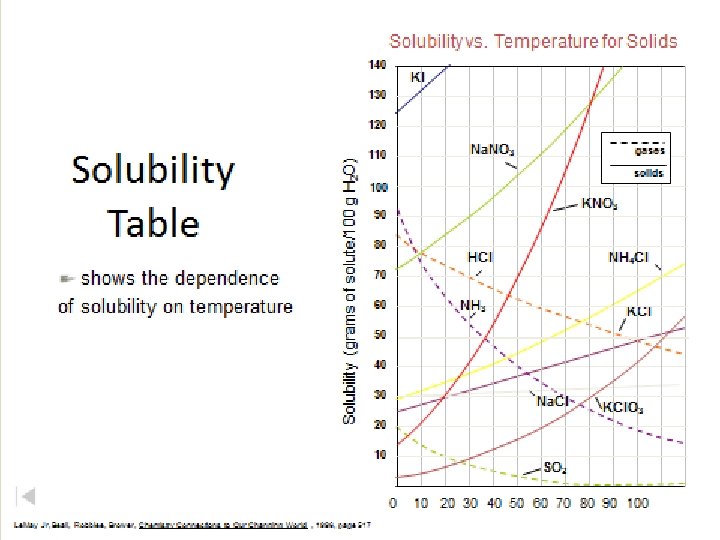
How to determine the solubility of a given substance? • Find out the mass of solute needed to make a saturated solution in 100 cm 3 of water for a specific temperature(referred to as the solubility). • This is repeated for each of the temperatures from 0ºC to 100ºC. The data is then plotted on a temperature/solubility graph, and the points are connected. These connected points are called a solubility curve.


How to use a solubility graph? A. IDENTIFYING A SUBSTANCE ( given the solubility in g/100 cm 3 of water and the temperature) • Look for the intersection of the solubility and temperature.

Example: What substance has a solubility of 90 g/100 cm 3 of water at a temperature of 25ºC ?


Example: What substance has a 3 solubility of 200 g/100 cm of water at a temperature of 90ºC ?


