Minimal Residual Disease and Biomarkers ASH 2015 Summary

Minimal Residual Disease and Biomarkers ASH 2015 Summary 57 th ASH Annual Meeting and Exposition. Orlando, FL

Mutational Profile And Prognostic Relevance of Circulating Tumor Cells in Multiple Myeloma Y Mishima *, B Paiva *, J Shi *, M Massoud, S Manier, L Flores, A Perilla. Glen, Y Aljawai, S Takagi, D Huynh, AM Roccaro, A Sacco, D Alginani, MV Mateos, J Blade, JJ Lahuerta, P Richardson, J Laubach, R Schlossman, K Anderson, N Munshi, F Prosper, JF San Miguel *, F Michor *, IM Ghobrial * 57 th ASH Annual Meeting and Exposition. Orlando, FL
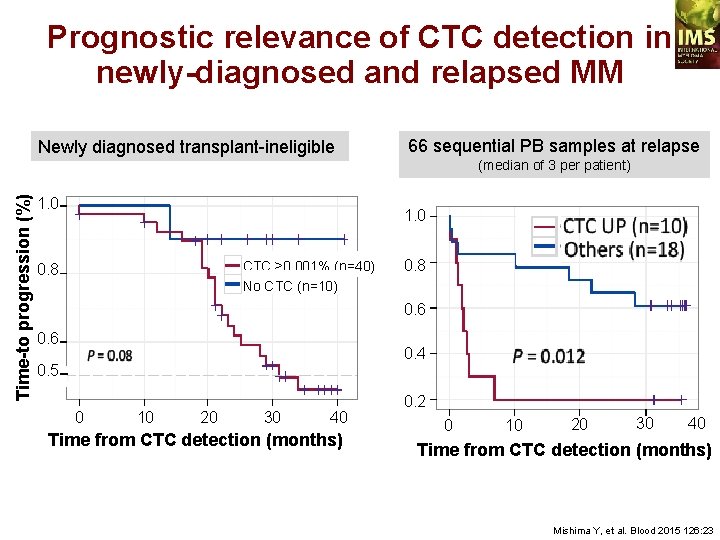
Prognostic relevance of CTC detection in newly-diagnosed and relapsed MM Newly diagnosed transplant-ineligible 66 sequential PB samples at relapse Time-to progression (%) (median of 3 per patient) 1. 0 CTC ≥ 0. 001% (n=40) No CTC (n=10) 0. 8 0. 6 0. 4 0. 5 0 10 20 30 40 Time from CTC detection (months) 0. 2 0 10 20 30 40 Time from CTC detection (months) Mishima Y, et al. Blood 2015 126: 23
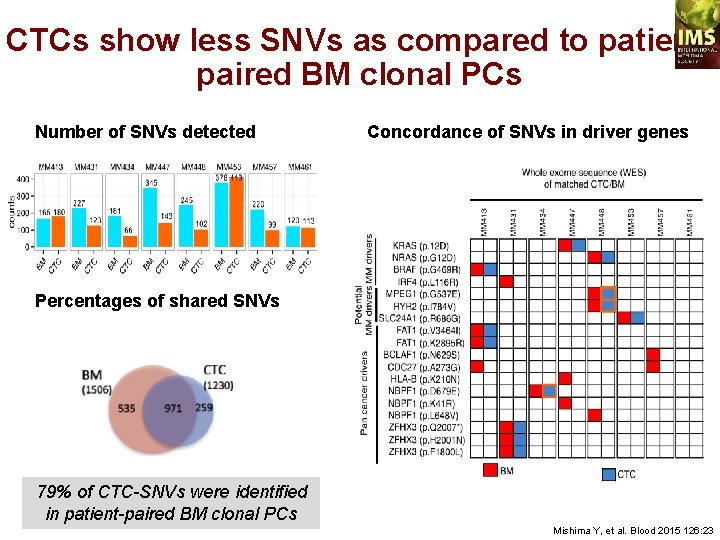
CTCs show less SNVs as compared to patientpaired BM clonal PCs Number of SNVs detected Concordance of SNVs in driver genes Percentages of shared SNVs 79% of CTC-SNVs were identified in patient-paired BM clonal PCs Mishima Y, et al. Blood 2015 126: 23
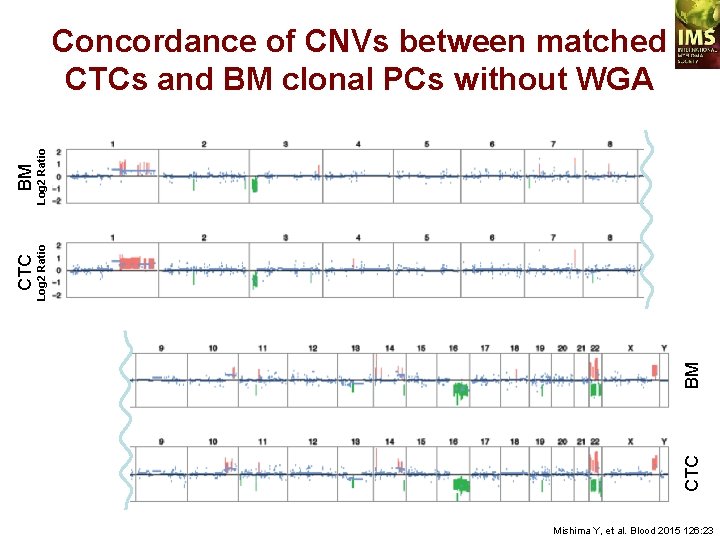
BM CTC BM Log 2 Ratio CTC Log 2 Ratio Concordance of CNVs between matched CTCs and BM clonal PCs without WGA Mishima Y, et al. Blood 2015 126: 23

