Minor1 Solutions P M V Subbarao Professor Mechanical

Minor-1 Solutions P M V Subbarao Professor Mechanical Engineering Department

Problem 1: • Few hundred years ago, huge New. Comen engines mentioned below served the world community as steam pumps. • Horsepower: 88 k. W @ 95 rpm Diameter of Cylinder (Bore) : 584 mm • Height of the Cylinder (Stroke): 1219 mm Draw a simple and neat sketch of this engine, and p-v diagram followed by this engine and calculate the torque and cycle work generated by this engine. • P-v diagram can be approximated as a rectangle. • Change in volume may taken as a fraction of cylinder volume (100% is also valid). • Torque • Cycle work

• Cylinder volume = • Expected from outstanding students, but not used for marking : • For a rectangular p-v diagram: • Engineering requirement: • Piston weight =

Problem 2 • A power plant with a capacity of 150 MW is following a Rankine Cycle. The maximum temperature and pressure of steam are 10 MPa and 500 C and the condenser is maintained at a pressure of 10 KPa. • The reheating is carried out at a pressure of 4 MPa. Reheated steam temperature is 450 C and regeneration is carried out using an Open Feed Water Heater at a pressure of 3 MPa. • Draw the layout of this cycle and locate important points where the properties are to be calculated on this cycle. • Calculate the efficiency of cycle, rate of heat addition required in main steam generator and reheater. Also calculate the mass flow rate of steam required to generate a power of 150 MW
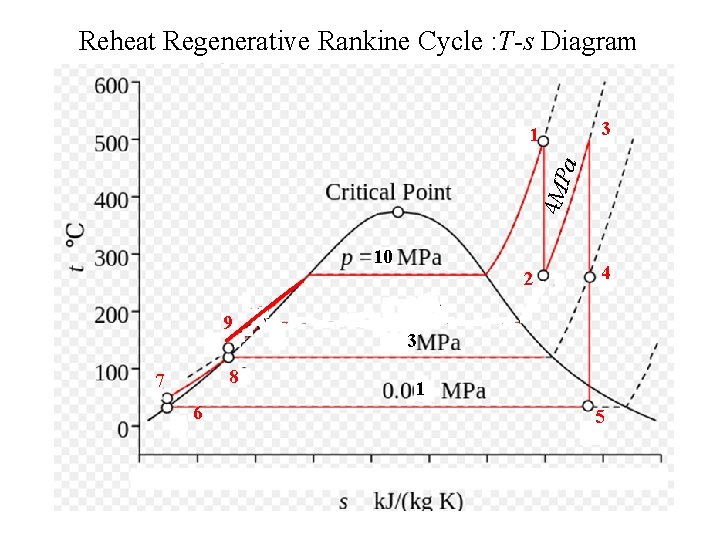
Reheat Regenerative Rankine Cycle : T-s Diagram 3 4 MP a 1 10 2 9 8 7 6 4 3 1 5
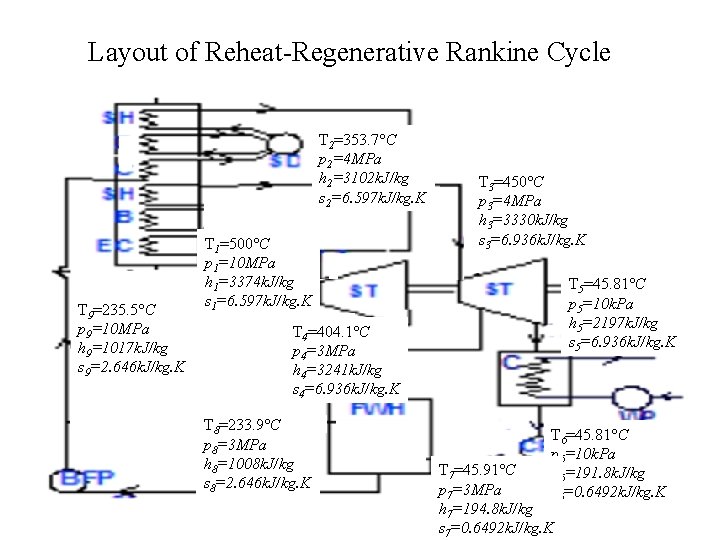
Layout of Reheat-Regenerative Rankine Cycle T 2=353. 7 C p 2=4 MPa h 2=3102 k. J/kg s 2=6. 597 k. J/kg. K T 9=235. 5 C p 9=10 MPa h 9=1017 k. J/kg s 9=2. 646 k. J/kg. K T 1=500 C p 1=10 MPa h 1=3374 k. J/kg s 1=6. 597 k. J/kg. K T 3=450 C p 3=4 MPa h 3=3330 k. J/kg s 3=6. 936 k. J/kg. K T 5=45. 81 C p 5=10 k. Pa h 5=2197 k. J/kg s 5=6. 936 k. J/kg. K T 4=404. 1 C p 4=3 MPa h 4=3241 k. J/kg s 4=6. 936 k. J/kg. K T 8=233. 9 C p 8=3 MPa h 8=1008 k. J/kg s 8=2. 646 k. J/kg. K T 6=45. 81 C p 6=10 k. Pa h 6=191. 8 k. J/kg s 6=0. 6492 k. J/kg. K T 7=45. 91 C p 7=3 MPa h 7=194. 8 k. J/kg s 7=0. 6492 k. J/kg. K

