Mitochondria and the Tri Carboxylic Acid Cycle TCA

Mitochondria and the Tri. Carboxylic Acid Cycle (TCA or Citric acid cycle) are central to energy production in eukaryotic cells! 10/14 and 10/16 • Overview of intracellular metabolism • What are the 4 Stages of aerobic energy production? • How are mitochondrial organized inside? • Steps in TCA: structures, enzymes, and energy compounds, and carbon dioxide. • The energy Balance sheet for oxidation of a glucose to carbon dioxide. 1 glucose 38(sometimes 36)ATP + 6 CO 2 • Fatty acids are oxidized by Beta Oxidation • Amino acids “can” become glucose, though the process is VERY expensive.
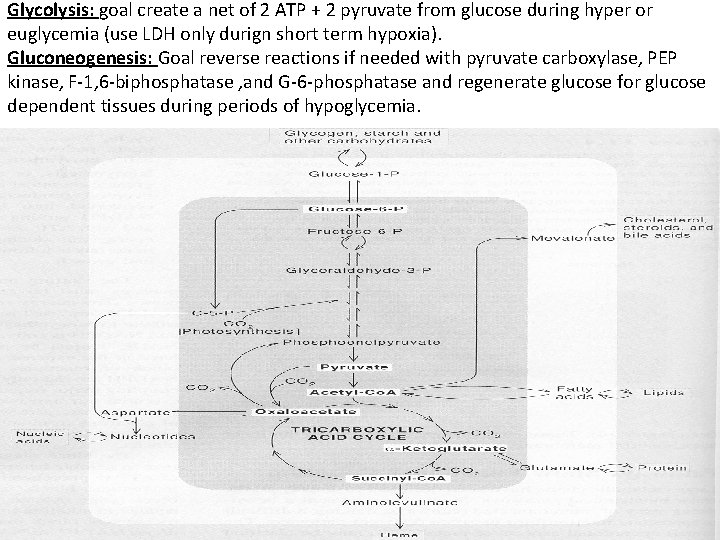
Glycolysis: goal create a net of 2 ATP + 2 pyruvate from glucose during hyper or euglycemia (use LDH only durign short term hypoxia). Gluconeogenesis: Goal reverse reactions if needed with pyruvate carboxylase, PEP kinase, F-1, 6 -biphosphatase , and G-6 -phosphatase and regenerate glucose for glucose dependent tissues during periods of hypoglycemia.

There are 4 steps in aerobic respiration: • Step 1: Glycolysis in cytosol – Glucose 2 Pyruvate+2 ATP+2 NADH+ 2 H+ • Step 2: Tricarboxylic Acid Cycle/Acetyl-CO: Occurs in mitochondrial matrix – Pyruvate oxidized to carbon dioxide – Priming: 2 Pyruvate 2 Acetly-Co. A + 2 CO 2 – Cycle yields CO 2, ATP, FADH and NADH • Step 3: Electron Transport and Proton Pump • Occurs on inner mitochondrial membrane – Energy in electrons pumps. H+ out of matrix! – Last Step: 4 e- and 4 H+ bind O 2 2 H 2 O • Step 4: ATP synthesis on inner membrane – Let H+ run back though a ATP-synthase enzyme and phosphorylate ADP to ATP!

