Mitglied der HelmholtzGemeinschaft Routen der CO 2 Abscheidung

Mitglied der Helmholtz-Gemeinschaft Routen der CO 2 -Abscheidung in Kraftwerken E. Riensche, J. Nazarko, S. Schiebahn, M. Weber, L. Zhao, D. Stolten Forschungszentrum Jülich Gmb. H, D-52425 Jülich Institut für Energie- und Klimaforschung – IEK-3: Brennstoffzellen Jahreshaupttagung der DPG - Arbeitskreis Energie (AKE) Dresden, 13. -16. März 2011

Introduction Three phases of power production from coal occur: • 20 th century: Continually increasing efficiency up to ……………. . …. … ~45 % • Ending with: Flue gas cleaning (De. NOx, Dedust, De. SOx) ………. . . … 1 -2 %-points loss • 21 th century: Necessity for CCS (Carbon Capture and Storage) … ~8 -14 %-points loss Challenge of CCS: • Collecting CO 2 as pure as possible • High efficiency of power production Current efficiency penalties of 12 -14 %-points • CO 2 separation degrees ~90% and • CO 2 purities between ~90 and 99 mol% R&D: • Gas separation • Integrated CCS systems CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 2
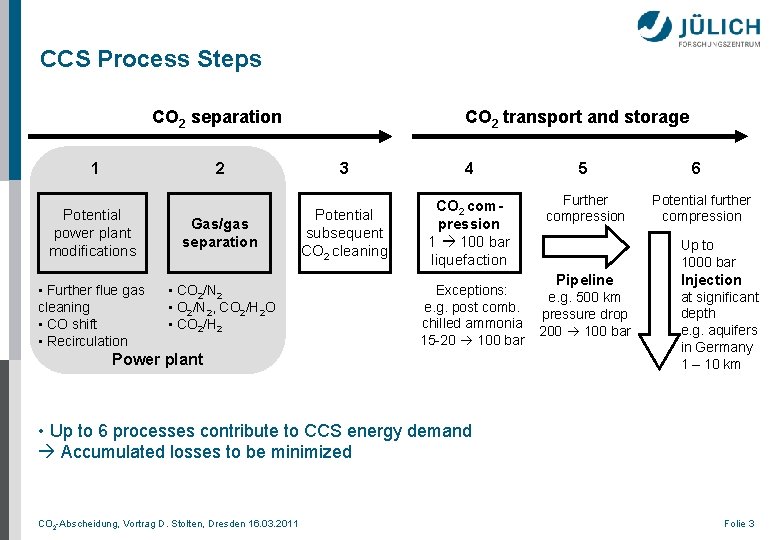
CCS Process Steps CO 2 separation 1 2 Potential power plant modifications Gas/gas separation • Further flue gas cleaning • CO shift • Recirculation • CO 2/N 2 • O 2/N 2, CO 2/H 2 O • CO 2/H 2 CO 2 transport and storage 3 4 5 Potential subsequent CO 2 cleaning CO 2 compression 1 100 bar liquefaction Further compression Exceptions: e. g. post comb. chilled ammonia 15 -20 100 bar Power plant 6 Potential further compression Up to 1000 bar Pipeline e. g. 500 km pressure drop 200 100 bar Injection at significant depth e. g. aquifers in Germany 1 – 10 km • Up to 6 processes contribute to CCS energy demand Accumulated losses to be minimized CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 3
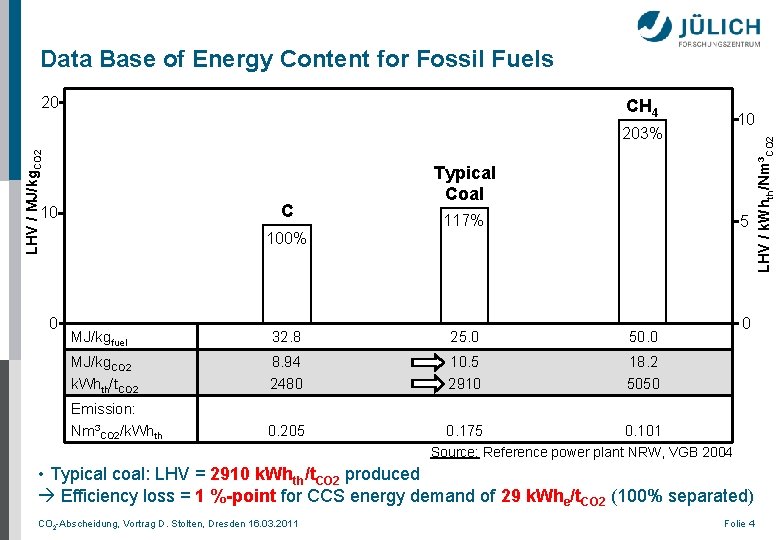
Data Base of Energy Content for Fossil Fuels CH 4 10 LHV / MJ/kg. CO 2 203% C 10 Typical Coal 117% 5 100% 0 MJ/kgfuel 32. 8 25. 0 50. 0 MJ/kg. CO 2 k. Whth/t. CO 2 8. 94 2480 10. 5 2910 18. 2 5050 Emission: Nm³CO 2/k. Whth 0. 205 0. 175 0. 101 0 Source: Reference power plant NRW, VGB 2004 • Typical coal: LHV = 2910 k. Whth/t. CO 2 produced Efficiency loss = 1 %-point for CCS energy demand of 29 k. Whe/t. CO 2 (100% separated) CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 4 LHV / k. Whth/Nm³CO 2 20