Conclusions • This study provides the rational for an integrated flowmolecular algorithm to detect CTCs in PB, and to identify candidate patients for noninvasive genomic characterization • Mutational profile of CTCs mirrors a significant fraction (~80%) of the genomic variants present in BM clonal PCs, but may miss up to one-third (35%) of mutations specifically detected in BM clonal PCs • CTCs could potentially be used as a noninvasive biomarker to monitor MM drivers (e. g. : KNRAS, BRAF, IRF 4) Mishima Y, et al. Blood 2015 126: 23

Evaluation of circulating tumour DNA (ct. DNA) for the mutational characterisation of multiple myeloma Andrew Spencer 1, Sridurga Mithraprabhu 1, Malarmathy Ramachandran 1, Daniela Klarica 1, Jay Hocking 1, Laura Mai 2, Stephanie Walsh 2, David Broemeling 2, Andre Marziali 2, Anna Kalff 1, Matthew Wiggin 2, Brian Durie 3, Tiffany Khong 1 1 Alfred Health-Monash University, Melbourne, Australia; 2 Boreal Genomics, Vancouver, Canada; 3 Samuel Oschin Comprehensive Cancer Institute, Los Angeles, USA
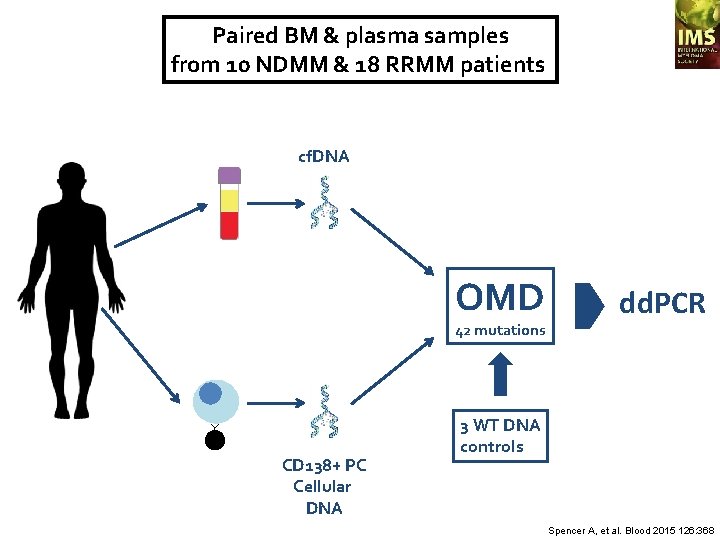