B. Look for the temperature or solubility • Locate the solubility curve needed and see for a given temperature, which solubility it lines up with and visa versa.
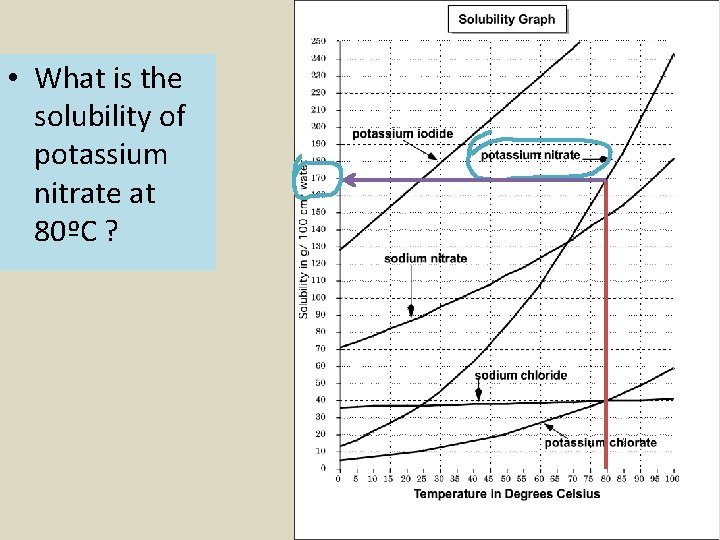
• What is the solubility of potassium nitrate at 80ºC ?
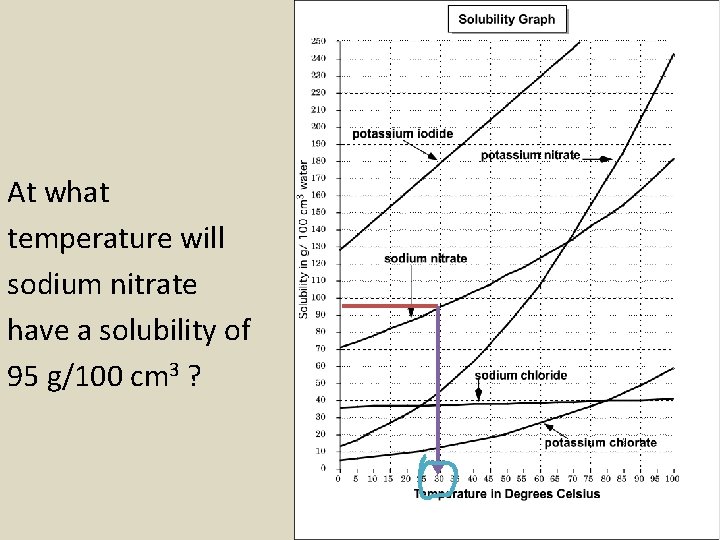
At what temperature will sodium nitrate have a solubility of 95 g/100 cm 3 ?
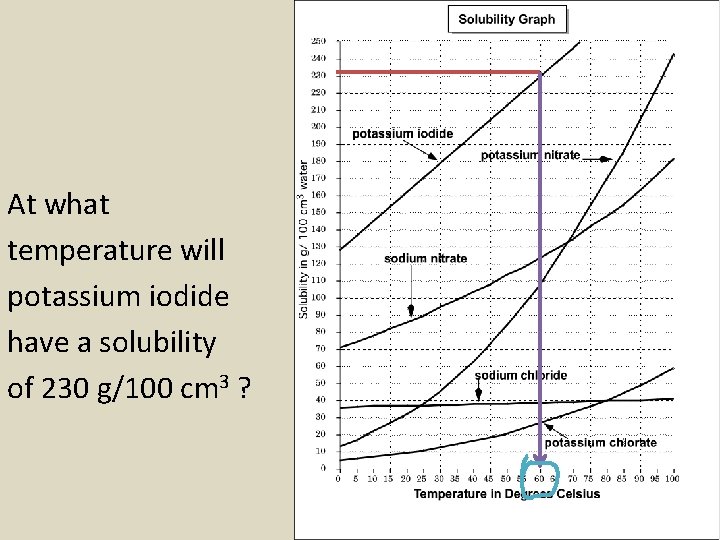
At what temperature will potassium iodide have a solubility of 230 g/100 cm 3 ?
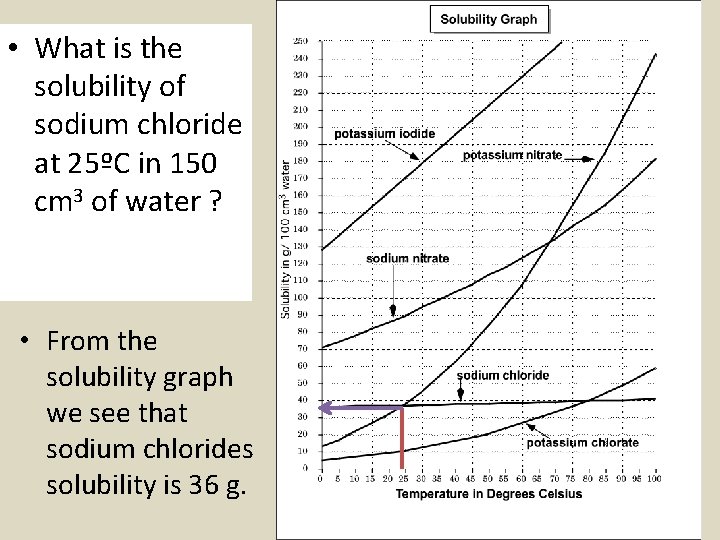
• What is the solubility of sodium chloride at 25ºC in 150 cm 3 of water ? • From the solubility graph we see that sodium chlorides solubility is 36 g.