Mitochondria are central to the huge ATP yield that the tricarboxylic acid cycle makes possible for eukaryotic cells. • Organelle with its own DNA: • Location inside cell: • Mitochondrial density relative to tissue type: – – Aerobic tissues: Anaerobic tissues: Why Light and Dark Turkey meat? Liver and kidney? • Maternal Inheritance of mitochondria: – You got it all from your mamma! • Mitochondrial Parts: – Matrix, Crista, Inner/Outer membranes, Porins – Unique double membrane system – Bacteria are sort of complex mitochondria
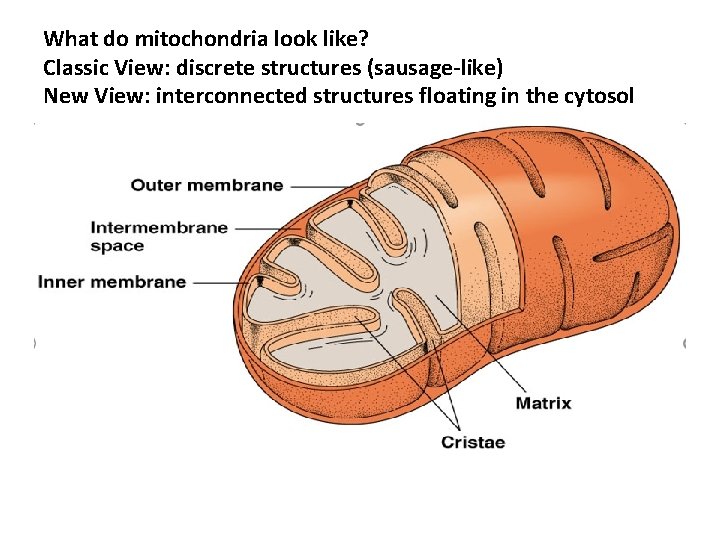
What do mitochondria look like? Classic View: discrete structures (sausage-like) New View: interconnected structures floating in the cytosol
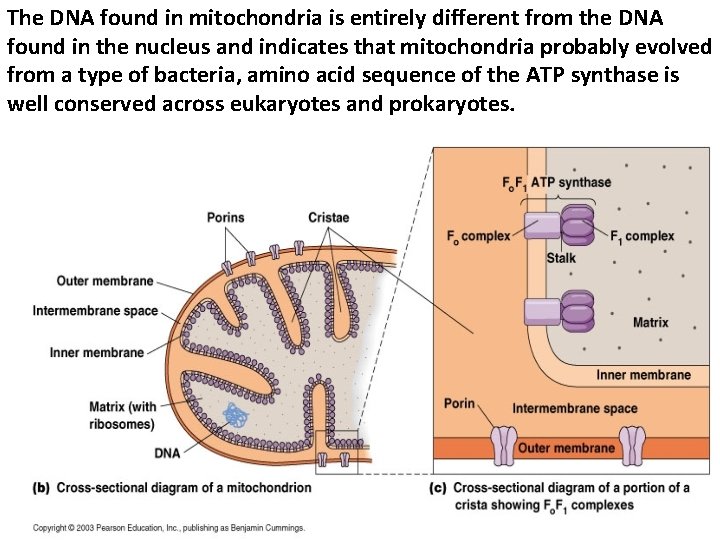
The DNA found in mitochondria is entirely different from the DNA found in the nucleus and indicates that mitochondria probably evolved from a type of bacteria, amino acid sequence of the ATP synthase is well conserved across eukaryotes and prokaryotes.

