Mixtures and solutions Physical combinations Mixtures and solutions

Mixtures and solutions Physical combinations

Mixtures and solutions o What is a mixture? n A mixture is a physical combination of different substances.

Mixtures and solutions o What are the different types of mixtures? n The 2 types are heterogeneous and homogeneous. o Heterogeneous have an uneven distribution of particles. o Homogeneous have an even distribution of particles.

Mixtures and solutions o What falls in the heterogeneous category? n Mixture: not mixed liquids n Suspension: liquids that settle o Examples: n n Pepper Salt, sand, and iron filings Muddy water Italian salad dressing

Mixtures and solutions o What falls into the homogenous category? n Solutions: mixtures in liquid that look like one substance. n Colloids: mixtures that are evenly distributed, but have larger particles. o Examples n n n Gelatin Vinegar Butter What is the Tyndall effect? What is Brownian motion?

Mixtures and solutions o What is solution concentration? n Concentration is how much substance is dissolved into a solution. o Solvent: substance in higher amount o Solute: dissolves into the solvent n Unsaturated-more room to dissolve. n Saturated-no more room to dissolve. n Super-saturated-more than can be held is dissolved in solution.

Mixtures and solutions o How do we show solution concentration? n There are several ways. . . o o o % by mass % by volume Molarity Molality Mole fraction

Mixtures and solutions o What is % by mass? n Mass of solute X 100 mass of solution What is the % by mass of 3. 6 g of Na. Cl in 100. 0 g of water?

Mixtures and solutions o What is % by volume? n Volume of solute x 100 Volume of solution n What is the % by volume of 35 m. L of ethanol in 155 m. L of water?

Mixtures and solutions o What is molarity? n Molarity is a comparison of moles of solute to liter of solution. o Molarity = moles solute/liter of solution o How strong is 73 grams of hydrogen chloride dissolved in 250 m. L of water? n 8. 0 M

Mixtures and solutions o What is the dilution equation? n M 1 V 1 = M 2 V 2 n M = molarity n V = volume o How can you dilute 250 m. L of 12 M HCl to 8 M HCl?

Mixtures and solutions o What is molality? n Molality is moles of solute per kilogram of solvent. o What is the molality of 4. 5 g of Na. Cl in 100. 0 g of water.

Mixtures and solutions o What is the mole fraction? o What is the mole fraction of 36 g HCl and 64 g of water?

Mixtures and solutions o What is solvation? n Solvation is the process of taking apart ions to make a solution. They become surrounded by polar water molecules. n Water is polar, this means each end has a slight charge due to the unbalanced sharing of electrons.

Mixtures and solutions o What factor affect solvation? n n Temperature changes speed of particles. Surface area changes contact points. Agitation changes speed of particles. Pressure changes some mixtures.

Mixtures and solutions o What happens to the properties of a mixture? n The properties of a mixture take on some of the properties of the substances involved because they keep their chemical structures.

Mixtures and solutions o What are colligative properties? n Colligative properties are changes in properties that depend on the concentration of solute. o o Vapor pressure lowering Boiling point elevation Freezing point depression Osmotic pressure
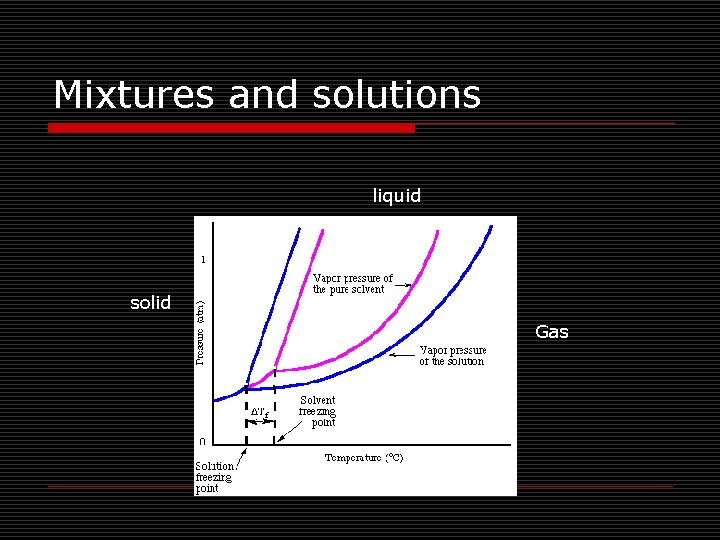
Mixtures and solutions liquid solid Gas
- Slides: 18