Minimally Invasive Delivery System for Bone Graft Materials

Minimally Invasive Delivery System for Bone Graft Materials Lauren Burdock (BME) Jon Witten (BME) Frank Zhao (BME)
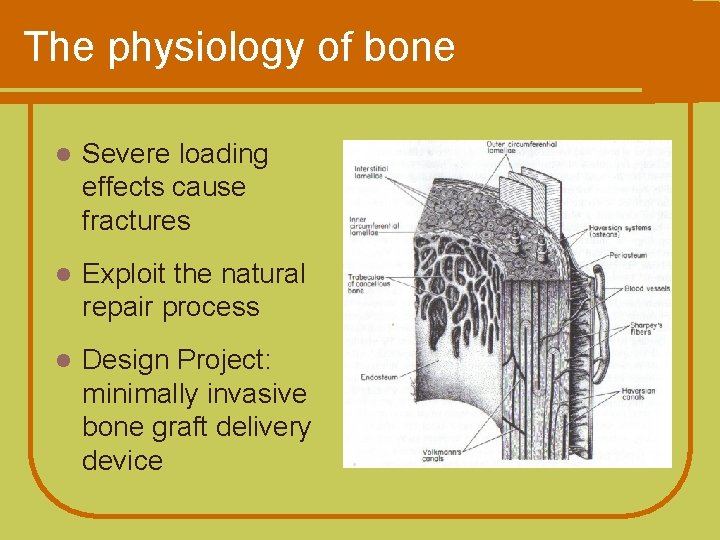
The physiology of bone l Severe loading effects cause fractures l Exploit the natural repair process l Design Project: minimally invasive bone graft delivery device
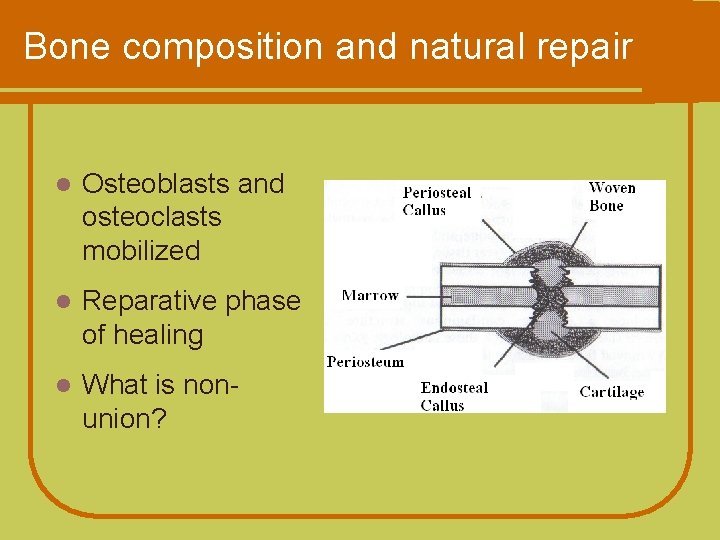
Bone composition and natural repair l Osteoblasts and osteoclasts mobilized l Reparative phase of healing l What is nonunion?
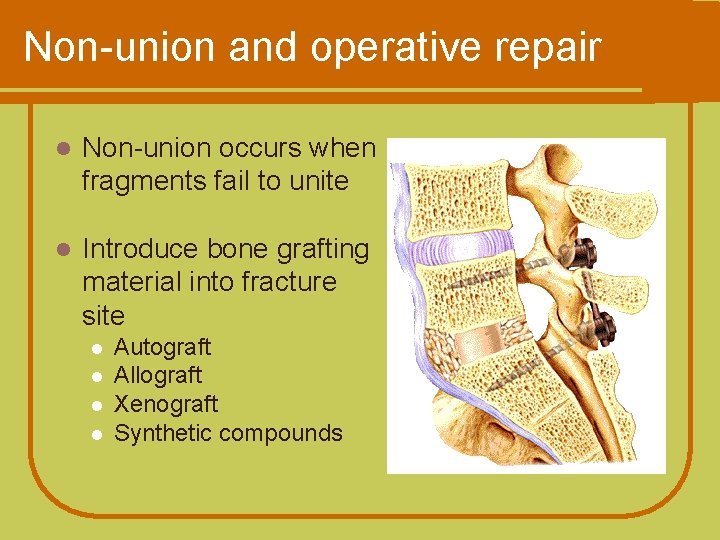
Non-union and operative repair l Non-union occurs when fragments fail to unite l Introduce bone grafting material into fracture site l l Autograft Allograft Xenograft Synthetic compounds

Current Bone Graft Insertion Procedures l Previous bone grafting techniques require incisions and manual application of bone pastes and putties into the fracture site, followed by closing of the surgical site. l Open surgery is more prone to contamination and infection than less invasive procedures.