Tricarboxylic Acid Cycle (TCA/Krebs Cycle) is the CENTRAL HUB for oxidation and energy production from sugars, fatty acids, and some amino acids! TCA is called a “cycle” because the last step creates the substrate for the first step! Acetyl-Co. A is main entry molecule! • Glucose Acetyl-Co. A • Fatty Acids Acetyl-Co. A • Amino Acids Some make Acetyl-Co. A Some aa turned into Glucose ( Acetyl-Co. A) Complete Oxidation of one Acetyl-Co. A GTP+3 NADH+FADH 2+2 CO 2 • GTP ATP • NADH 3 ATP • FADH 2 2 ATP Acetyl-Co. A 1 ATP+9 ATP+2 CO

What are the key steps in TCA? Please work at recognizing compound names, structures, enzymes and locations of energy generation? • Overview: CH 3 -CO-S-Co. A 2 CO 2 • Entry Point: • Oxaloacetate merges with Acetyl-Co. A • Citrate is formed (3 carboxyl groups; TCA) • Last Step in Cycle: • Oxaloacetate(OAA) re-created from malate • Starting point is recycled! • VVIP: Remember: removal of any one intermediate in the cycle prevents OAA regeneration! @Intermediates back up into glycolysis too!@
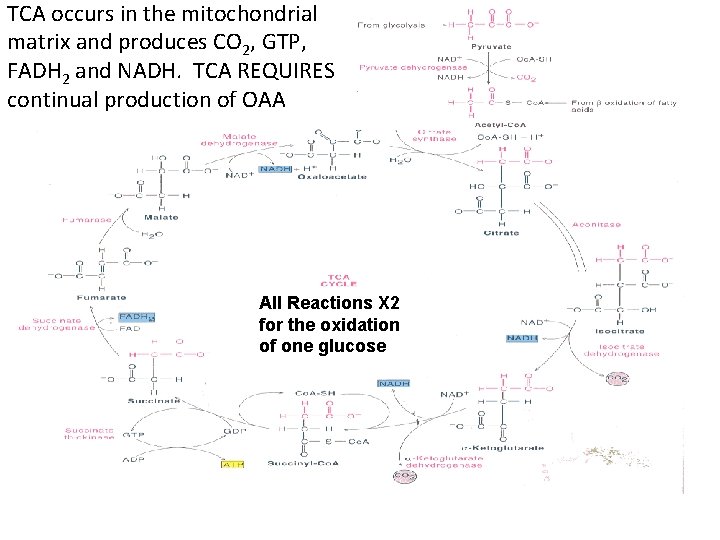
TCA occurs in the mitochondrial matrix and produces CO 2, GTP, FADH 2 and NADH. TCA REQUIRES continual production of OAA All Reactions X 2 for the oxidation of one glucose

Energy Balance Sheet for complete oxidation of a single glucose molecule to carbon dioxide. • Glycolysis in Cytosol: (8 ATP) – – 1 glucose 2 pyruvate + 2 ATP + 2 NADH System feeds forward only if NAD+ is available Aerobic and sometimes anaerobic Pyruvate transported into matrix • Pyruvate Decarboxylase in Matrix: (6 ATP) 2 pyruvate 2 Acetyl-Co. A+2 NADH + 2 H+ and 2 CO 2 • TCA in Matrix: (24 ATP) – 2 Acetyl-Co. A 4 CO 2+2 GTP+2 FADH 2+6 NADH – Conversion to ATP 2 ATP + 4 ATP + 18 ATP • Net Yield: 1 glucose 8+6+24=38 ATP (assumes oxygen ) • Sometimes a less efficient system is used to transport cytosolic NADH into the mitochondrial matrix such that these two NADHs yield only 4 ATP, not 6.

Remember that a negative Delta G can help drive a reaction with a positive value if there is “No Membrane In Between”. This is why Delta G from the reactions in the mitochondria cannot help to drive the reactions of glycolysis in the cytosol. Although molecules such as pyruvate can “carry” the energy between different compartments.
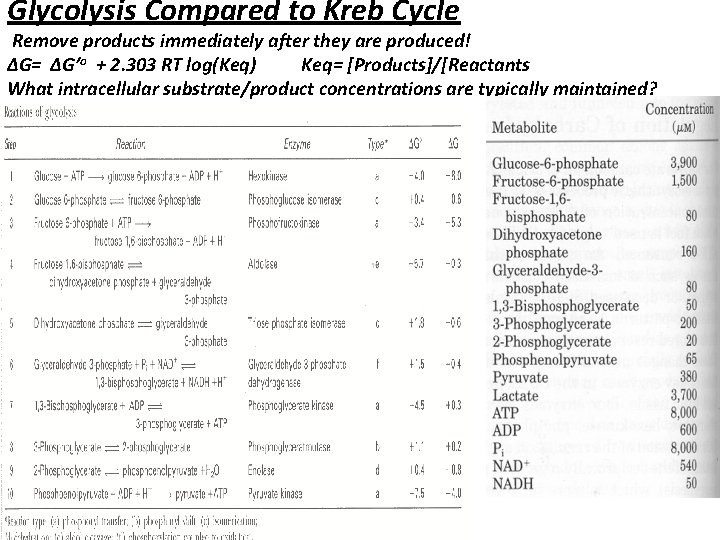
Glycolysis Compared to Kreb Cycle Remove products immediately after they are produced! ∆G= ∆G’o + 2. 303 RT log(Keq) Keq= [Products]/[Reactants What intracellular substrate/product concentrations are typically maintained?

