MIXTURE and COMOUNDS A MIXTURE is a combination

MIXTURE and COMOUNDS A MIXTURE is a combination of two or more substances that are not chemically united and do not exist in fixed proportions to each other. Most natural substances are mixtures. Example air is the mixture of several gases, petroleum etc. MIXTURES PURE COMPOUNDS A mixture can be A pure compound has a physically constant composition with separated into pure fixed ratios of elements. compounds or elements. Mixtures may exhibit a changing set of physical properties. For example, mixture of alcohol and water boils over a range of temperatures. Physical properties such as boiling point or melting point of pure substances are invariant. For example, pure water boils at 100 degrees o. C.

Types of Mixtures • Homogeneous mixture: Having visibly indistinguishable parts. Physical properties are the same throughout the material. A homogeneous mixture a solution (example: vinegar). • Heterogeneous mixtures: Having visibly distinguishable parts. Physical properties are different at different points in a material (example: bottle of ranch dressing).

NEW CHAPTER History of Chemistry

History of Chemistry Antoine Lavoisier in 1783 presented law of conservation of mass (Mass is neither created nor destroyed in a chemical reaction). He is Known as “father of modern chemistry. ” John Dalton (1766– 1844), an English schoolteacher stated that elements were composed of tiny individual particles (atoms), a given compound should always contain the same combination of these atoms.

Dalton’s Atomic Theory In 1808 Dalton published his theory of atoms; 1 - Each element is made up of tiny particles called atoms. 2. The atoms of a given element are identical; the atoms of different elements are different. 3. Chemical compounds are formed when atoms of different elements combine with each other. 4. Chemical reactions involve reorganization of the atoms —changes in the way they are bound together. The atoms themselves are not changed in a chemical reaction.

History of Chemistry has been important since ancient (very old) times. The Greeks about 400 B. C. first explained why chemical changes occur. Greeks had proposed that all matter was composed of four fundamental substances fire, earth, water, and air. The foundations of modern chemistry were laid in the sixteenth century. By the late eighteenth century, combustion had been studied extensively. Antoine Lavoisier (1743– 1794), a French chemist , who finally explained the true nature of combustion.

• In 1809 Gay-Lussac performed experiments in which he measured (under the same conditions of temperature and pressure) the volumes of gases that reacted with each other. • For example, Gay-Lussac found that 2 volumes of hydrogen react with 1 volume of oxygen to form 2 volumes of gaseous water and that 1 volume of hydrogen reacts with 1 volume of chlorine to form 2 volumes of hydrogen chloride.

Avogadro’s Hypothesis In 1811 Avogadro interpreted that; At the same temperature and pressure, equal volumes of different gases contain the same number of particles. (1 mole of any gas contains 6. 022 x 1023 atoms/ions or molecules) Joseph Proust (1754– 1826) presented Law of Constant Composition, also known as the law of definite proportions. • It states that the elemental composition of a pure substance never varies.

DISCOVERY OF ELECTRON • The English physicist J. J. Thomson, performed experiments to understand composition of the atom. He studied electrical discharges in partially evacuated tube called cathode-ray tube. • Thomson found that when high voltage was applied to the tube, a “ray” he called a cathode ray (because it emanated from the negative electrode, or cathode) was produced.

Because this ray was produced at the negative electrode and was repelled by the negative pole and attracted towards positive pole of applied electric field, Thomson postulated that the ray was a stream of negatively charged particles, now called electrons. Thompson also measured the charge/mass ratio of the electron to be 1. 76 108 coulombs/g.