Paired BM & plasma samples from 10 NDMM & 18 RRMM patients cf. DNA OMD 42 mutations CD 138+ PC Cellular DNA dd. PCR 3 WT DNA controls Spencer A, et al. Blood 2015 126: 368
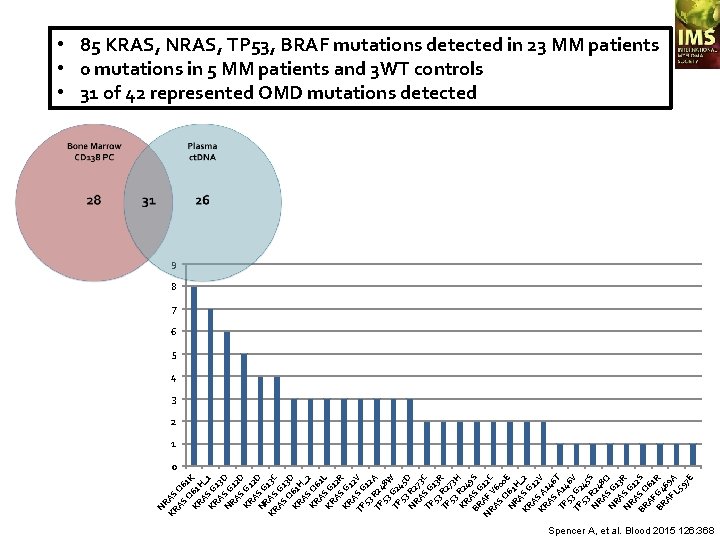
N R KR AS AS Q 6 Q 1 K KR 61 A H_ KR S G 1 A 13 D N SG RA 12 S D KR G 1 AS 2 D N R G KR AS 13 C AS G 1 Q 3 D KR 61 AS H_2 KR Q A 61 L KR S G A 12 R KR S G A 12 TP S G V 53 12 TP R 24 A 53 8 W TP G 2 53 45 D N R 2 RA 73 TP S G C 53 13 R TP R 2 53 73 H KR R 24 A 9 BR S G S N AF 12 C RA V S 600 Q E N 61 RA H _ KR S G 2 AS 12 KR A V AS 146 TP A 1 T 53 46 TP G 2 V 53 45 S N R 24 RA 8 Q N SG RA 13 N SG R RA 1 2 BR S Q S AF 61 BR G 4 R AF 69 L 5 A 97 E • 85 KRAS, NRAS, TP 53, BRAF mutations detected in 23 MM patients • 0 mutations in 5 MM patients and 3 WT controls • 31 of 42 represented OMD mutations detected 9 8 7 6 5 4 3 2 1 0 Spencer A, et al. Blood 2015 126: 368
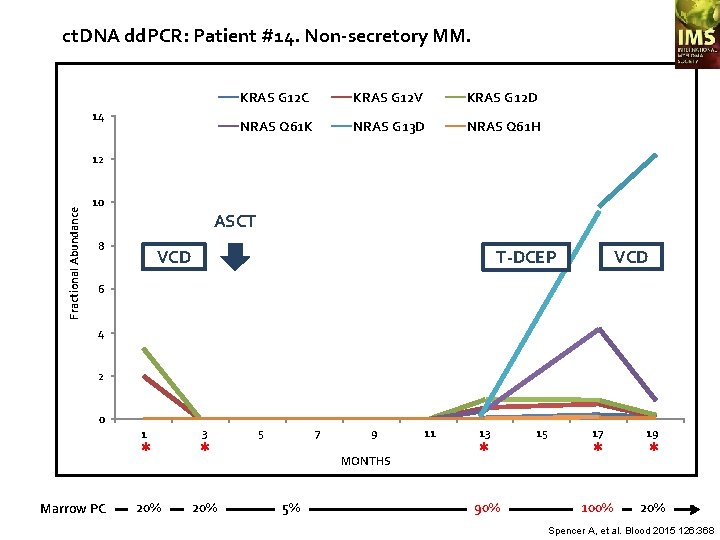
ct. DNA dd. PCR: Patient #14. Non-secretory MM. 14 KRAS G 12 C KRAS G 12 V KRAS G 12 D NRAS Q 61 K NRAS G 13 D NRAS Q 61 H Fractional Abundance 12 10 ASCT 8 T-DCEP VCD 6 4 2 0 1 * Marrow PC 20% 23 * 20% 35 47 59 MONTHS 5% 11 6 13 7 * 90% 15 8 17 9 19 10 100% 20% * * Spencer A, et al. Blood 2015 126: 368