Beta-oxidation: is an enzyme pathway that oxidizes fatty acids into acetyl-Co. A molecules that feed directly into TCA which produces 3 NADH, 1 FADH 2 and 1 ATP for each ACETYL-Co. A! Fatty Acids: 9 kcal/g Carbohydrates: 4 kcal/g Amino. Acids: 4 kcal/g Triglycerides + Lipase Glycerol + 3 fatty acids C-16 palmitate 8 acetyl-Co. A(7 cuts) Each acetyl-Co. A TCAX 8 (8 X 3 NADH)+(8 X 1 FADH 2)+(8 X 1 GTP) 72 ATP + 16 ATP + 8 ATP = 96 ATP PLUS: each time acetyl-Co. A cut off the FA chain: 1 NADH+1 FADH 2 created Additional Energy: (7 XNADH)+(7 XFADH 2) (7 X 3)+ (7 X 2)= 35 ATP Total oxidation of palmitic acid to CO 2= 96+35=131 (130)ATP Compare to the 38 ATP from a single glucose molecule! Fats create tons of ATP!
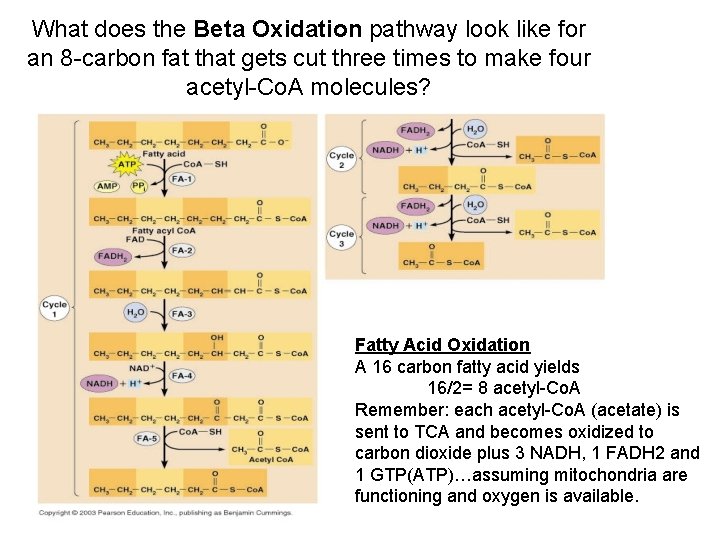
What does the Beta Oxidation pathway look like for an 8 -carbon fat that gets cut three times to make four acetyl-Co. A molecules? Fatty Acid Oxidation A 16 carbon fatty acid yields 16/2= 8 acetyl-Co. A Remember: each acetyl-Co. A (acetate) is sent to TCA and becomes oxidized to carbon dioxide plus 3 NADH, 1 FADH 2 and 1 GTP(ATP)…assuming mitochondria are functioning and oxygen is available.