Separation Routes, Tasks and Methods CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 5
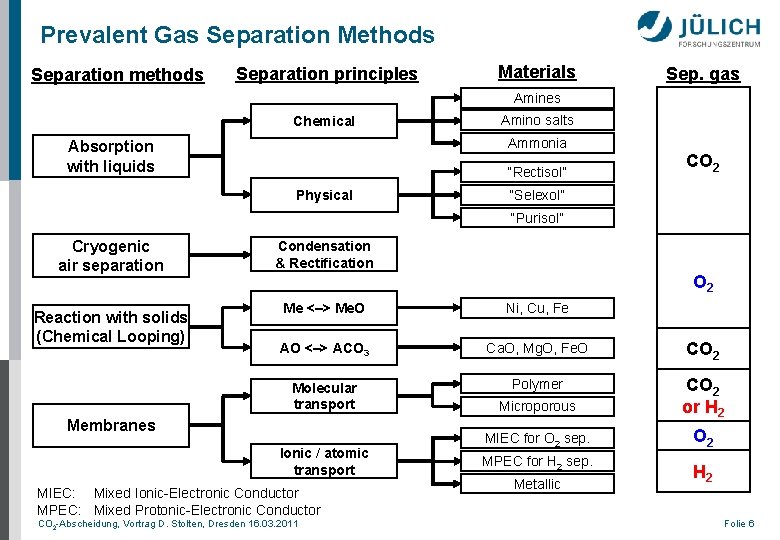
Prevalent Gas Separation Methods Separation methods Separation principles Materials Sep. gas Amines Chemical Amino salts Ammonia Absorption with liquids “Rectisol” Physical CO 2 “Selexol” “Purisol” Cryogenic air separation Reaction with solids (Chemical Looping) Condensation & Rectification O 2 Me <–> Me. O Ni, Cu, Fe AO <–> ACO 3 Ca. O, Mg. O, Fe. O CO 2 Molecular transport Polymer Microporous CO 2 or H 2 MIEC for O 2 sep. O 2 Membranes Ionic / atomic transport MIEC: Mixed Ionic-Electronic Conductor MPEC: Mixed Protonic-Electronic Conductor CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 MPEC for H 2 sep. Metallic H 2 Folie 6
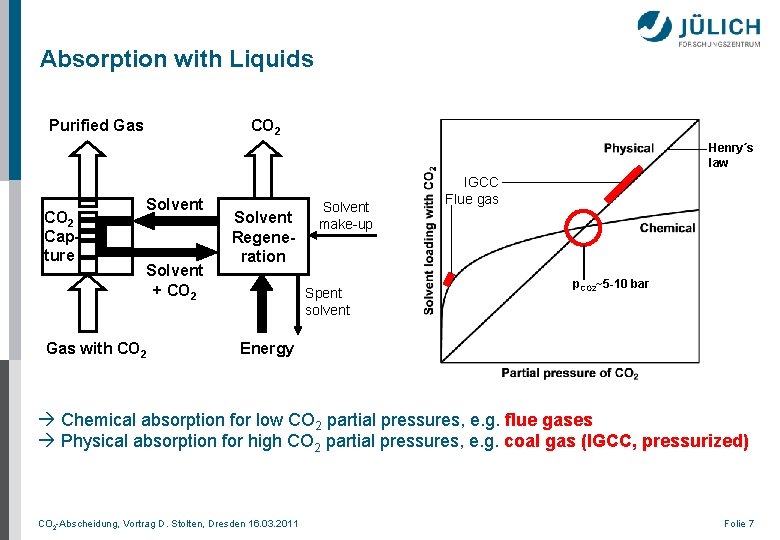
Absorption with Liquids Purified Gas CO 2 Henry´s law CO 2 Capture Solvent + CO 2 Gas with CO 2 Solvent Regeneration Solvent make-up Spent solvent IGCC Flue gas p. CO 2~5 -10 bar Energy Chemical absorption for low CO 2 partial pressures, e. g. flue gases Physical absorption for high CO 2 partial pressures, e. g. coal gas (IGCC, pressurized) CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 7
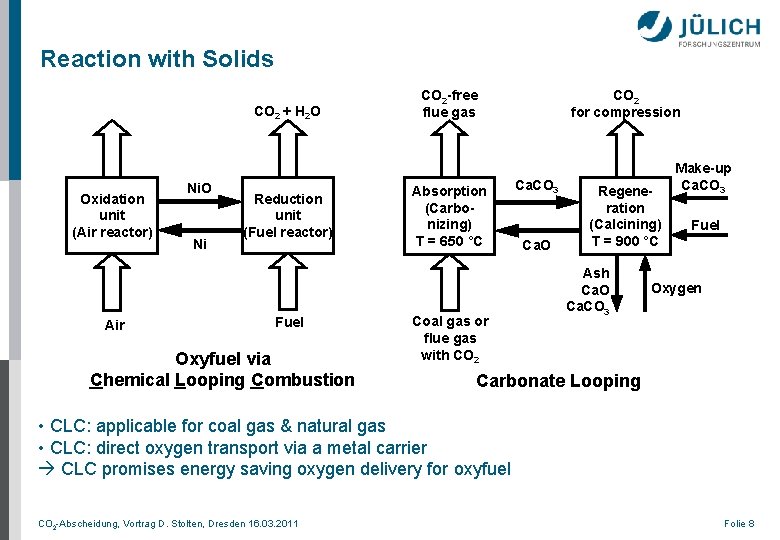
Reaction with Solids CO 2 + H 2 O Oxidation unit (Air reactor) Air Ni. O Ni Reduction unit (Fuel reactor) Fuel Oxyfuel via Chemical Looping Combustion CO 2 -free flue gas Absorption (Carbonizing) T = 650 °C Coal gas or flue gas with CO 2 for compression Ca. CO 3 Ca. O Regeneration (Calcining) T = 900 °C Ash Ca. O Ca. CO 3 Make-up Ca. CO 3 Fuel Oxygen Carbonate Looping • CLC: applicable for coal gas & natural gas • CLC: direct oxygen transport via a metal carrier CLC promises energy saving oxygen delivery for oxyfuel CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 8