Goal l We want to reduce morbidity associated with current bone grafting procedures l To do this, a minimally invasive technique would be needed to replace the procedures that are currently in place l This is a proof-of-concept project. We intend to construct a prototype that could then be adopted by the industry
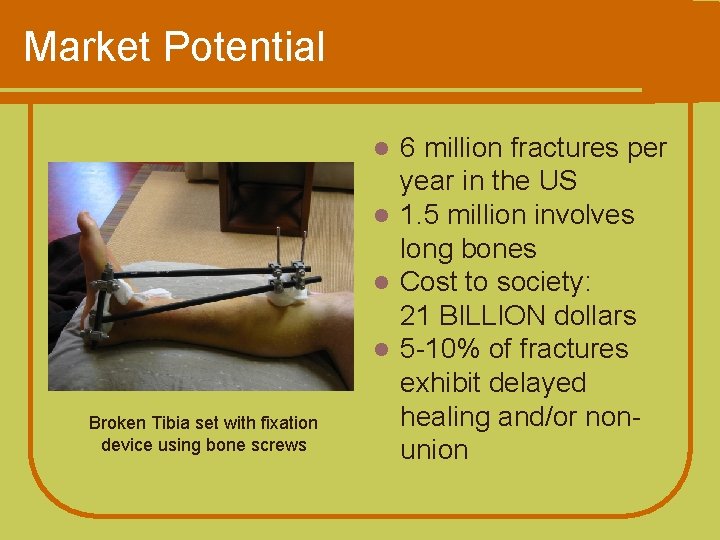
Market Potential 6 million fractures per year in the US l 1. 5 million involves long bones l Cost to society: 21 BILLION dollars l 5 -10% of fractures exhibit delayed healing and/or nonunion l Broken Tibia set with fixation device using bone screws
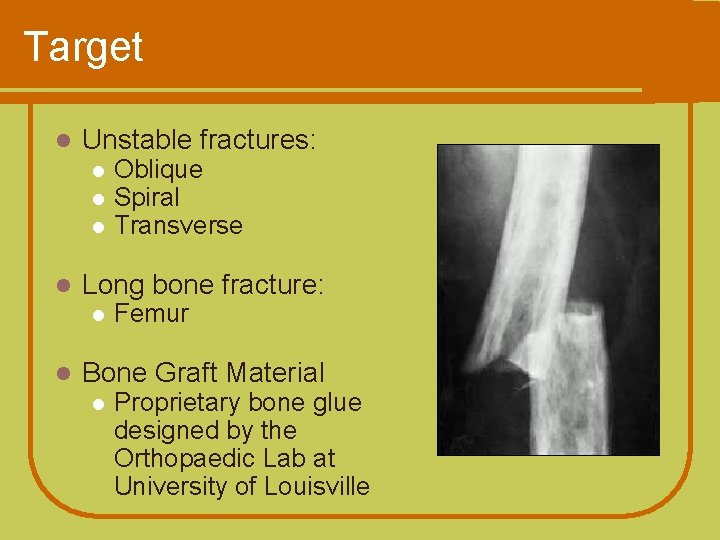
Target l Unstable fractures: l l Long bone fracture: l l Oblique Spiral Transverse Femur Bone Graft Material l Proprietary bone glue designed by the Orthopaedic Lab at University of Louisville

Design Concept External fixation device holds bone sections together to allow proper healing and alignment

Design Concept l Hollow needle design l Diameter of lumen must allow passage of bone graft material l Potentially attached to the external fixation device Pin will be inserted into this area to fill the gap with bone graft materials.
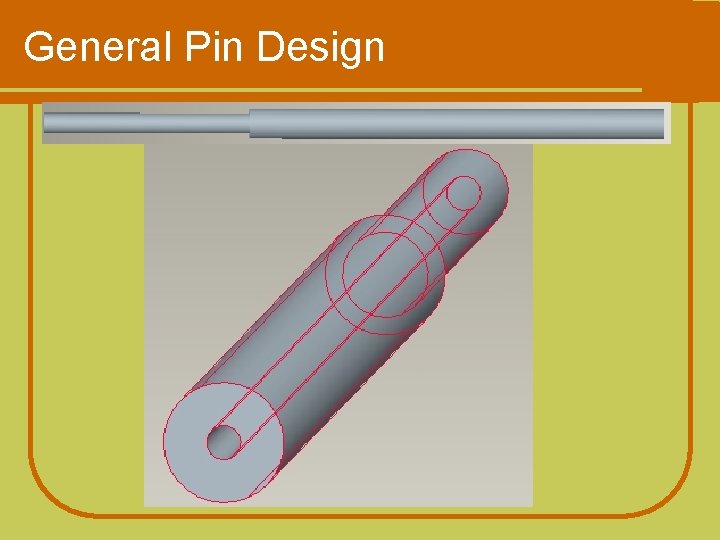
General Pin Design