If fat yields SO MUCH energy why don’t cells burn fat all the time? Problems: • Fatty acid solubility in blood is a problem for transport! • Fatty acids not always completely oxidized to carbon dioxide! • Beta-oxidation requires plenty of oxygen and mitochondria! Beta-oxidation creates toxins during a diabetic crisis: • 1) Acetone • 2) Acetoacetone • 3) Ketones • 4) Beta-hydroxybutyric acid

Why do amino acids make a poor fuel for making ATP? Answer: It is really expensive AND potentially toxic! • 1) Use results in protein breakdown!! expensive • 2) Not all A. A. can feed into glucose or the TCA!! expensive • 3) Ammonia and urea are created by degradation!! toxic • 4) Ketones are created by accident!! Toxic • During Starvation: Proteolysis occurs in liver cells so glucose can be produced for the other cells that MUST use glucose, like glucose dependent red blood cells. • These are “gluconeogenic” amino acids! • Some amino acids “can” feed into TCA following transamination!
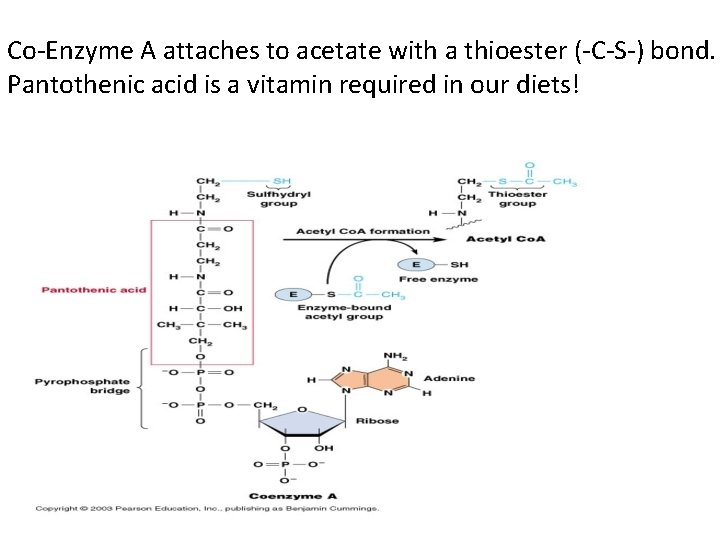
Co-Enzyme A attaches to acetate with a thioester (-C-S-) bond. Pantothenic acid is a vitamin required in our diets!
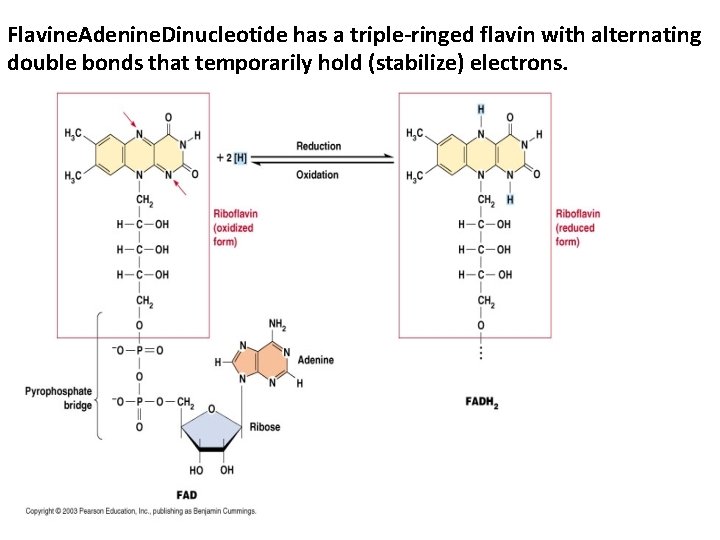
Flavine. Adenine. Dinucleotide has a triple-ringed flavin with alternating double bonds that temporarily hold (stabilize) electrons.