C. Determine if a solution is saturated, unsaturated, or supersaturated. • If the solubility for a given substance places it anywhere on it's solubility curve it is saturated. • If it lies above the solubility curve, then it's supersaturated, • If it lies below the solubility curve it's an unsaturated solution. Remember though, if the volume of water isn't 100 cm 3 to use a proportion first as shown above.
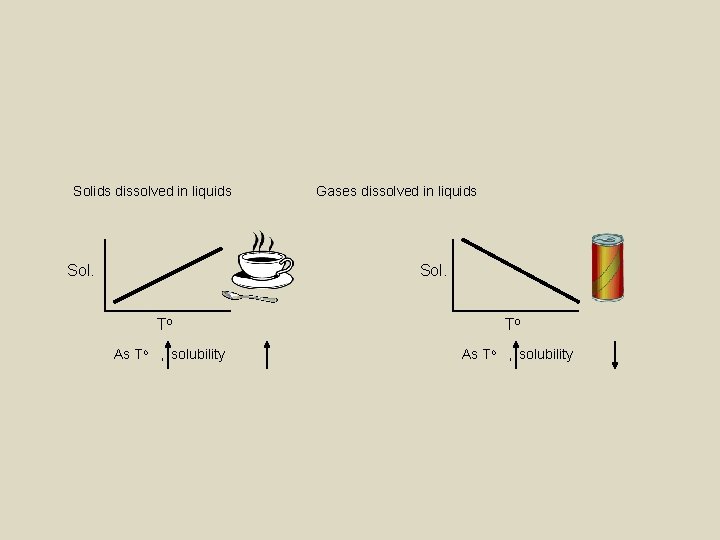
Solids dissolved in liquids Sol. Gases dissolved in liquids Sol. To As To , solubility

Sometimes you'll need to determine how much additional solute needs to be added to a unsaturated solution in order to make it saturated. For example, 30 grams of potassium nitrate has been added to 100 cm 3 of water at a temperature of 50ºC.
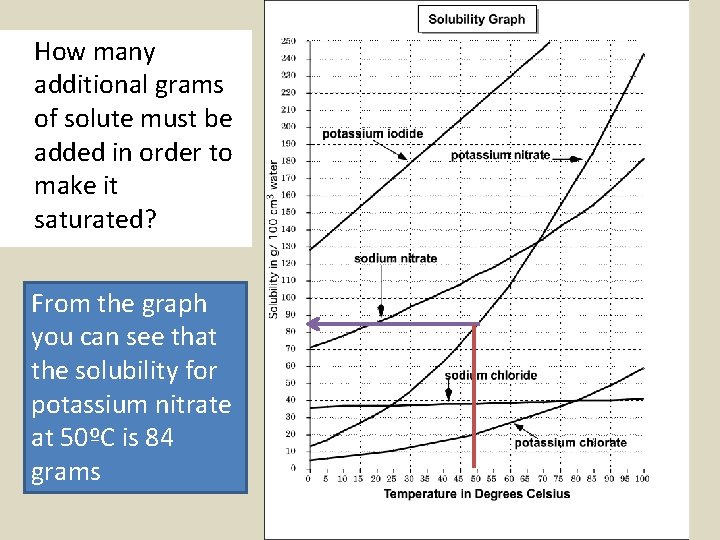
How many additional grams of solute must be added in order to make it saturated? From the graph you can see that the solubility for potassium nitrate at 50ºC is 84 grams

If there already 30 grams of solute in the solution, all you need to get to 84 grams is 54 more grams ( 84 g-30 g )
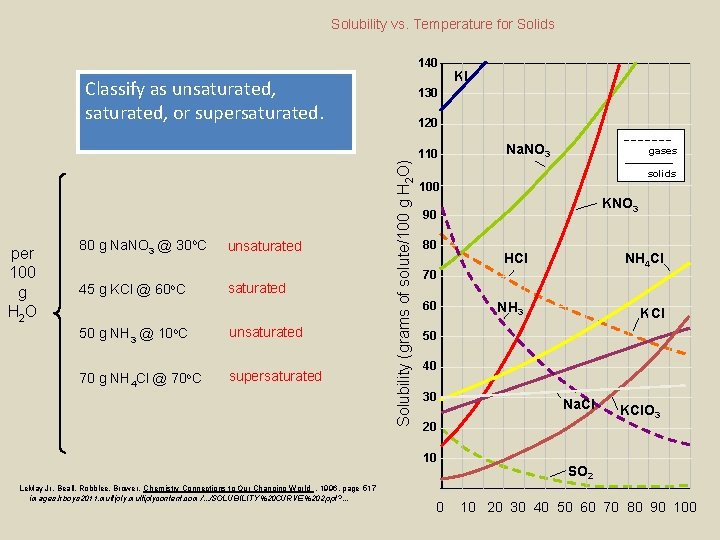
Solubility vs. Temperature for Solids 140 Classify as unsaturated, or supersaturated. 80 g Na. NO 3 @ 30 o. C 45 g KCl @ 60 o. C unsaturated 50 g NH 3 @ 10 o. C unsaturated 70 g NH 4 Cl @ 70 o. C supersaturated 130 120 Solubility (grams of solute/100 g H 2 O) per 100 g H 2 O 110 Na. NO 3 gases solids 100 KNO 3 90 80 HCl NH 4 Cl 70 60 NH 3 KCl 50 40 30 Na. Cl 20 10 Le. May Jr, Beall, Robblee, Brower, Chemistry Connections to Our Changing World , 1996, page 517 images. hboys 2011. multiplycontent. com /. . . /SOLUBILITY%20 CURVE%202. ppt? . . . KI KCl. O 3 SO 2 0 10 20 30 40 50 60 70 80 90 100

