Mixtures and Pure Substances All matter is either

Mixtures and Pure Substances All matter is either a pure substance or a mixture Physical and chemical properties show us whether a substance is “pure” or a mixture

Pure Substances • Made of only ONE kind of matter • Has a unique set of properties • May be either an element or a compound
Element Material that cannot be broken down into any simpler substance Are the basic building blocks for all compounds Hydrogen (H) ; Carbon (C) ; Oxygen (O)

Compound Combination of two or more elements in a fixed proportion When elements combine in specific proportions, they form a compound Water (H 2 O) ; Carbon Dioxide (CO 2)

Mixtures • Combination of pure substances However: substances in a mixture do not combine chemically as happens when a compound is formed There are 4 main types of mixtures

Mechanical mixtures Different substances that make up the mixture are visible Also called heterogeneous mixture • Example: soil; package of mixed vegetables

Solution Different substances that make it up are not separately visible One substance is dissolved in another • Example: sugar in coffee; gas dissolved in liquid (Carbonated pop)

Did you Know? • Chemist call a substance dissolved in water an aqueous solution

Suspension Is a cloudy mixture in which tiny particles of one substance are held within another Can be separated when poured through a filter • Example: Tomato juice

Colloid Also a cloudy mixture The particles of the suspended substance are so small that they cannot be easily separated out from the other substance • Example: Milk; Ketchup
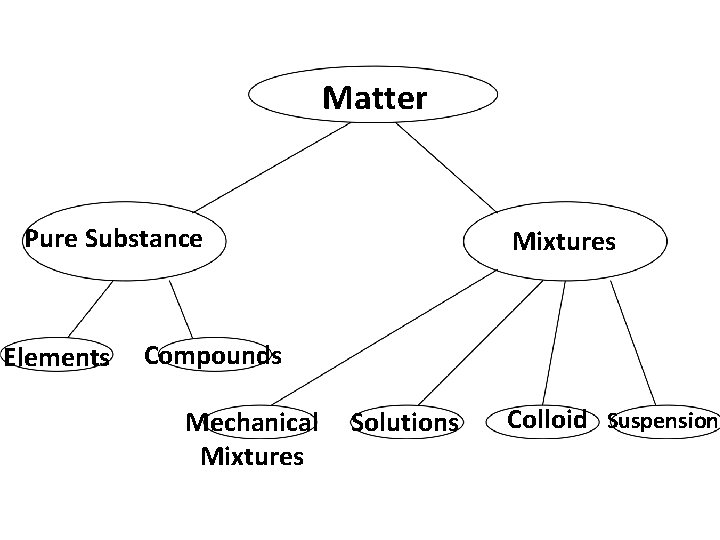
Matter Pure Substance Elements Mixtures Compounds Mechanical Mixtures Solutions Colloid Suspension

Observing Change in Matter • Matter can undergo two types of change • Physical Change One in which a material changes from one state to another Material can also physically change back into its original state

Chemical Change Occurs when two or more materials react and create new materials New materials have completely different properties

Evidence of Chemical Change • Change in Colour • Change in Odour • Formation of a solid or gas • Release or absorption of energy

Freeze Drying Food • Food is Frozen-water turns into ice • Frozen food up in a pressure- pressure reduced until solid turns to gas • Leaves food 10% original mass & does not have to be refrigerated

Meal, Ready to Eat • Uses freeze dried food in a special package called a “Flameless Ration Heater” • Good for Soldiers, astronauts, mountain climbers

Corn based Products used by Humans • Chemical made from corn can be used to make soda pop bottles, remove paint, nail polish, fuel for cars • Products are not as harmful to the environment, renewable form of fuel

Difference between Chemical and Physical Change • Physical Change is a change in state, can be reversed • Chemical Change there is a change in properties, new substance formed.
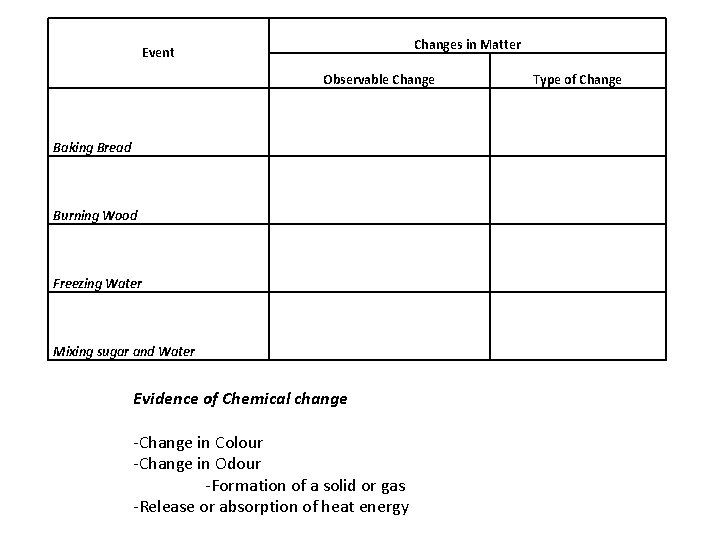
Changes in Matter Event Observable Change Type of Change Baking Bread Burning Wood Freezing Water Mixing sugar and Water Evidence of Chemical change -Change in Colour -Change in Odour -Formation of a solid or gas -Release or absorption of heat energy
- Slides: 19