CONCLUSIONS • These data provide proof of concept that evaluable plasmaderived ct. DNA is present in MM and that it can be interrogated to provide additional mutational data on patients. • The sub-clonal architecture in some patients may be more complex than suggested by prior NGS studies with a greater prevalence of RAS mutations than previously described. • Sequential ct. DNA analysis may represent a non-invasive approach for therapeutic monitoring and to inform treatment decisions. Spencer A, et al. Blood 2015 126: 368

What Is the Frequency of Transplant-Eligible Multiple Myeloma Patients Being Cured? The Impact of an MGUS-like Signature at Diagnosis and MRD-Negativity B Paiva, MB Vídriales, N Puig, MT Cedena, L Cordon, MV Mateos, J Martínez. López, L Rosiñol, A García de Coca, N de las Heras, J Galende, J Hernández, L Palomera, J De la Rubia, J Bladé, JF San Miguel, and JJ Lahuerta, on behalf of the Spanish Myeloma Group (GEM/PETHEMA) 57 th ASH Annual Meeting and Exposition. Orlando, FL

PETHEMA/GEM 2000 protocol Diagnosis Induction (n=1075) 6 alternating cycles of VBMCP/VBAD HDT/ASCT Maintenance Interferon/prednisone for 2 y • Patients’ follow-up was updated for the present analysis, and the median follow-up of the series is now of 12 -years • All patients remaining progression-free and alive but for which the follow-up was inferior to 10 -years were excluded from the analysis • From a total of 1071 patients, 1030 were eligible for this analysis because they either relapsed or died during the first 10 -years from diagnosis, or remained progression-free and alive for more than 10 -years • 91 patients (9%) remained progression-free and alive for more than 10 years Paiva B, et al. Blood 2015 126: 725
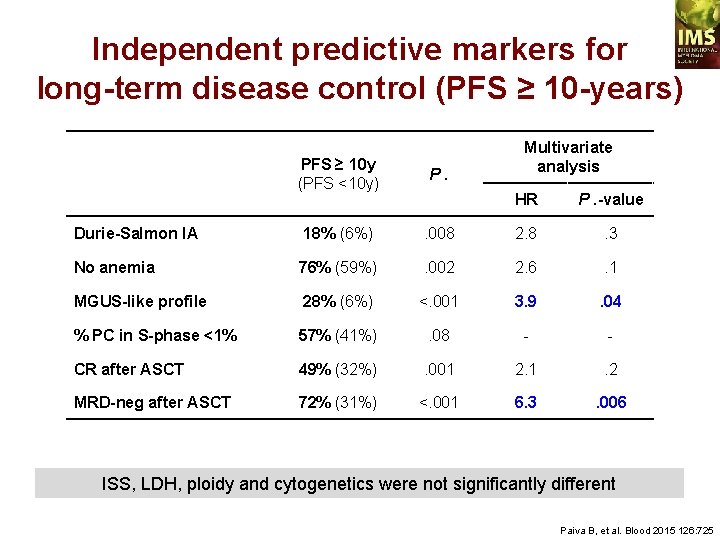
Independent predictive markers for long-term disease control (PFS ≥ 10 -years) PFS ≥ 10 y (PFS <10 y) P. Durie-Salmon IA 18% (6%) No anemia Multivariate analysis HR P. -value . 008 2. 8 . 3 76% (59%) . 002 2. 6 . 1 MGUS-like profile 28% (6%) <. 001 3. 9 . 04 % PC in S-phase <1% 57% (41%) . 08 - - CR after ASCT 49% (32%) . 001 2. 1 . 2 MRD-neg after ASCT 72% (31%) <. 001 6. 3 . 006 ISS, LDH, ploidy and cytogenetics were not significantly different Paiva B, et al. Blood 2015 126: 725
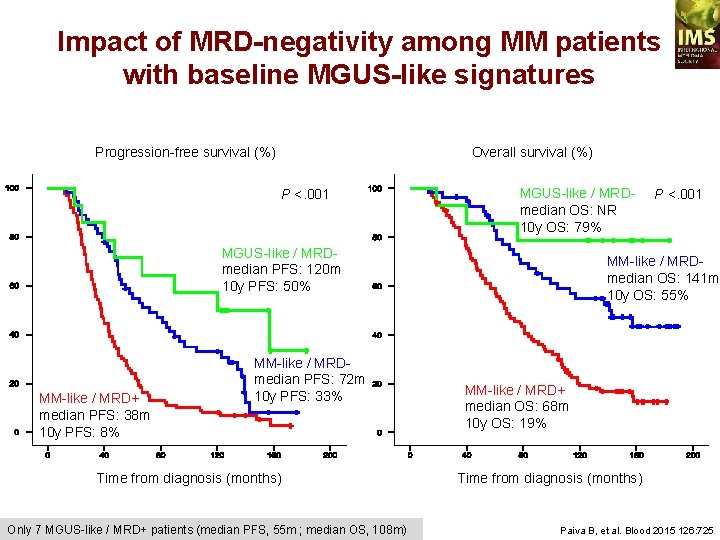
Impact of MRD-negativity among MM patients with baseline MGUS-like signatures Progression-free survival (%) Overall survival (%) P <. 001 MGUS-like / MRDmedian OS: NR 10 y OS: 79% MGUS-like / MRDmedian PFS: 120 m 10 y PFS: 50% MM-like / MRD+ median PFS: 38 m 10 y PFS: 8% MM-like / MRDmedian PFS: 72 m 10 y PFS: 33% Time from diagnosis (months) Only 7 MGUS-like / MRD+ patients (median PFS, 55 m ; median OS, 108 m) P <. 001 MM-like / MRDmedian OS: 141 m 10 y OS: 55% MM-like / MRD+ median OS: 68 m 10 y OS: 19% Time from diagnosis (months) Paiva B, et al. Blood 2015 126: 725

Conclusions • Operational cure (i. e. : >10 -years PFS) was possible for 9% of transplant-eligible MM patients before the era of novel agents. • Curability rates were particularly frequent among patients with a benign phenotypic signature at diagnosis and MRD negativity after HDT/ASCT • Our results suggest a remarkable clinical benefit of attaining deep remissions after intensive treatment for patients with early MM. Paiva B, et al. Blood 2015 126: 725