Robert Millikan Experiment In 1909 Robert Millikan, performed an experiments involving charged oil drops. These experiments allowed him to determine the magnitude of the electron charge. With the charge-to-mass ratio determined by Thomson, Millikan was able to calculate the mass of the electron as me = 9. 11 X 10─31 kilogram or me = 9. 11 x 10 ─ 28 g. Charge-tomass ratio of electron is given below; Magnitude of Charge on electron = ─1. 60 x 10 ─19 C Magnitude of Charge/mass ratio (e/m) = ─ 1. 76 x 108 C/g
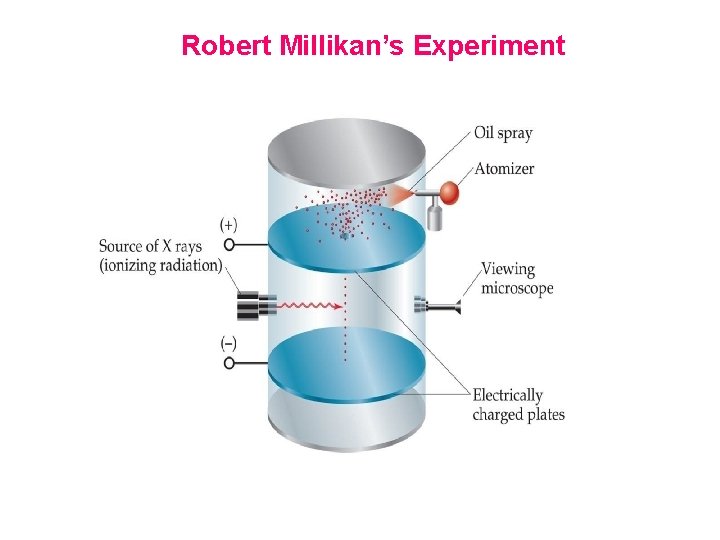
Robert Millikan’s Experiment

In 1911 Ernest Rutherford shot a beam of α-particles at a thin sheet of gold foil and observed the pattern caused by their scattering. α -particles are helium nuclei, 4 He 2+. He observed the particles to travel through the foil with, at the most, very minor deflections in their paths. The results of the experiment were very different from those Rutherford anticipated.
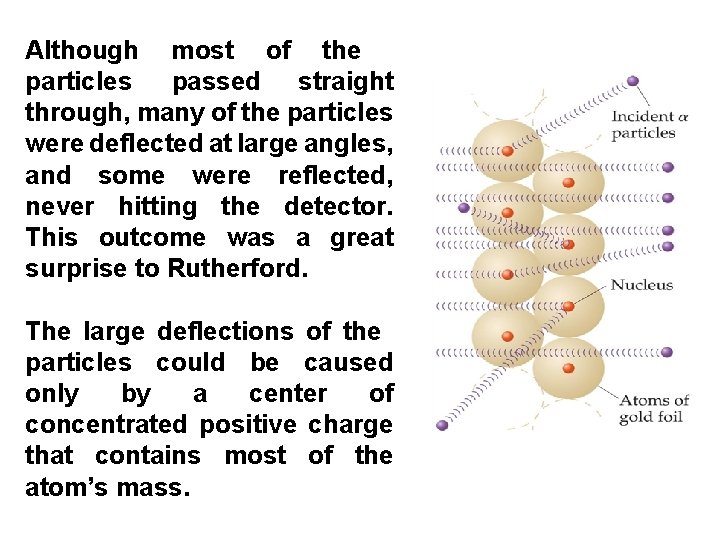
Although most of the particles passed straight through, many of the particles were deflected at large angles, and some were reflected, never hitting the detector. This outcome was a great surprise to Rutherford. The large deflections of the particles could be caused only by a center of concentrated positive charge that contains most of the atom’s mass.

Atoms are made up of three major parts: Part Found Mass Charge Protons Part of Nucleus 1. 7 x 10 -24 g (1. 0 AMU) +1 Neutrons Part of Nucleus Electrons Outside Nucleus 1. 7 x 10 -24 g (1. 0 AMU) 9. 1 x 10 -28 g (small) 0 -1 Notes: 1) Neutral atoms contain equal number of electrons and protons. 2) Atoms can loose or gain electrons to become charged = ions 3) Electrons are reshuffled in a chemical reaction. 4) Number of protons (Atomic number) determine the identity of the atom or ion. 5) Mass of atom = Number of Protons plus Number of Neutrons
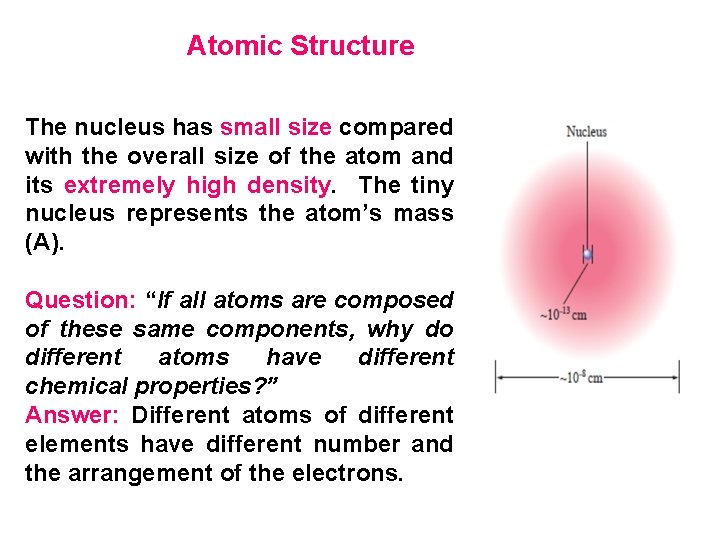
Atomic Structure The nucleus has small size compared with the overall size of the atom and its extremely high density. The tiny nucleus represents the atom’s mass (A). Question: “If all atoms are composed of these same components, why do different atoms have different chemical properties? ” Answer: Different atoms of different elements have different number and the arrangement of the electrons.