Polymer Membranes: CO 2 Separation from Natural Gas Example for a natural gas field p ~100 bar Natural gas p. CO 2 ~10 bar p. CO 2 ~1 bar p ~1 bar CO 2 • Transport: solution diffusion mechanism • Driving force: partial pressure difference • Compressors: not required in natural gas fields • Integration in coal power plants: - Limitation in operating temperature - Compression energy to be considered CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 9
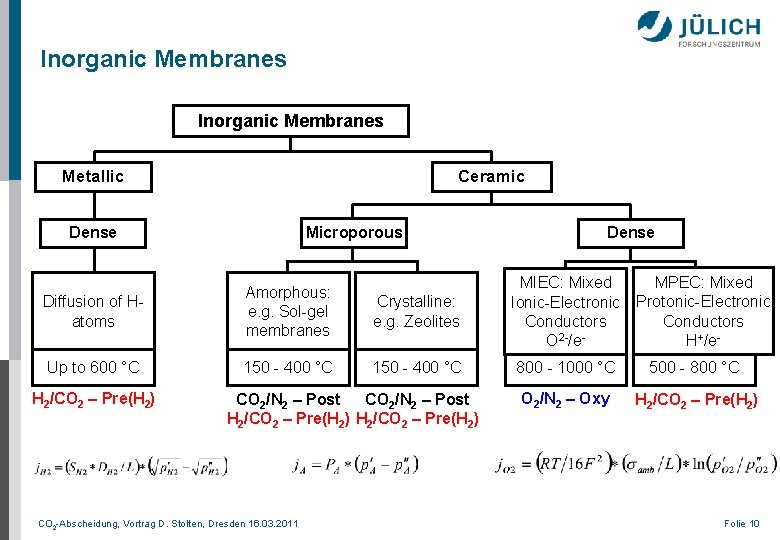
Inorganic Membranes Metallic Ceramic Microporous Dense MPEC: Mixed Protonic-Electronic Conductors H+/e- Diffusion of Hatoms Amorphous: e. g. Sol-gel membranes Crystalline: e. g. Zeolites MIEC: Mixed Ionic-Electronic Conductors O 2 -/e- Up to 600 °C 150 - 400 °C 800 - 1000 °C 500 - 800 °C O 2/N 2 – Oxy H 2/CO 2 – Pre(H 2) CO 2/N 2 – Post H 2/CO 2 – Pre(H 2) CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 10