Prognostic Value of Immune Profiling Multiple Myeloma Patients During Minimal Residual Disease Monitoring in the PETHEMA/GEM 2010 MAS 65 Study P Arana, B Paiva, N Puig, MT Cedena, L Cordon, MB Vidriales, J Martinez-Lopez, EM Ocio, MT Hernandez, AI Teruel, M Gironella, MA Echeveste, L Rosiñol, R Martinez, A Oriol, J Bladé, JJ Lahuerta, MV Mateos, JF San Miguel, on behalf of the Spanish Myeloma Group (GEM/PETHEMA) 57 th ASH Annual Meeting and Exposition. Orlando, FL
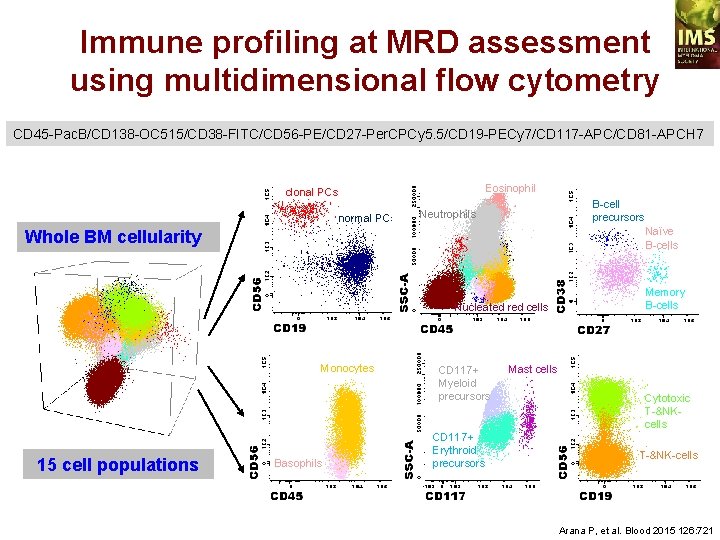
Immune profiling at MRD assessment using multidimensional flow cytometry CD 45 -Pac. B/CD 138 -OC 515/CD 38 -FITC/CD 56 -PE/CD 27 -Per. CPCy 5. 5/CD 19 -PECy 7/CD 117 -APC/CD 81 -APCH 7 Eosinophil s clonal PCs normal PCs Neutrophils Naïve B-cells Whole BM cellularity Nucleated red cells Monocytes 15 cell populations B-cell precursors Basophils CD 117+ Myeloid precursors CD 117+ Erythroid precursors Memory B-cells Mast cells Cytotoxic T-&NKcells T-&NK-cells Arana P, et al. Blood 2015 126: 721
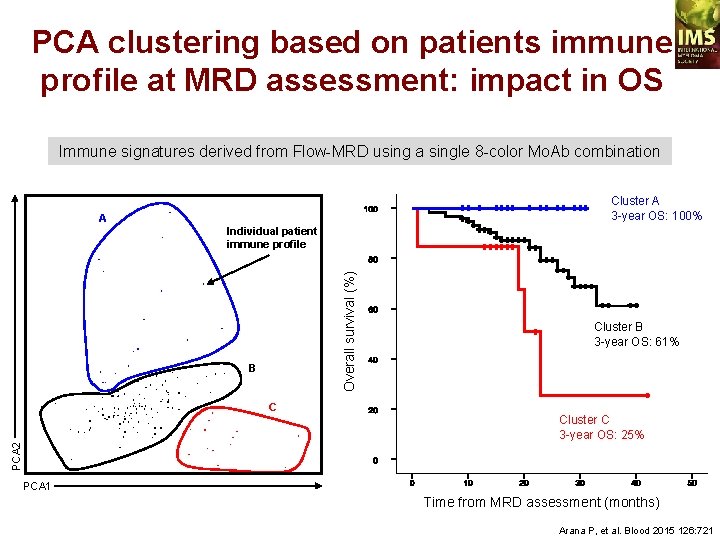
PCA clustering based on patients immune profile at MRD assessment: impact in OS Immune signatures derived from Flow-MRD using a single 8 -color Mo. Ab combination Cluster A 3 -year OS: 100% A Overall survival (%) Individual patient immune profile B Cluster C 3 -year OS: 25% PCA 2 C Cluster B 3 -year OS: 61% PCA 1 Time from MRD assessment (months) Arana P, et al. Blood 2015 126: 721
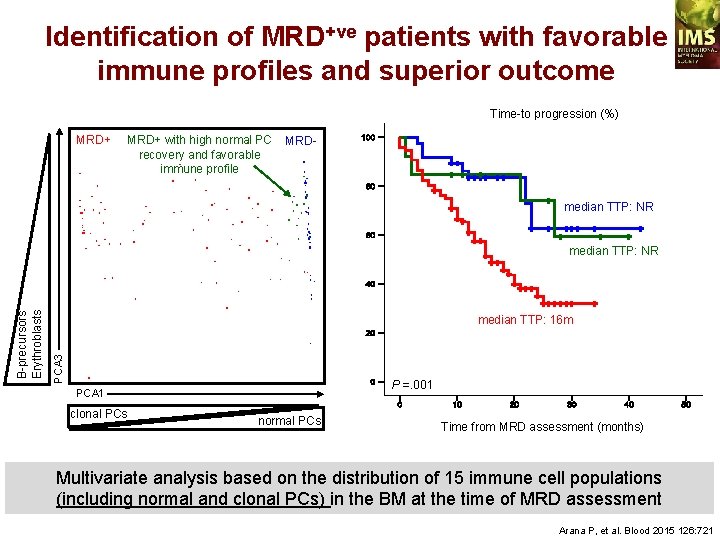
Identification of MRD+ve patients with favorable immune profiles and superior outcome Time-to progression (%) MRD+ with high normal PC MRDrecovery and favorable immune profile median TTP: NR median TTP: 16 m PCA 3 B-precursors Erythroblasts median TTP: NR P =. 001 PCA 1 clonal PCs normal PCs Time from MRD assessment (months) Multivariate analysis based on the distribution of 15 immune cell populations (including normal and clonal PCs) in the BM at the time of MRD assessment Arana P, et al. Blood 2015 126: 721

Conclusions • Immune profiling in MM after therapy during MRD monitoring is prognostically relevant and allows the identification of patients with either poor survival or sustained disease control • Flow-based MRD monitoring offers complementary information to quantification of MRD levels, and may contribute to identify patients that albeit being MRD-positive can still experience prolonged survival due to high normal PC recovery and a unique immune signature particularly characterized by increased B-cell maturation • Flow-based immune profiling at MRD assessment may become particularly relevant for patients treated with immunotherapeutic strategies Arana P, et al. Blood 2015 126: 721

