Mitralign Program Update Lutz Buellesfeld MD FSCAI FESC

Mitralign Program Update Lutz Buellesfeld MD FSCAI FESC Department of Cardiology Bern University Hospital Switzerland

Lutz Buellesfeld, MD Consulting: Mitralign Medtronic Core. Valve Edwards Lifesciences, LLC Abbott Vascular
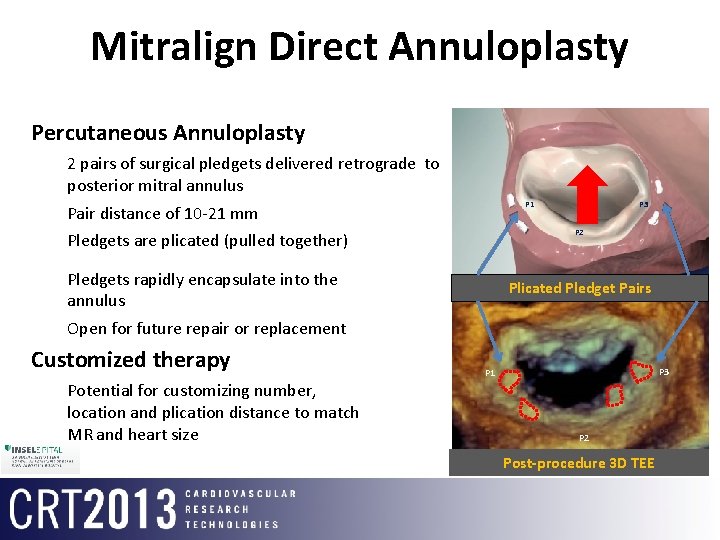
Mitralign Direct Annuloplasty Percutaneous Annuloplasty 2 pairs of surgical pledgets delivered retrograde to posterior mitral annulus P 1 Pair distance of 10 -21 mm P 3 P 2 Pledgets are plicated (pulled together) Pledgets rapidly encapsulate into the annulus Plicated Pledget Pairs Open for future repair or replacement Customized therapy Potential for customizing number, location and plication distance to match MR and heart size P 3 P 1 P 2 Post-procedure 3 D TEE