The Mass Number (A): The Mass Number of an atom is the total number of protons and neutrons in the atom. The Atomic Number (Z): It is Number of the protons or number of electrons in an atom. All atoms of the same element have the same number of protons or electrons.

Atomic Structure Neutral atoms have the same number of protons and electrons. Ions are charged atoms as Cl ─, OH─ , Na+, H+. Cations = They have more protons than electrons and are positively charged as Na+, H+ Aions = They have more electrons than protons and are negatively charged as Cl ─, OH─. 18

Isotope The term isotope is formed from the Greek roots isos (equal) and topos (place). Hence: "the same place, " meaning that different isotopes of a single element occupy the same position on the periodic table. The number of protons within the atom's nucleus uniquely identifies an element, The number of nucleons (protons and neutrons) in the nucleus is the mass number, and each isotope of a given element has a different mass number.
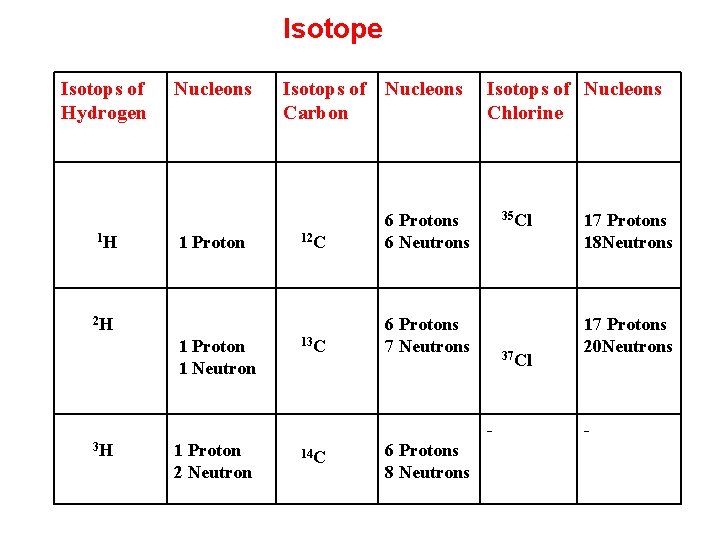
Isotope Isotops of Hydrogen 1 H Nucleons 1 Proton Isotops of Nucleons Carbon 12 C 6 Protons 6 Neutrons 13 C 6 Protons 7 Neutrons 2 H 1 Proton 1 Neutron Isotops of Nucleons Chlorine 35 Cl 37 Cl 3 H 1 Proton 2 Neutron 14 C 6 Protons 8 Neutrons 17 Protons 18 Neutrons 17 Protons 20 Neutrons -

Isotopic Effect A neutral atom has the same number of electrons as protons. Thus, different isotopes of a given element all have the same number of protons and share a similar electronic structure. Because the chemical behavior of an atom is largely determined by its electronic structure, different isotopes exhibit nearly identical chemical behavior. However, heavier isotopes tend to react somewhat more slowly than lighter isotopes of the same element.