Are you now able to…. Calculate solubility using graphs I still feel unsure. I need some more help to understand. I feel ok. I need to do some more work on my own to check my understanding. I am happy and feel I understand can explain the main points.
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Solutions Methods to speed up dissolving Crushing • a solute increases the surface area of the solute allowing more solvent to surround it thus dissolving it faster Ex. Sugar cubes versus granulated sugar

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Solutions Methods to speed up dissolving (cont. ) Heating • a solution increases the energy of the molecules making them move more quickly thus spreading the solute throughout the solvent and speeding up the rate at which the solute dissolves Ex- cooking iced tea
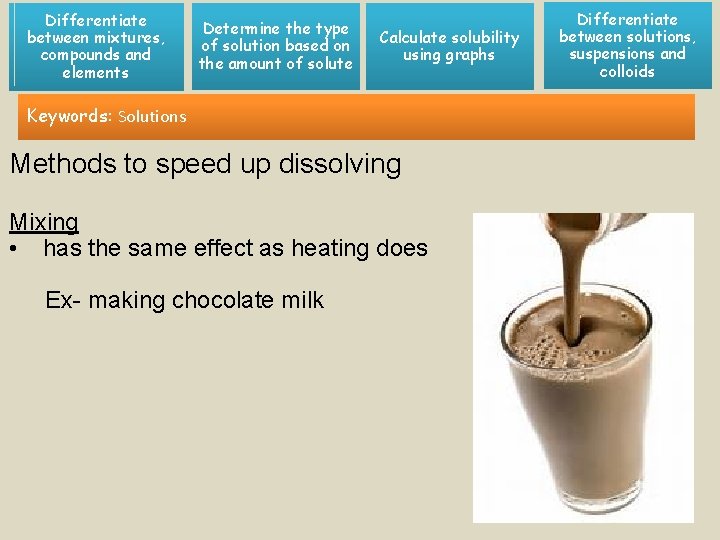
Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Keywords: Solutions Methods to speed up dissolving Mixing • has the same effect as heating does Ex- making chocolate milk Differentiate between solutions, suspensions and colloids


Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Suspensions Suspension: • A suspension is a mixture in which particles of material are dispersed throughout a liquid or gas and are large enough to settle out • The particles are insoluble • Heterogeneous mixtures Ex snow globe

Differentiate between mixtures, compounds and elements Determine the type of solution based on the amount of solute Calculate solubility using graphs Differentiate between solutions, suspensions and colloids Keywords: Colloids • Have properties of both solutions and suspensions • Particles in a colloid are much smaller than particles in a suspension • Colloids have the same definition as suspensions in that a colloid is a mixture in which particles of material are dispersed throughout a liquid or gas, but the particles of a colloid are not large enough to settle out • Particles in a colloid scatter light Ex milk, fog, Jell-o

Are you now able to…. Differentiate between solutions, suspensions and colloids I still feel unsure. I need some more help to understand. I feel ok. I need to do some more work on my own to check my understanding. I am happy and feel I understand can explain the main points.
- Slides: 66