MIXTURES AND SOLUTIONS MIXTURE n n a combination

MIXTURES AND SOLUTIONS

MIXTURE n n a combination of substances in which the individual components retain their own properties. EXAMPLES;

SOLUTION n Mixture in which one or more substances are distributed evenly in another substance Examples: SOLUTE- the substance that YOU put into the solution (gets dissolved) SOLVENT- the substance that does the dissolving

How do we measure solutions? n n CONCENTRATION- amount of solute dissolved in a fixed amount of solution. (usually 100 m. L) SATUARATED SOLUTION- when no more solute can be dissolved in the solution

Differences between a mixture and solution n n Solution has a uniform color Mixture is much easier to separate out the substances

ACIDS AND BASES Created by the DISSOCIATION of a water molecule. DISSOCIATION- is the breaking apart of a water molecule H 2 O H+ + OHOH- is known as the HYDROXIDE ION and a high number of these makes the solution a base (alkaline)

ACIDS AND BASES n n The H+ ion can bond with other water molecules to form a HYDRONIUM ION H 3 O + A high amount of HYDRONIUM IONS makes the solution an acid.

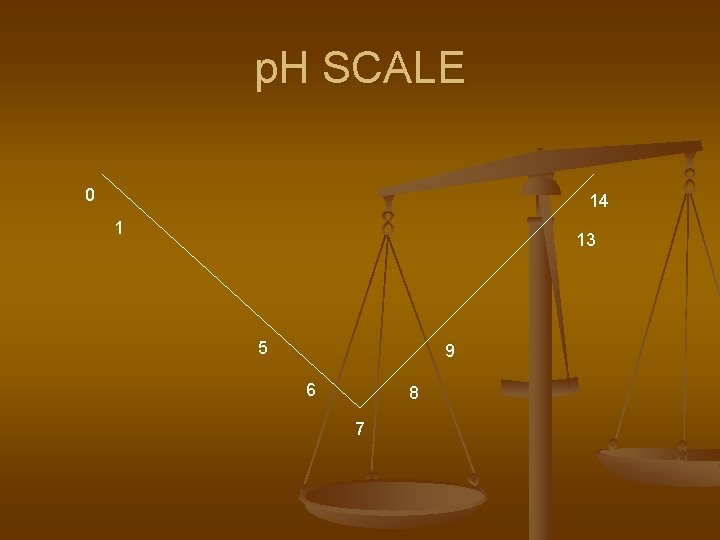
p. H SCALE n n n We measure the acidity or alkalinity (basicity) by finding the ratio of hydroxide ions to hydronium ions. We measure it on a p. H scale of 0 to 14 with 7 being neutral. Pure water has a p. H of 7.

ACID n ACID- has a greater number of hydronium ions -have sour taste (examples) -highly corrosive to metal - 0 to 6. 9 on the p. H scale

BASE- has a greater number of OH- ions -has a bitter taste (examples) - highly corrosive to organic matter -7. 1 to 14 on the p. H scale -feels slippery
p. H SCALE 0 14 1 13 5 9 6 8 7

p. H SCALE How to use the p. H scale. Moving one number away from neutral increases the strength 10 X Example: In order to NEUTRALIZE an acid it is best to use a base of the same strength. Example:
- Slides: 12