Chemical Bonding The forces that hold atoms together in compounds are called chemical bonds. Covalent Bonds: One way that atoms can form bonds is by sharing electrons. These bonds are called covalent bonds, and the resulting collection of atoms is called a molecule. Molecules can be represented by chemical, For example, the formula for carbon dioxide is CO 2, meaning that each molecule contains 1 atom of carbon and 2 atoms of oxygen. Examples of molecules that contain covalent bonds are hydrogen (H 2), water (H 2 O), oxygen (O 2), ammonia (NH 3), and methane (CH 4).
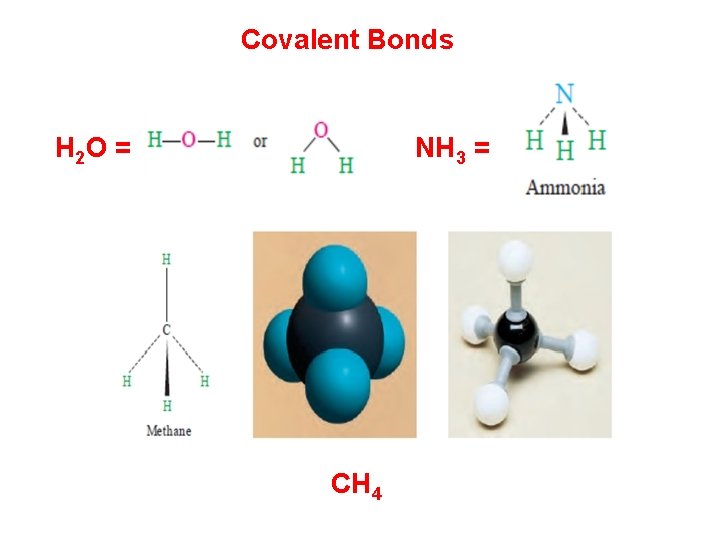
Covalent Bonds H 2 O = NH 3 = CH 4

Ionic Bonds A second type of chemical bond results from attractions among ions. An ion is an atom or group of atoms that has a net positive or negative charge. Common table salt is or sodium chloride (Na. Cl) is example of ionic compound. Sodium (Na) losses one electron and remaining 10 electrons, now has a net 1 charge (Na+)—it has become a positive ion. A positive ion is called a cation. The Chlorine atom gains one electron and 18 electrons produce a net 1 charge; the chlorine has become an ion with a negative charge—an anion (Cl─). The chloride ion is written as Cl, and the process is represented as;

Ionic Bonds

Ionic Bonds Because anions and cations have opposite charges, they attract each other. This force of attraction between oppositely charged ions is called ionic bonding. So, they form Na. Cl. A solid consisting of oppositely charged ions is called an ionic solid. Ionic solids can consist of simple ions, as in sodium chloride, or of polyatomic (many atom) ions, as in ammonium nitrate (NH 4 NO 3), which contains ammonium ions (NH 4) and nitrate ions (NO 3).

Compounds Naming The most important rules for naming compounds other than organic compounds are given below; The systems for naming inorganic binary compounds (compounds composed of two elements) are classified into type-1. Binary Ionic Compounds (Type I) Binary ionic compounds contain a positive ion (cation) always written first in the formula and a negative ion (anion). In naming these compounds, the following rules apply:

Naming Type I Binary Compounds 1. The cation is always named first and the anion second. 2. A monatomic (meaning “one-atom”) cation takes its name from the name of the element. For example, Na is called sodium in the names of compounds containing this ion. 3. A monatomic anion is named by taking the root of the element name and adding -ide. Thus the Cl ion is called chloride.


Type I Binary Compounds Q. Name each binary compound. a. Cs. F b. Al. Cl 3 c. Li. H Solution: a. Cs. F is cesium fluoride. b. Al. Cl 3 is aluminum chloride. c. Li. H is lithium hydride. Notice that, in each case, the cation is named first, and then the anion is named.

Binary Ionic Compounds (Type II) There are many metals that form more than one type of positive ion and thus form more than one type of ionic compound with a given anion. For example, the compound Fe. Cl 2 contains Fe+2 ions, and the compound Fe. Cl 3 contains Fe+3 ions. In a case such as this, the charge on the metal ion must be specified. The systematic names for these two iron compounds are iron (II) chloride and iron (III) chloride, respectively, where the Roman numeral indicates the charge of the cation.

Examples:

Binary Ionic Compounds (Type II) Another system for naming these ionic compounds that is seen in the older literature was used for metals that form only two ions. The ion with the higher charge has a name ending in -ic, and the one with the lower charge has a name ending in -ous. In this system, for example, Fe+3 is called the ferric ion, and Fe+2 is called the ferrous ion. The names for Fe. Cl 3 and Fe. Cl 2 are then ferric chloride and ferrous chloride, respectively.