Résumé: CCS Power Plant Classes Basic concept Route Gas separation task Post CO 2 Oxy O 2 Gas separation method Cond. Absorption / Reaction Membrane separation Cryog. Absorption w. liquids Reaction with solids air sepa. Polymer Chemical Physical Adsorption Reaction ration SPP IGCC* SPP IGCC CO 2 Pre H 2 SPP IGCC + Shift Mixed cond. Metallic SPP IGCC* SPP IGCC + Shift SPP IGCC* Porous SPP IGCC + Shift** IGCC + Shift** * Flue gas recycle for higher CO 2 concentration ** Flue gas recycle for membrane sweep with a large O 2 -poor N 2 gas stream • Today: two power plant technologies: Steam Power Plant (SPP) and IGCC • Identified: 32 CCS power plant classes CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 11
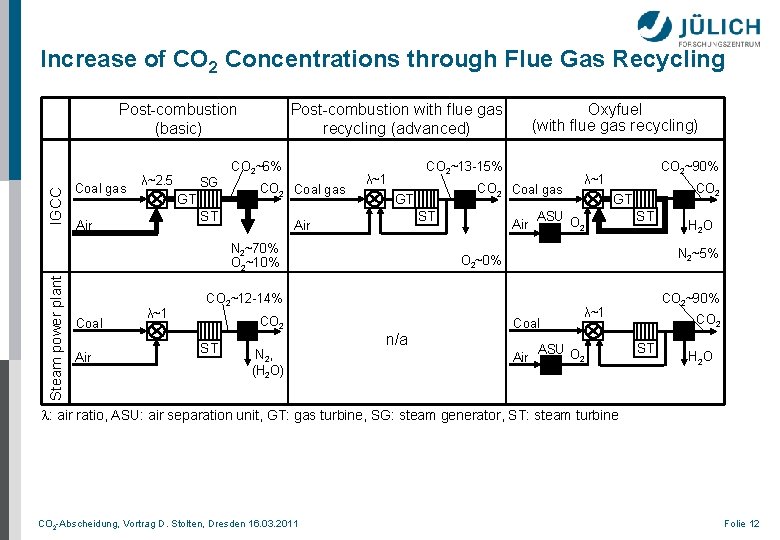
Increase of CO 2 Concentrations through Flue Gas Recycling IGCC Post-combustion (basic) Coal gas λ~2. 5 SG GT Post-combustion with flue gas recycling (advanced) CO 2~6% CO 2 Coal gas ST Air CO 2~13 -15% λ~1 Steam power plant Air λ~1 CO 2 Coal gas GT ST Air N 2~70% O 2~10% Coal Oxyfuel (with flue gas recycling) Air ST N 2, (H 2 O) λ~1 CO 2 GT ASU O 2 ST n/a H 2 O N 2~5% O 2~0% CO 2~12 -14% CO 2~90% Coal Air CO 2~90% λ~1 ASU O 2 CO 2 ST H 2 O l: air ratio, ASU: air separation unit, GT: gas turbine, SG: steam generator, ST: steam turbine CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 12