NEXT GENERATION FLOW (NGF) FOR HIGH SENSITIVE DETECTION OF MRD IN MM CANCER RESEARCH CENTER-IBSAL UNIVERSITY & UNIVERSITY HOSPITAL, SALAMANCA (SPAIN) Session 651: “Multiple Myeloma: Biology and physiopathology excluding therapy. Novel technologies to evaluate biology and prognosis” 57 th ASH Annual Meeting Orlando (FL), December 6 th, 2015
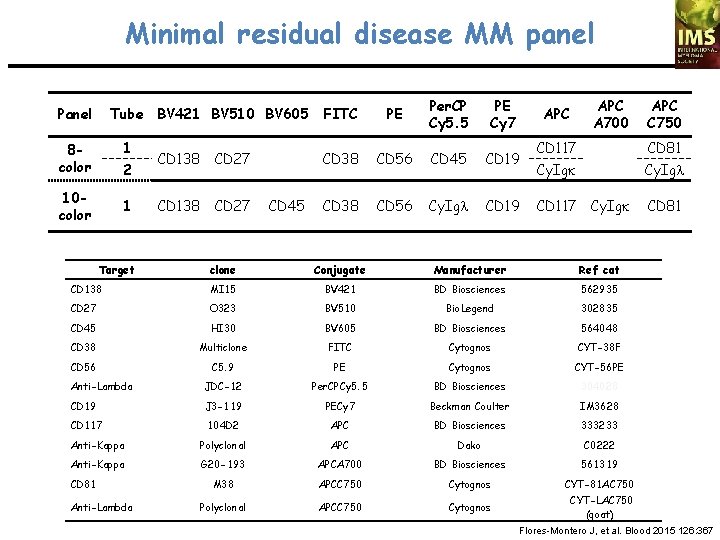
Minimal residual disease MM panel Panel Tube BV 421 BV 510 BV 605 1 8 color 2 10 color 1 Target CD 138 CD 27 CD 45 FITC PE Per. CP Cy 5. 5 PE Cy 7 CD 38 CD 56 CD 45 CD 19 CD 38 CD 56 Cy. Igl CD 19 APC A 700 APC C 750 CD 117 CD 81 Cy. Igk Cy. Igl CD 117 Cy. Igk CD 81 clone Conjugate Manufacturer Ref cat CD 138 MI 15 BV 421 BD Biosciences 562935 CD 27 O 323 BV 510 Bio. Legend 302835 CD 45 HI 30 BV 605 BD Biosciences 564048 CD 38 Multiclone FITC Cytognos CYT-38 F CD 56 C 5. 9 PE Cytognos CYT-56 PE Anti-Lambda JDC-12 Per. CPCy 5. 5 BD Biosciences 304028 CD 19 J 3 -119 PECy 7 Beckman Coulter IM 3628 CD 117 104 D 2 APC BD Biosciences 333233 Anti-Kappa Polyclonal APC Dako C 0222 Anti-Kappa G 20 -193 APCA 700 BD Biosciences 561319 M 38 APCC 750 Cytognos CYT-81 AC 750 Polyclonal APCC 750 Cytognos CYT-LAC 750 (goat) CD 81 Anti-Lambda Flores-Montero J, et al. Blood 2015 126: 367
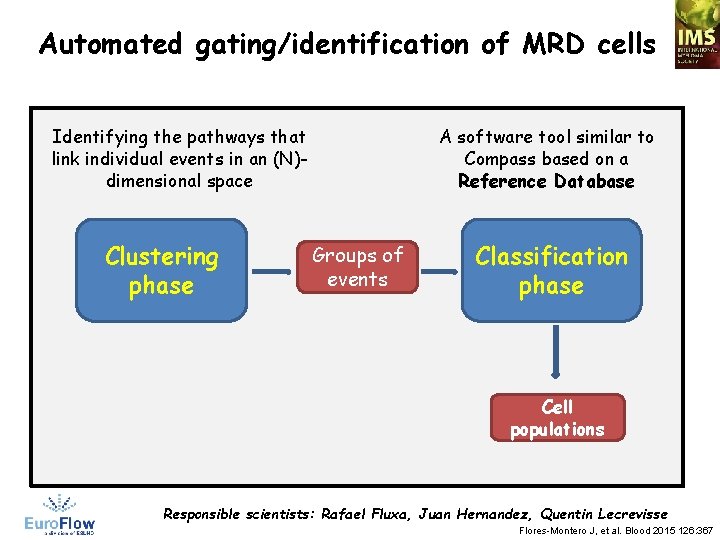
Automated gating/identification of MRD cells Identifying the pathways that link individual events in an (N)dimensional space Clustering phase A software tool similar to Compass based on a Reference Database Groups of events Classification phase Cell populations Responsible scientists: Rafael Fluxa, Juan Hernandez, Quentin Lecrevisse Flores-Montero J, et al. Blood 2015 126: 367
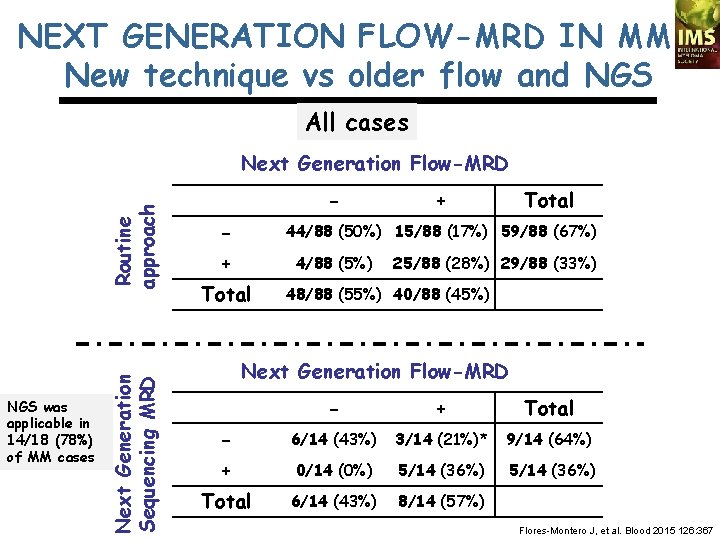
NEXT GENERATION FLOW-MRD IN MM: New technique vs older flow and NGS All cases NGS was applicable in 14/18 (78%) of MM cases Next Generation Sequencing MRD Routine approach Next Generation Flow-MRD - + Total 44/88 (50%) 15/88 (17%) 59/88 (67%) + 4/88 (5%) Total 25/88 (28%) 29/88 (33%) 48/88 (55%) 40/88 (45%) Next Generation Flow-MRD - + Total - 6/14 (43%) 3/14 (21%)* 9/14 (64%) + 0/14 (0%) 5/14 (36%) Total 6/14 (43%) 8/14 (57%) Flores-Montero J, et al. Blood 2015 126: 367