Binary Covalent Compounds (Type III) Binary covalent compounds are formed between two nonmetals. Although these compounds do not contain ions, they are named very similarly to binary ionic compounds. Naming Binary Covalent Compounds 1. The first element in the formula is named first, using the full element name. 2. The second element is named as if it were an anion. 3. The prefix mono- is never used for naming the first element. For example, CO is called carbon monoxide, not monocarbon monoxide.
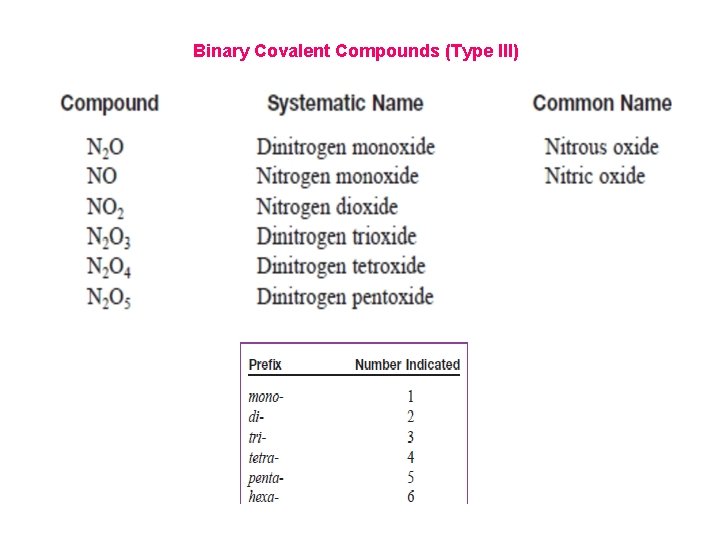
Binary Covalent Compounds (Type III)

Examples:

Acids When dissolved in water, certain molecules produce a solution containing free H+ ions (protons). An acid is a molecule in which one or more H ions are attached to an anion. The rules for naming acids depend on whether the anion contains oxygen. If the name of the anion ends in -ide, the acid is named with the prefix hydro- and the suffix -ic. For example, when gaseous HCl is dissolved in water, it forms hydrochloric acid. When the anion contains oxygen, the acidic name is formed from the root name of the anion with a suffix of ic or -ous, depending on the name of the anion

Acids 1. If the anion name ends in -ate, the suffix -ic is added to the root name. For example, H 2 SO 4 contains the sulfate anion (SO 4)─2 and is called sulfuric acid; H 3 PO 4 contains the phosphate anion (PO 4) ─3 and is called phosphoric acid; and HC 2 H 3 O 2 contains the acetate ion (C 2 H 3 O 2) ─ 1 and is called acetic acid. 2. If the anion has an -ite ending, the -ite is replaced by ous. For example, H 2 SO 3, which contains sulfite (SO 3) ─2, is named sulfurous acid; and HNO , which contains 2 nitrite (NO 2) ─ 1 , is named nitrous acid.
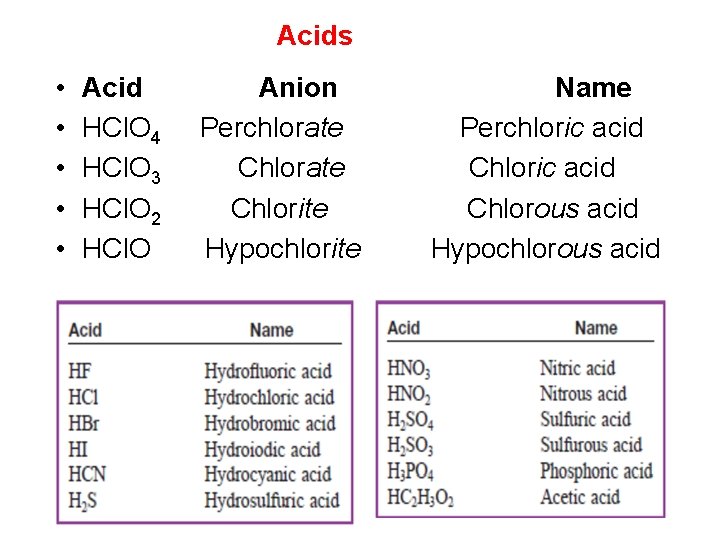
Acids • • • Acid HCl. O 4 HCl. O 3 HCl. O 2 HCl. O Anion Perchlorate Chlorite Hypochlorite Name Perchloric acid Chlorous acid Hypochlorous acid