Types of Output Ports
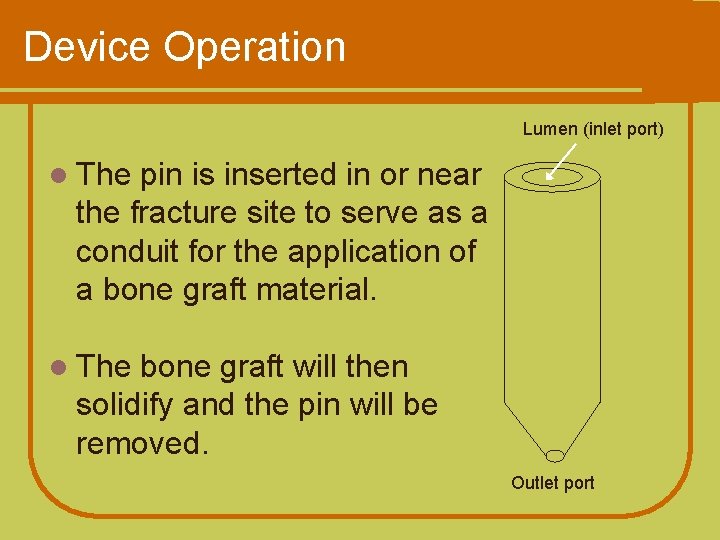
Device Operation Lumen (inlet port) l The pin is inserted in or near the fracture site to serve as a conduit for the application of a bone graft material. l The bone graft will then solidify and the pin will be removed. Outlet port
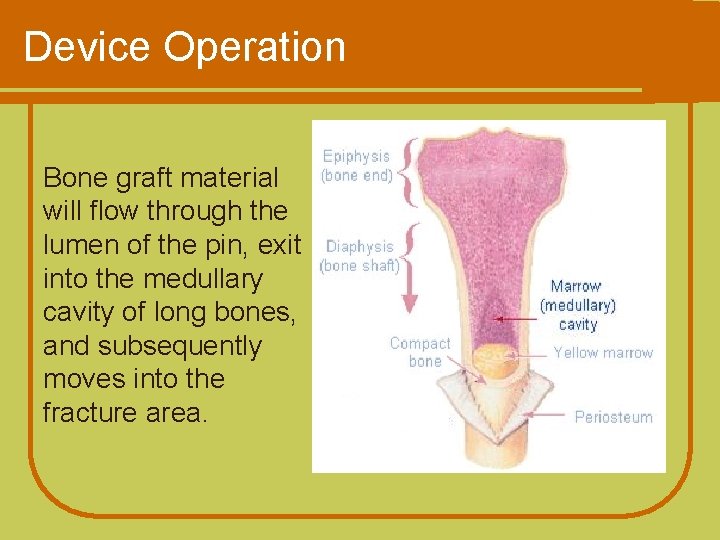
Device Operation Bone graft material will flow through the lumen of the pin, exit into the medullary cavity of long bones, and subsequently moves into the fracture area.

Testing l Phase I: Bone substitution: acrylic tubing l Bone graft material substitution: Plaster of Paris l l Phase II: Porcine/Cadaver femur bones (provided by U of L) l Possibly bone graft material designed by the Orthopaedic Lab of U of L l

FDA Considerations l There has been a recent proposal to reclassify intervertebral body fusion devices from class III to class II. l We are still researching whether this applies to our device. l Class II: material characterization, mechanical testing, animal testing, clinical testing, sterility, biocompatibility, and labeling.

Potential Problems l Contamination at l Viscosity of bone inlet port graft material change diameter of lumen l Clogging of output port experiment with size and position l Accurate delivery to fracture site experiment with pin placement l Pin material must be biocompatible l Pin strength (a problem of lumen versus outer diameter size)

Progress l Work Accomplished l l Used Pro. E to design prototypes Bioengineering Lab at Uof. L received schematics on Friday, February 17 th Submitted a Request for Quote on a forum for machine shops in the region Future Work l l Use a QFD diagram to compare designs Try to obtain bone graft samples for viscosity testing and subsequent calculations.
- Slides: 18