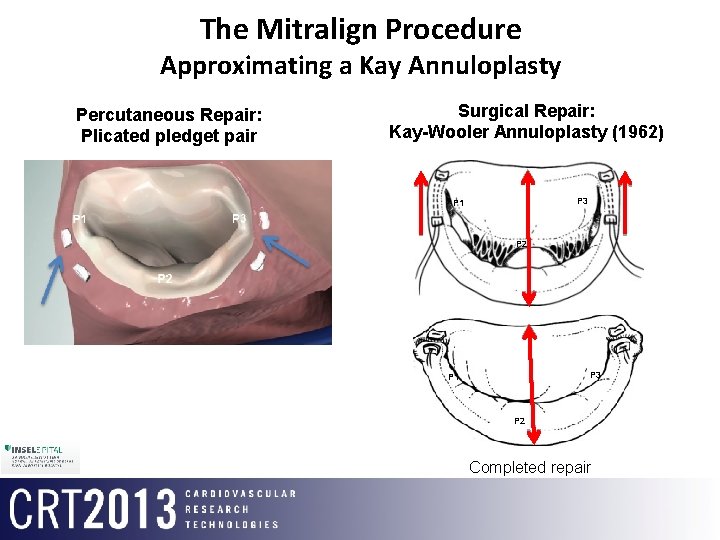
The Mitralign Procedure Approximating a Kay Annuloplasty Percutaneous Repair: Plicated pledget pair Surgical Repair: Kay-Wooler Annuloplasty (1962) P 3 P 1 P 2 Completed repair
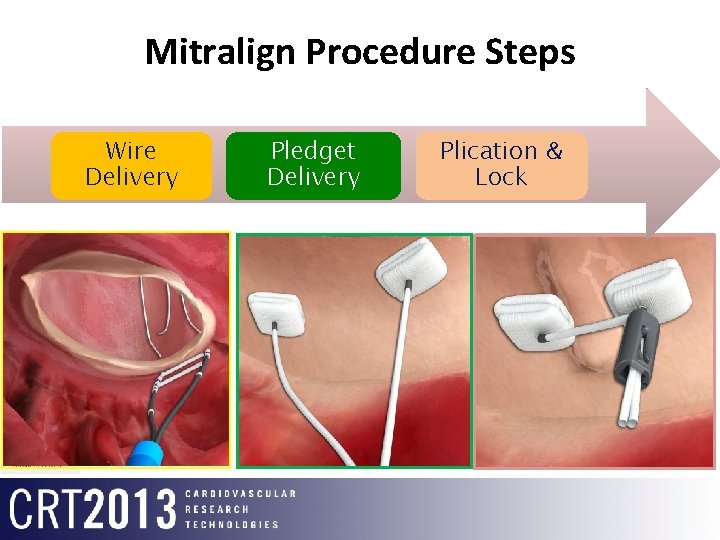
Mitralign Procedure Steps Wire Delivery Pledget Delivery Plication & Lock
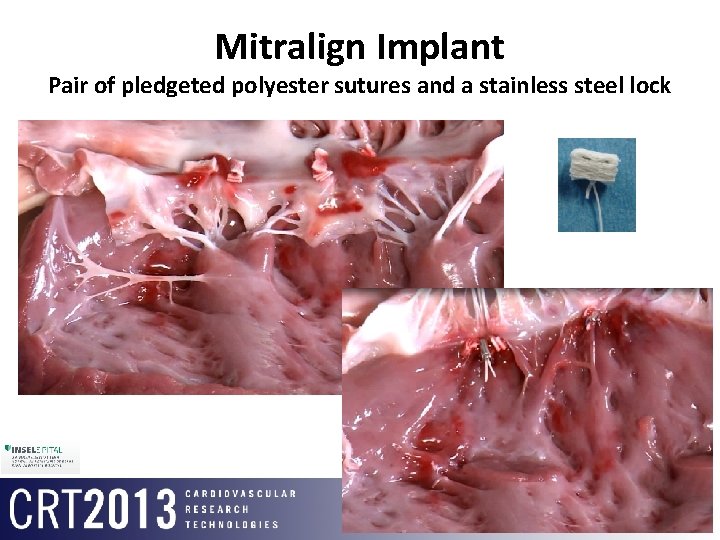
Mitralign Implant Pair of pledgeted polyester sutures and a stainless steel lock
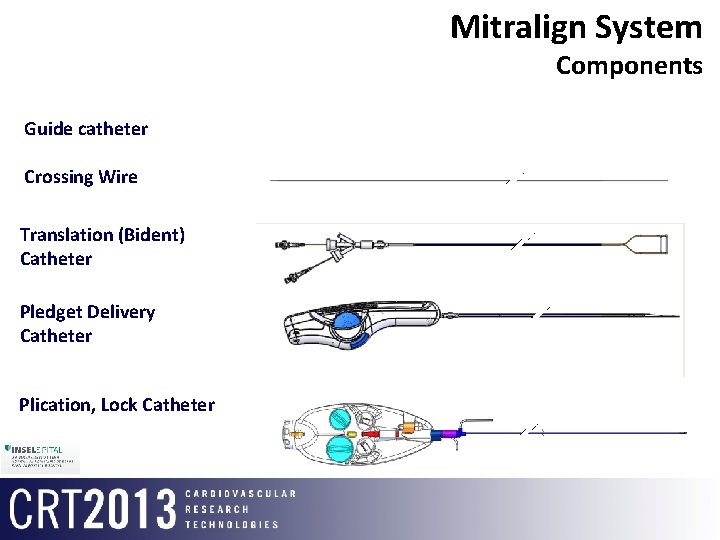
Mitralign System Components Guide catheter Crossing Wire Translation (Bident) Catheter Pledget Delivery Catheter Plication, Lock Catheter
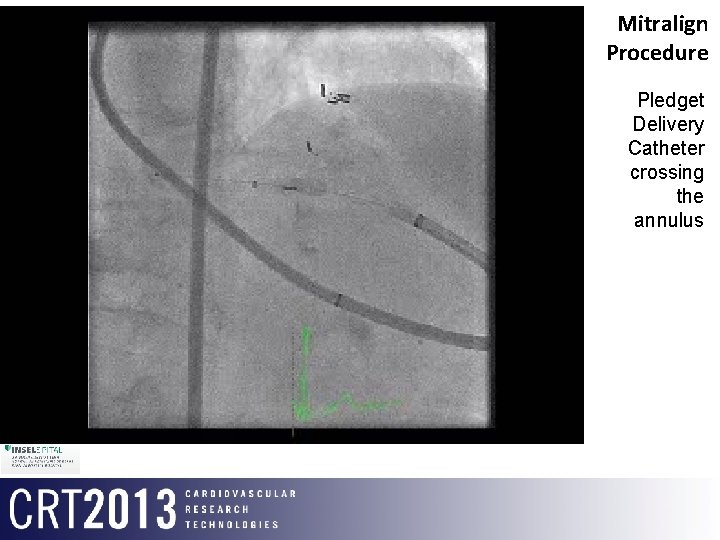
Mitralign Procedure Pledget Delivery Catheter crossing the annulus
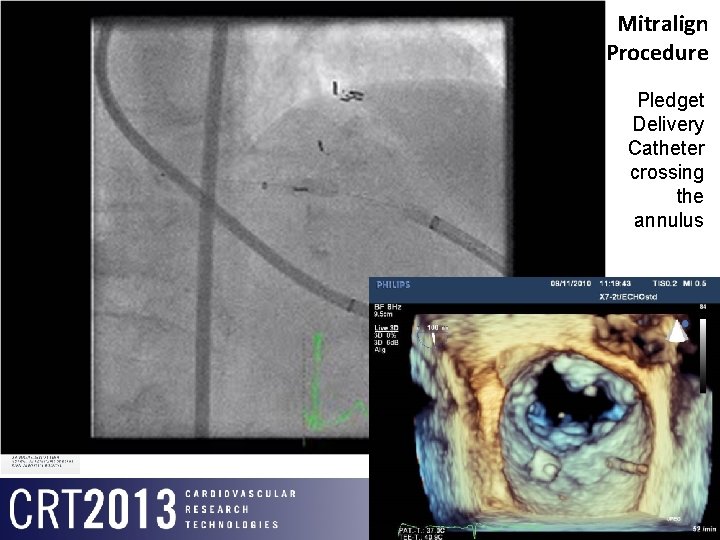