Calculation of Specific Turbine Work Output Specific work done in HP turbine Bleed steam fraction Specific work done in LP turbine Specific turbine work output= 272+854. 3=1126. 3 k. J/kg

Calculation of Net specific Work output Specific work consumed by LP (condensate) pump Specific work consumed by HP (Boiler Feed) pump Net specific Work output = 1126. 3 -2. 2 -9 = 1115. 1 k. J/kg

Calculation of Mass Flow Rate of Steam Mass flow rate of Steam =

Capacity of Heaters Capacity of Main Steam generator Capacity of Reheator

Cycle Efficiency 44. 54%

Problem 3 • The boiler of the steam engine discussed in Problem 1 was using coal as a fuel. A test on this engine shows following volumetric Dry Exhaust Gas Analysis. • CO 2: 11. 7%, O 2: 3. 9%, CO: 1. 6%, & N 2: 82. 8%. • A combined Proximate and Ultimate Analysis (As Received Basis) of the coal are given as: Carbon Content (%) 42. 6 Moisture Content (%) 6. 0 Hydrogen Content (%) 6. 3 Oxygen Content (%) 8. 5 Sulphur Content (%) 0. 9 Ash Content (%) 35. 7 Express the ultimate anlaysis on dry-ash free basis. Calculate actual air-fuel ratio used in this boiler furnace.

Combined Ultimate and Proximate Analyses : As Received Basis Carbon Content (%) 42. 6 Moisture Content (%) 6. 0 Hydrogen Content (%) 6. 3 Oxygen Content (%) 8. 5 Sulphur Content (%) 0. 9 Ash Content (%) 35. 7

Ultimate Analysis: Dry-Ash Free Basis Carbon Content (%) 42. 6/(100 -35. 7 -6) 100= 73. 07 Moisture Content (%) 0. 0 Hydrogen Content (%) 6. 3/(100 -35. 7 -6) 100= 10. 81 Oxygen Content (%) 8. 5(100 -35. 7 -6) 100= 14. 58 Sulphur Content (%) 0. 9(100 -35. 7 -6) 100= 1. 54 Ash Content (%) 0. 0

Equivalent Chemical Formula of Dry-ash free Basis • Ultimate Analysis of dry (moisture free) fuel: Gravimetric • Percentage of carbon : 73. 07 --- Number of moles, X=73. 07 /12= 6. 09 • Percentage of hydrogen : 10. 81 --- Number of atomic moles, Y = 10. 81/1=10. 81 • Percentage of oxygen: 14. 58 --- Number of atomic moles, K = 14. 58/16=0. 91 • Percentage of sulfur: 1. 54 – Number of atomic moles, Z = 1. 54/32=0. 05 • Equivalent chemical formula : C 6. 09 H 10. 81 S 0. 05 O 0. 91 • Equivalent Molecular weight : 100 kgs. • Note: You can develop ECF using as received basis too…. .

Stoichiometry of Actual Combustion

Clue from Theory of Proportions : School Knowledge • Volumetric analysis of few selected gases in exhaust gas are given. • We need to develop a stoichiometry equation to generated 100 kmols of these selected gases. • Let us say n kmoles of dry-ash free coal is burned with an excess air of to generate 100 kmoles of selected gas mixture, water vapour and SO 2

Stoichiometry of Actual Combustion with known Dry Exhaust Gas Analysis • Conservation species: • Conservation of Carbon: 6. 09 n = 11. 7+1. 6 n = 2. 184 • Conservation of Nitrogen: e n 31. 396 = 82. 8 = 1. 2076

Recognition of Actual Air Fuel Ratio Air-fuel ratio on dry-moisture free basis: 100 kg of as received coal contains 58. 3 kg of dry-ash free coal. Air-fuel ratio on as-received basis:
- Slides: 19