Post-combustion: Amine Scrubbing CO 2 -free flue gas CO 2+H 2 O 40°C 60°C 90°C Absorber Desorber Heat exchanger Flue gas 100°C 55°C Heat supply • Absorption heat is released at low temperature • Desorption requires heat at higher temperature • Heat supplied by steam condensation at the desorber CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 13
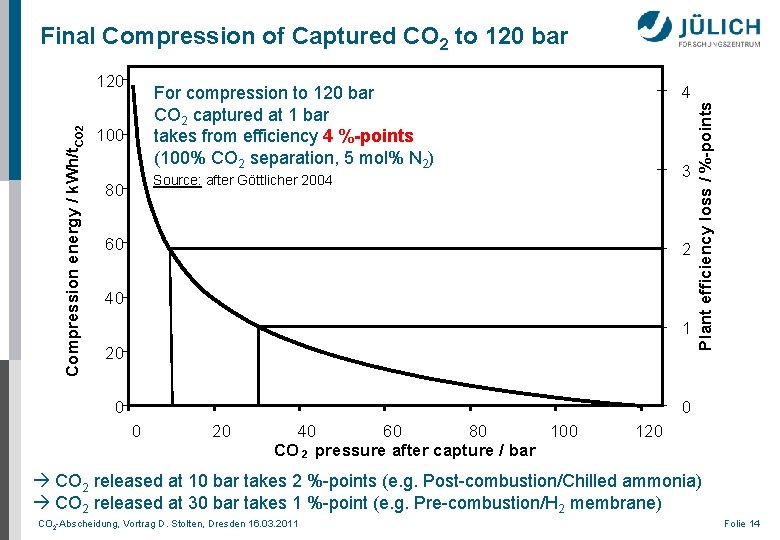
Compression energy / k. Wh/t. CO 2 120 4 For compression to 120 bar CO 2 captured at 1 bar takes from efficiency 4 %-points (100% CO 2 separation, 5 mol% N 2) 100 3 Source: after Göttlicher 2004 80 60 2 40 1 20 0 Plant efficiency loss / %-points Final Compression of Captured CO 2 to 120 bar 0 0 20 40 60 80 100 CO 2 pressure after capture / bar 120 CO 2 released at 10 bar takes 2 %-points (e. g. Post-combustion/Chilled ammonia) CO 2 released at 30 bar takes 1 %-point (e. g. Pre-combustion/H 2 membrane) CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 14

CO 2 Phase Diagram for Pure CO 2 and CO 2 -N 2 Mixtures Pipeline Source: Goos, Riedel, Zhao, Blum, GHGT-10, Amsterdam 2010 • Pure CO 2: Two-phase behaviour only at the saturation line • Impure CO 2: Two-phase regions occur - exceeding 100 bar Work hypothesis for pipeline transport: 5 mol% N 2 tolerable CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 15

Conclusions CCS concepts encompass a broad variety of solutions • Post-combustion, Oxyfuel, Pre-combustion • Gas separation: Absorption, Adsorption, Reaction with solids, Rectification, Membranes. All concepts show potentials for further improvement • Materials´ and componenent development • Integration of components and “CCS waste heat” (from capture and compression). The minimum efficiency penalty for CCS is estimated to be • 4 %-points for CO 2 capture from flue gas (90% separation) and • Even potentially lower, if separation of pure gases is avoided, e. g. by - Membrane sweep (permeation - dilution) and - Chemical looping (e. g. reaction of O 2 with a metal carrier – directly in air). Successful development of CCS concepts will require in-depth dialogue between process engineers and material scientists. CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 16

Thank You for Your Attention! June 20 -22 -2011, Frankfurt am Main Efficient Carbon Capture for Coal Power Plants www. icepe 2011. de CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 17

Thank You for Your Attention! 2 nd International Conference on Process Engineering Efficient Carbon Capture for Coal Power Plants June 20 -22, 2011 Frankfurt am Main/ Germany Registration: www. icepe 2011. de CO 2 -Abscheidung, Vortrag D. Stolten, Dresden 16. 03. 2011 Folie 18
- Slides: 18