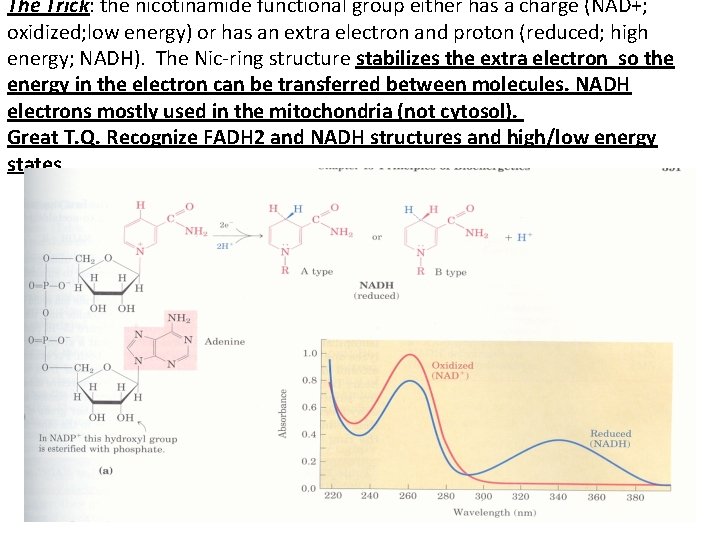
The Trick: the nicotinamide functional group either has a charge (NAD+; oxidized; low energy) or has an extra electron and proton (reduced; high energy; NADH). The Nic-ring structure stabilizes the extra electron so the energy in the electron can be transferred between molecules. NADH electrons mostly used in the mitochondria (not cytosol). Great T. Q. Recognize FADH 2 and NADH structures and high/low energy states
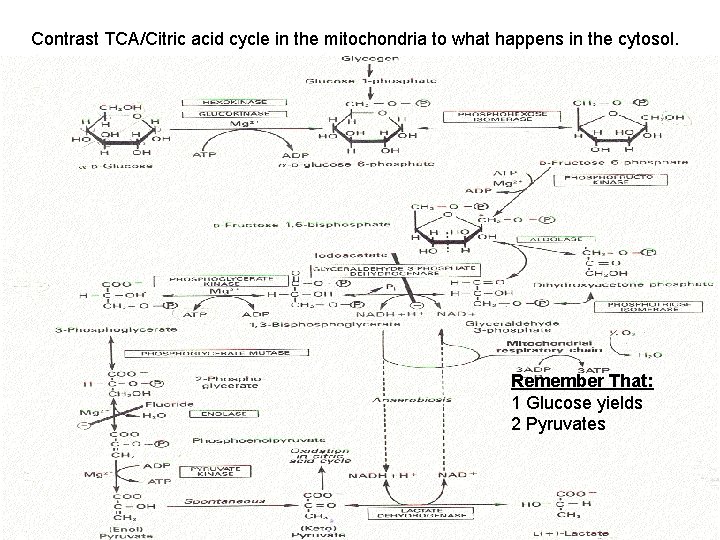
Contrast TCA/Citric acid cycle in the mitochondria to what happens in the cytosol. Remember That: 1 Glucose yields 2 Pyruvates
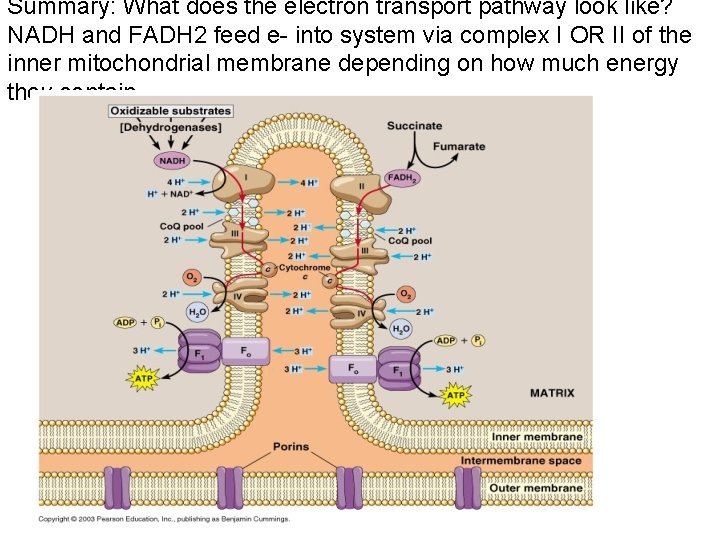
Summary: What does the electron transport pathway look like? NADH and FADH 2 feed e- into system via complex I OR II of the inner mitochondrial membrane depending on how much energy they contain.