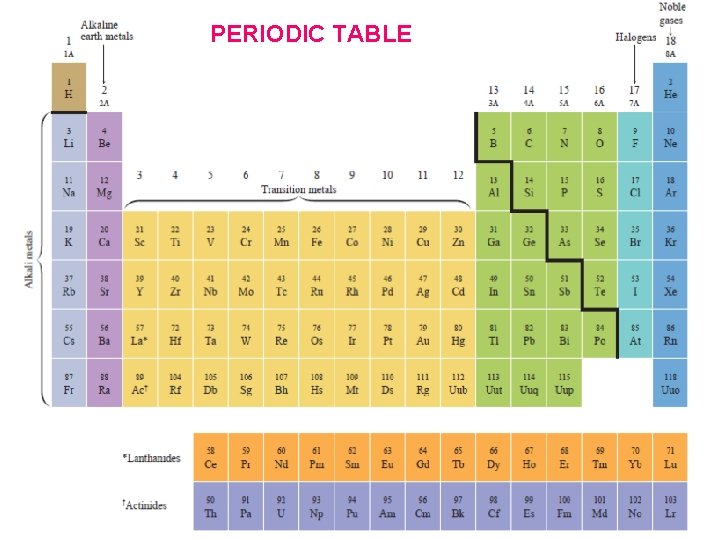
Periodic Table Mendeleev’s Periodic Table • Mendeleev (1869) arranged elements in the periodic table by their increasing mass numbers. • He left open spaces in his periodic table to account for elements not yet discovered. • However, It was rejected due to some serious flaws.

Henry Moseley In 1913, through his work with X-rays, he determined the actual nuclear charge (atomic number) of the elements*. He rearranged the elements in order of increasing atomic number. *“There is in the atom a fundamental quantity which increases by regular steps as we pass from each element to the next. This quantity can only be the charge on the central positive nucleus. ” 1887 - 1915

Periodic Law When elements are arranged in order of increasing atomic number, there is a periodic pattern in their physical and chemical properties.
PERIODIC TABLE

PERIODS Li, Be, B, C, N. O, F, Ne The horizontal rows of the periodic table are called PERIODS. There are 7 periods in the periodic table. GROUPS or FAMILIES The vertical columns of the periodic table are called GROUPS, or FAMILIES. H Li Na K B Cs Fr The elements in any group of the periodic table have similar physical and chemical properties! There are 8 groups in the periodic table.