Concluding Remarks Next Generation Flow cytometry (NGF) Advantages Fast (within 3 -4 h) Highly standardized with automated gating (e. g. Infinicyt) Efficient data storage / management with easy data comparison and review. Accurate quantitation Increased sensitivity (10 -5 -10 -6) Information on normal and malignant cells (sample quality and reconstitution of normal compartments) External and internal QC in a per sample basis Ready for IVD development Limitations - Education and training still required - Many cells needed to reach the required sensitivity, e. g. 5. 0 x 106 , if quantitation down to <10 -5 is needed - Fresh (<24 h) samples Adapted from van Dongen JJM, van der Velden VHJ, Bruggemann M & Orfao A, Blood 2015 Flores-Montero J, et al. Blood 2015 126: 367

Evaluation of Minimal Residual Disease (MRD) by Next Generation Sequencing (NGS) is highly predictive of PFS in the IFM/DFCI 2009 trial. Herve Avet-Loiseau, MD, Ph. D 1*, Jill Corre 2*, Valerie Lauwers-Cances, MD 3*, Marie-Lorraine Chretien 4*, Nelly Robillard, 5*, Xavier Leleu, MD, Ph. D 6, Cyrille Hulin, MD 7*, Catherine Gentil, Engineer 8*, Bertrand Arnulf 9*, Karim Belhadj, MD 10*, Sabine Brechignac, MD 11*, Laurent Garderet, MD 12, Lionel Karlin 13*, Gerald Marit 14*, Lotfi Benboubker 15*, Frederique Orsini-Piocelle, MD 16*, Bruno Royer, MD 17, 18*, Bernard Drenou, MD 19*, Mourad Tiab, MD 20*, Thierry Lamy, MD, Ph. D 21, Margaret MACRO, MD 22*, Paul G. Richardson, MD 23, Kenneth C Anderson, MD 24, Malek Faham, MD, Ph. D 25, Thierry Facon 26, Philippe Moreau 27, 28, 29*, Michel Attal 30 and Nikhil C. Munshi, MD 31
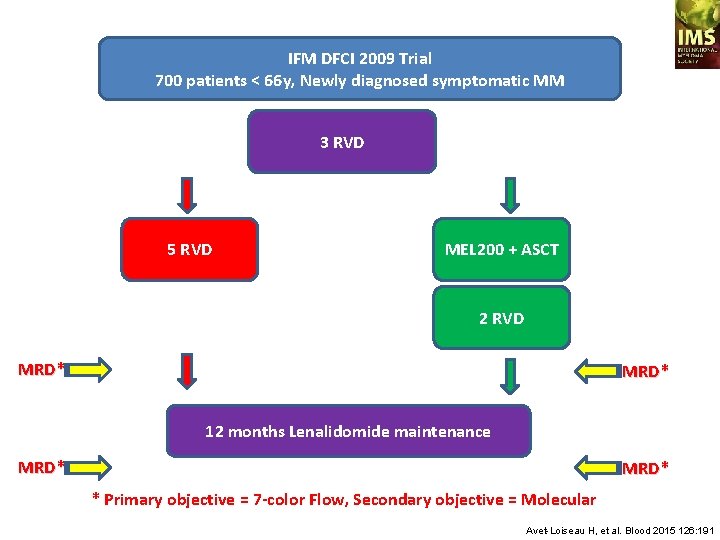
IFM DFCI 2009 Trial 700 patients < 66 y, Newly diagnosed symptomatic MM 3 RVD 5 RVD MEL 200 + ASCT 2 RVD MRD* 12 months Lenalidomide maintenance MRD* * Primary objective = 7 -color Flow, Secondary objective = Molecular Avet-Loiseau H, et al. Blood 2015 126: 191
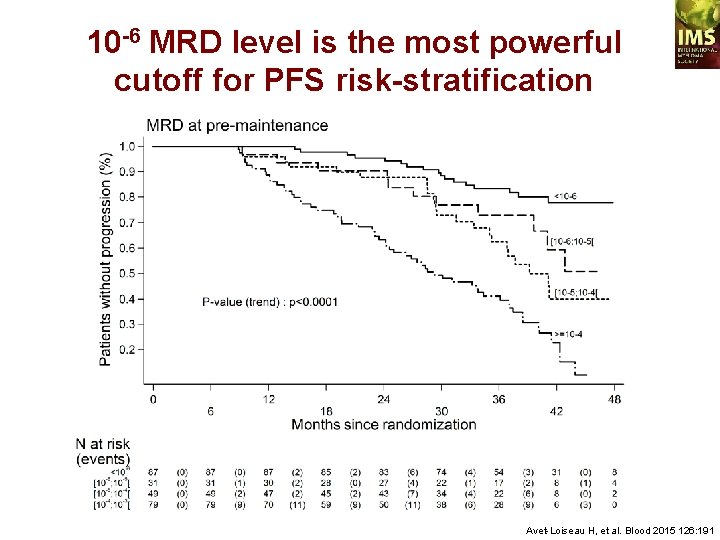
10 -6 MRD level is the most powerful cutoff for PFS risk-stratification Avet-Loiseau H, et al. Blood 2015 126: 191
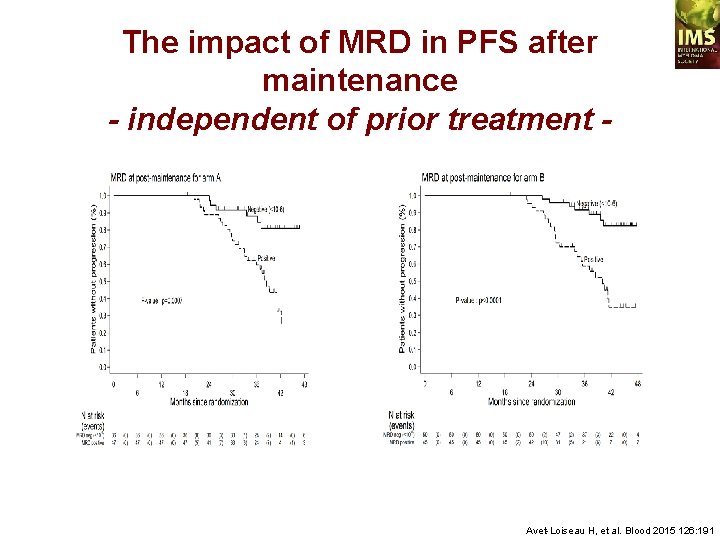
The impact of MRD in PFS after maintenance - independent of prior treatment - Avet-Loiseau H, et al. Blood 2015 126: 191