The Electron Transport System describes how cells use electron energy to create a proton gradient used for ATP synthesis! • THE FOUR DISCRETE ENZYME COMPLEXES • NADH dehydrogenase: Complex I- 23 peptides – Accepts e- from highest energy NADH and pumps proton out! • Succinate dehydrogenase: Complex II- 4 -5 peptides – Accepts e- from lower energy FADH 2 • Complex I and II Feed e- into Co. Enzyme Q (“Ubiquinone”) • Co-E Q feeds e- into Cytochrome bc: Complex III 9 peptides – Pumps proton out and feeds e- into Complex IV! • Cytochrome Oxidase: Complex IV- 9 more
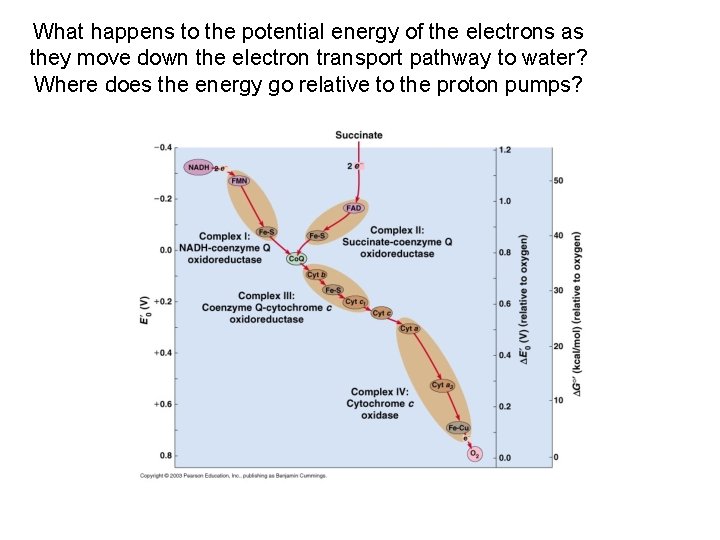
What happens to the potential energy of the electrons as they move down the electron transport pathway to water? Where does the energy go relative to the proton pumps?
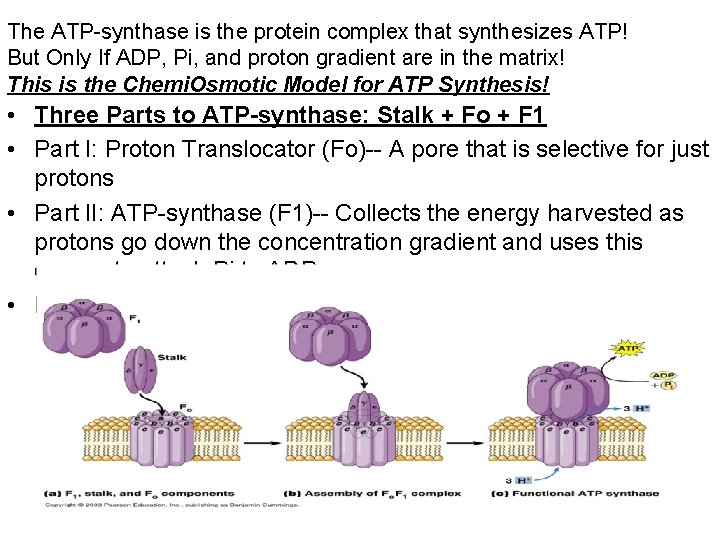
The ATP-synthase is the protein complex that synthesizes ATP! But Only If ADP, Pi, and proton gradient are in the matrix! This is the Chemi. Osmotic Model for ATP Synthesis! • Three Parts to ATP-synthase: Stalk + Fo + F 1 • Part I: Proton Translocator (Fo)-- A pore that is selective for just protons • Part II: ATP-synthase (F 1)-- Collects the energy harvested as protons go down the concentration gradient and uses this energy to attach Pi to ADP. • Part III: Stalk -- Links the F 1 and Fo parts together
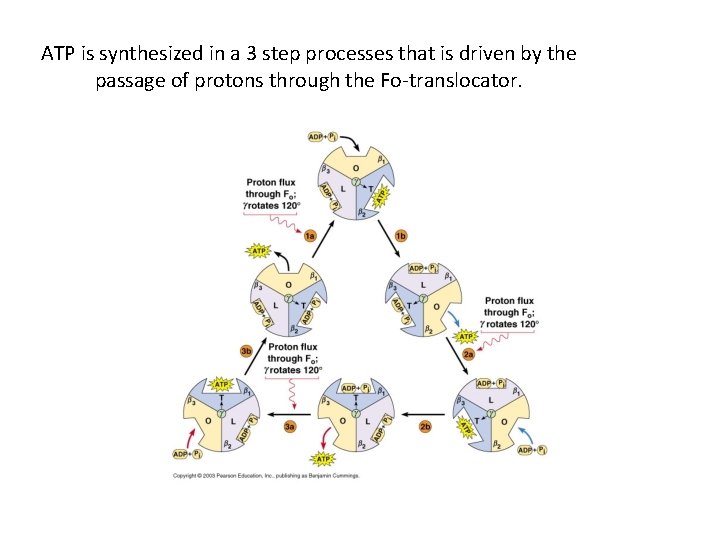
ATP is synthesized in a 3 step processes that is driven by the passage of protons through the Fo-translocator.

What are some metabolic toxins and inhibitors that are of major biological significance to eukaryotes? • Rotenone: Complex I/Co. Q • Cyanide and Carbon Monoxide: Complex IV • Oligomycin antibiotic: ADP phosphorylation • Alcohol and membrane permeability to H+ • Brown fat cells: uncoupler proteins and heat generation • Why might some antibiotics given for bacterial infections affect mitochondrial function in human cells?
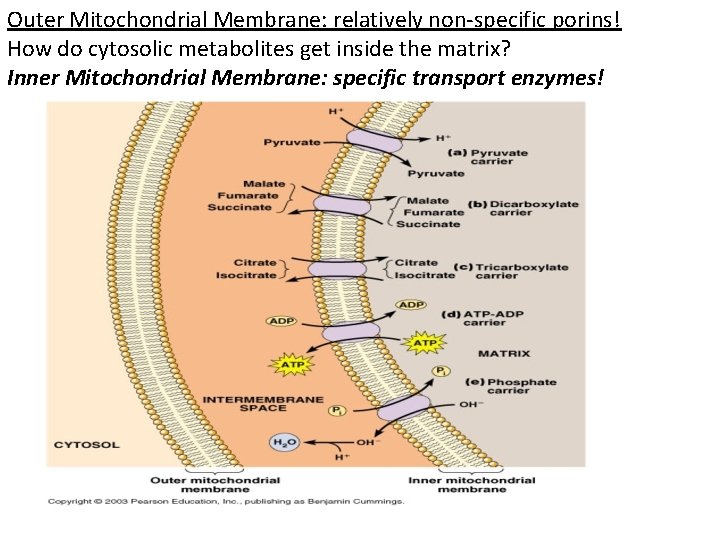
Outer Mitochondrial Membrane: relatively non-specific porins! How do cytosolic metabolites get inside the matrix? Inner Mitochondrial Membrane: specific transport enzymes!
- Slides: 27