Mitralign Procedure Pledget Delivery Catheter crossing the annulus
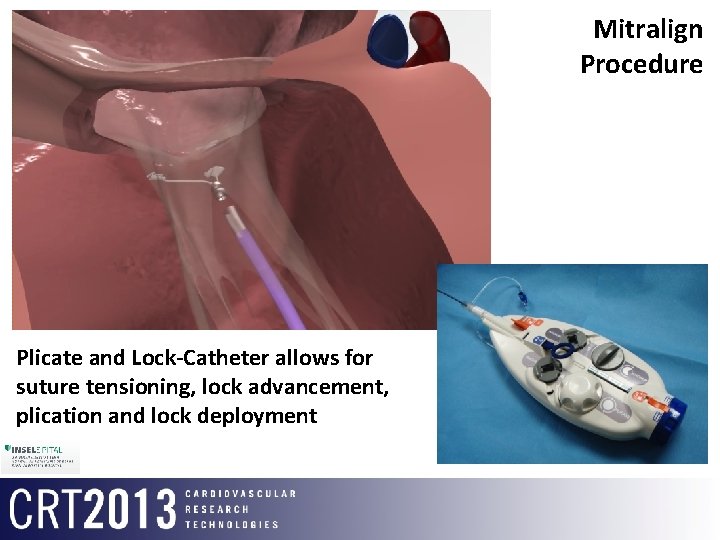
Mitralign Procedure Plicate and Lock-Catheter allows for suture tensioning, lock advancement, plication and lock deployment
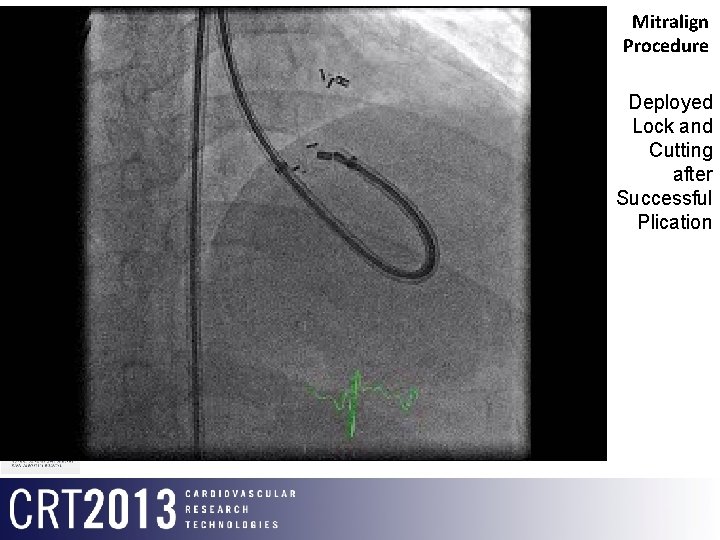
Mitralign Procedure Deployed Lock and Cutting after Successful Plication
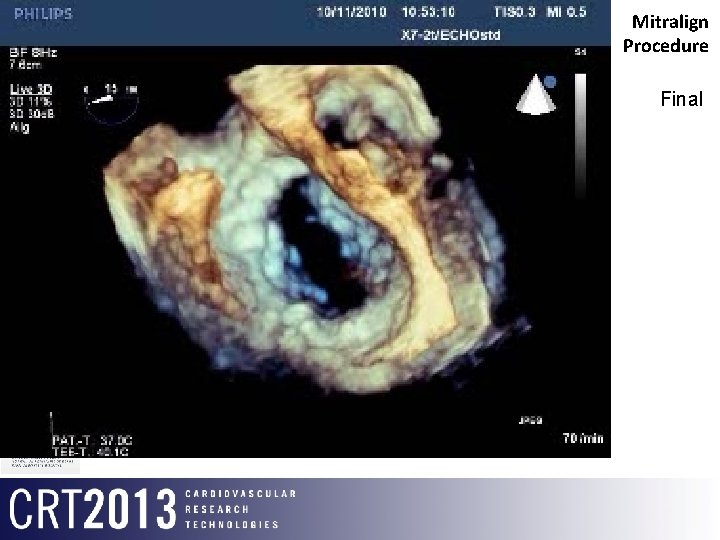
Mitralign Procedure Final
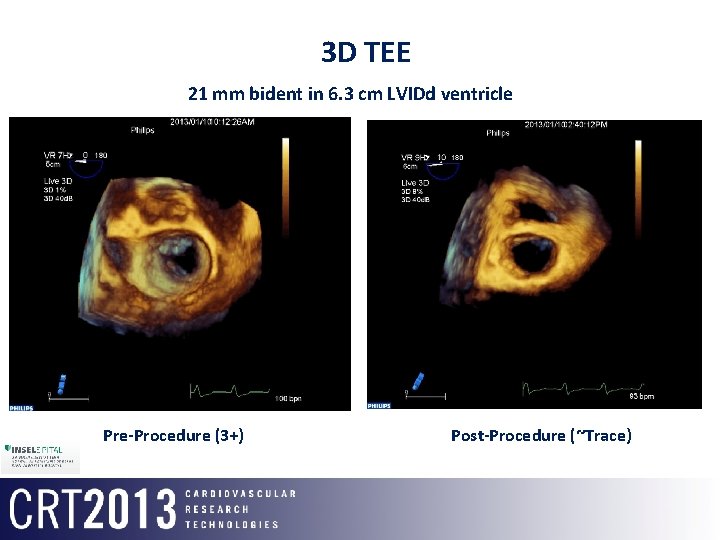
3 D TEE 21 mm bident in 6. 3 cm LVIDd ventricle Pre-Procedure (3+) Post-Procedure (~Trace)
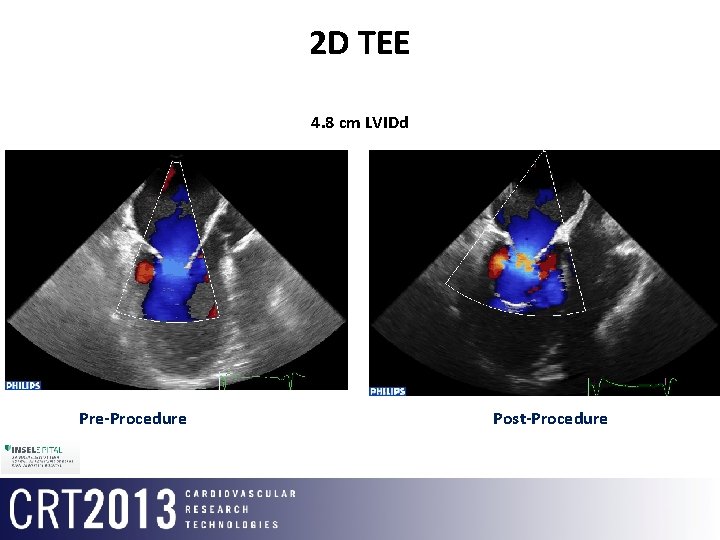
2 D TEE 4. 8 cm LVIDd Pre-Procedure Post-Procedure

Feasibility Study Design and Patient Population • Design: Prospective, single-arm, multicenter, safety and feasibility study • Patient