The Periodic Table Becomes Useful!! • Elements in the same group have similar chemical and physical properties!! • (Mendeleev did that on purpose. ) Why? ? • They have the same number of valence electrons. • They will form the same kinds of ions.
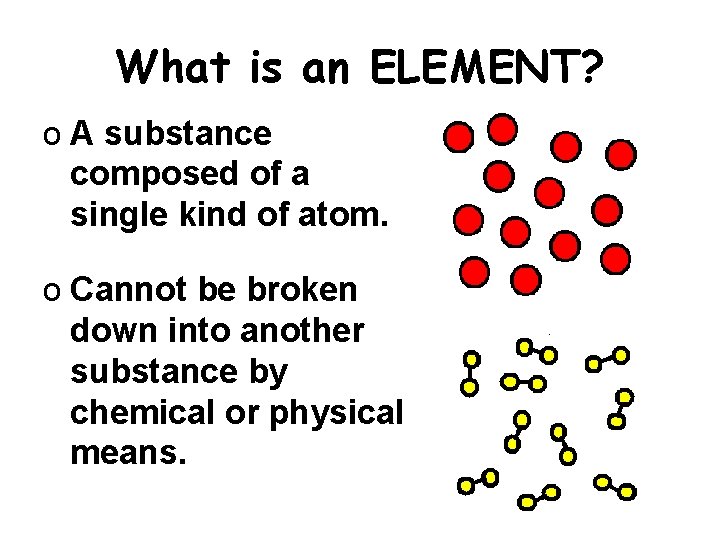
What is an ELEMENT? o A substance composed of a single kind of atom. o Cannot be broken down into another substance by chemical or physical means.
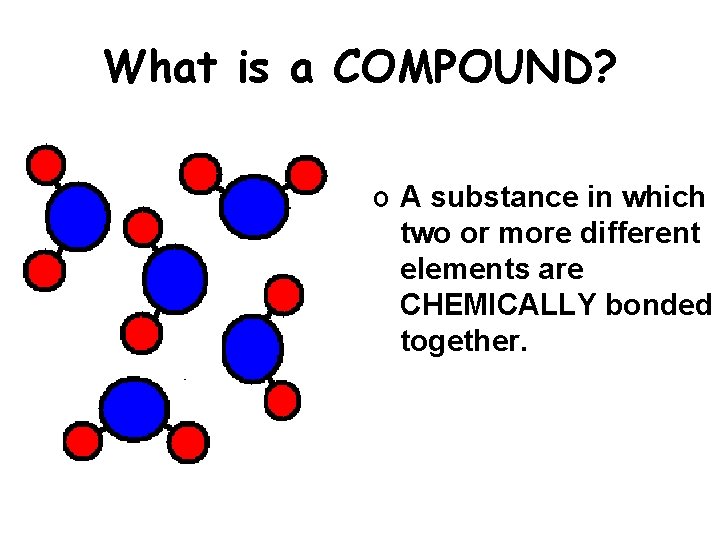
What is a COMPOUND? o A substance in which two or more different elements are CHEMICALLY bonded together.
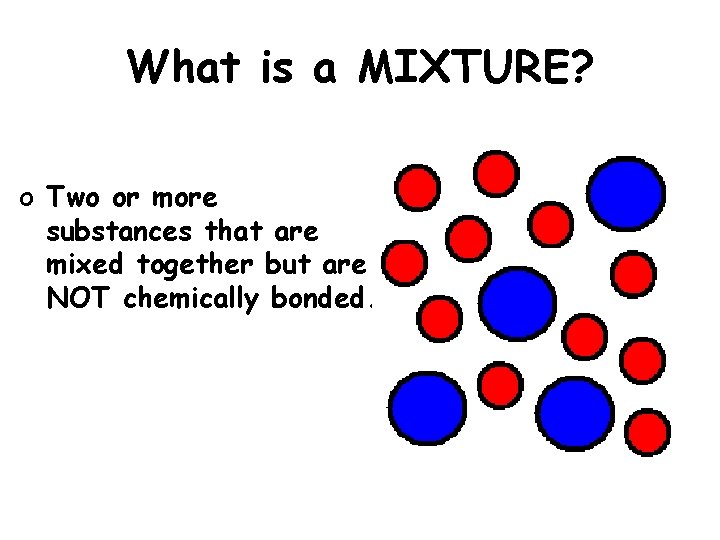
What is a MIXTURE? o Two or more substances that are mixed together but are NOT chemically bonded.
The periodic table is the most important tool in the chemist’s toolbox!

What Millikan did was to put a charge on a tiny drop of oil, and measure how strong an applied electric field had to be in order to stop the oil drop from falling. Since he was able to work out the mass of the oil drop, and he could calculate the force of gravity on one drop, he could then determine the electric charge that the drop must have. By varying the charge on different drops, he noticed that the charge was always a multiple of -1. 6 x 10 -19 C, the charge on a single electron. This meant that it was electrons carrying this unit charge. Here's how it worked. Have a look at the apparatus he used:

An atomizer sprayed a fine mist of oil droplets into the chamber. Some of these tiny droplets fell through a hole in the upper floor. Millikan first let them fall until they reached terminal velocity. Using the microscope, he measured their terminal velocity, and by use of a formula, calculated the mass of each oil drop. Next, Millikan applied a charge to the falling drops by illuminating the bottom chamber with x-rays. This caused the air to become ionized, and electrons to attach themselves to the oil drops. By attaching a battery to the plates above and below this bottom chamber, he was able to apply an electric voltage. The electric field produced in the bottom chamber by this voltage would act on the charged oil drops; if the voltage was just right, the electromagnetic force would just balance the force of gravity on a drop, and the drop would hang suspended in mid-air.

Now you try it. Click here to open a simulation of Millikan's chamber. First, allow the drops to fall. Notice how they accelerate at first, due to gravity. But quickly, air resistance causes them to reach terminal velocity. Now focus on a single falling drop, and adjust the electric field upwards until the drop remains suspended in mid-air. At that instant, for that drop, the electric force on it exactly equals the force of gravity on it. Some drops have more electrons than others, so will require a higher voltage to stop. When you've finished playing with the apparatus, close the window and we'll continue. http: //highered. mcgrawhill. com/sites/0072512644/student_view 0/chapter 2/animations_center. h tml#
- Slides: 52