Population: Symptomatic Patients with Functional Mitral Regurgitation • Time points: Baseline, discharge, 30 day, 6 month, 12 -36 months • Enrollment: • Study sites: CAUTION: Investigational Device Only. Not approved for sale in the United States 26 of 36 patients completed
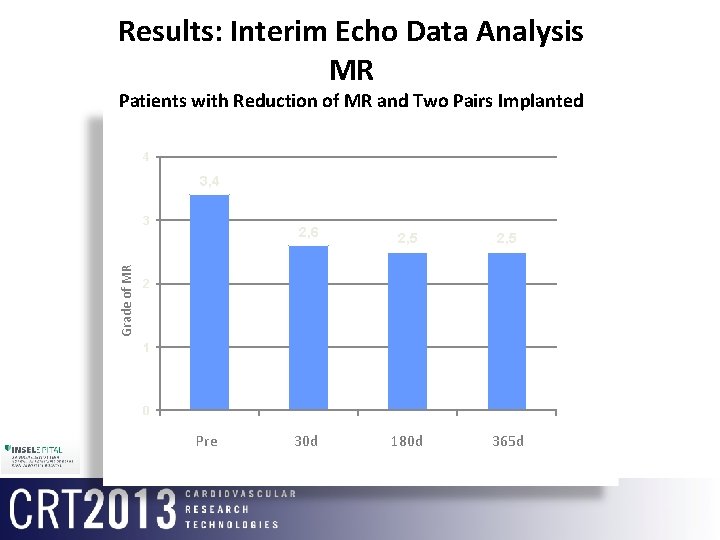
Results: Interim Echo Data Analysis MR Patients with Reduction of MR and Two Pairs Implanted 4 3, 4 Grade of MR 3 2, 6 2, 5 2 30 d 3 180 d 4 365 d 2 1 0 1 Pre

Results: Interim Echo Data Analysis LV Dimensions 0, 20 0, 30 0, 00 ∆ LVESD (cm) Δ LVEDD (cm) LV End Diastolic Diameter -0, 20 -0, 40 -0, 60 0, 10 -0, 30 -0, 50 -0, 70 Baseline 30 D 6 M 12 M Baseline 20, 00 LV End Diastolic Volume ∆ LVESV (ml) 0, 00 ∆ LVEDV (ml) LV End Systolic Diameter -20, 00 -40, 00 -60, 00 -80, 00 Baseline 30 D 6 month timepoint n=10, 12 -month timepoint n=5 6 M 12 M 20, 00 10, 00 -10, 00 -20, 00 -30, 00 -40, 00 -50, 00 -60, 00 30 D 6 M 12 M LV End Systolic Volume Baseline 30 D 6 M 12 M

Mitralign Regulatory Status • CE Marking Strategy: Feasibility Study + Expanded Use Study = CE Mark • Target enrollment completion: Q 2 2013 • US Strategy: • Feasibility Study commencement: Targeted 2013

Conclusions • Mitralign annuloplasty with the new BIDENT System is a safe and feasible procedure • Mitralign annuloplasty has the potential to reduce FMR, inducing a reverse LV remodeling and improving clinical symptoms • Early results are promising and further studies are ongoing / in planning to demonstrate the efficacy of this approach

Thank you
- Slides: 20