CONCLUSION MRD NGS is feasible in 92% of the patients MRD NGS is highly sensitive (< 10 -6) This sensitivity is achieved in 100% of the patients 10 -6 level is the most powerful cutoff for PFS 13/26 pts with t(4; 14) achieved MRD negativity vs no with del(17 p) May identify patients cured from myeloma Avet-Loiseau H, et al. Blood 2015 126: 191

Prospective Evaluation of MRI and PET-CT at Diagnosis and before Maintenance Therapy in Symptomatic Patients with Multiple Myeloma Included in the IFM/DFCI 2009 Trial P. Moreau, M. Attal, L. Karlin, L. Garderet, T. Facon, L. Benboubker, M. Macro, D. Caillot, M. Escoffre-Barbe, A. M. Stoppa, K. Laribi, C. Hulin, G. Marit, J. R. Eveillard, F. Caillon, C. Bodet-Millin, J. M. Nguyen, B. Pégourié, V. Dorvaux, C. Chaleteix, K. Anderson, P. Richardson, H. Avet. Loiseau, A. Gaultier, J. M. Nguyen, B. Dupas and F. Bodéré
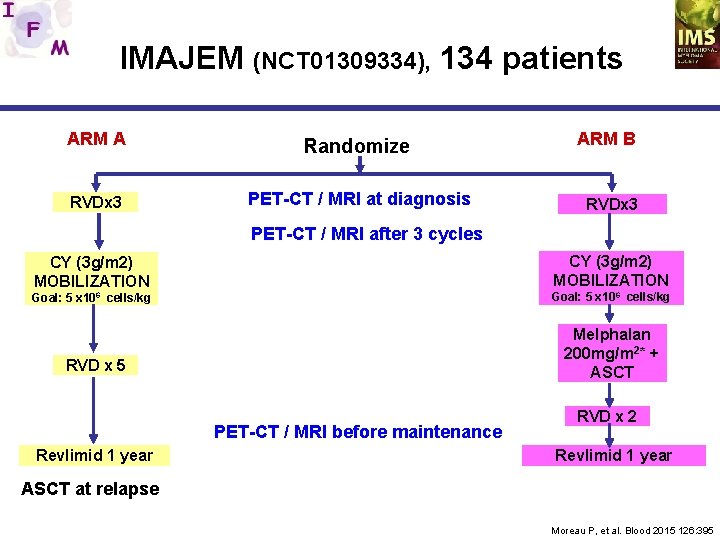
IMAJEM (NCT 01309334), 134 patients ARM A Randomize RVDx 3 PET-CT / MRI at diagnosis ARM B RVDx 3 PET-CT / MRI after 3 cycles CY (3 g/m 2) MOBILIZATION Goal: 5 x 106 cells/kg Melphalan 200 mg/m 2* + ASCT RVD x 5 PET-CT / MRI before maintenance Revlimid 1 year RVD x 2 Revlimid 1 year ASCT at relapse Moreau P, et al. Blood 2015 126: 395
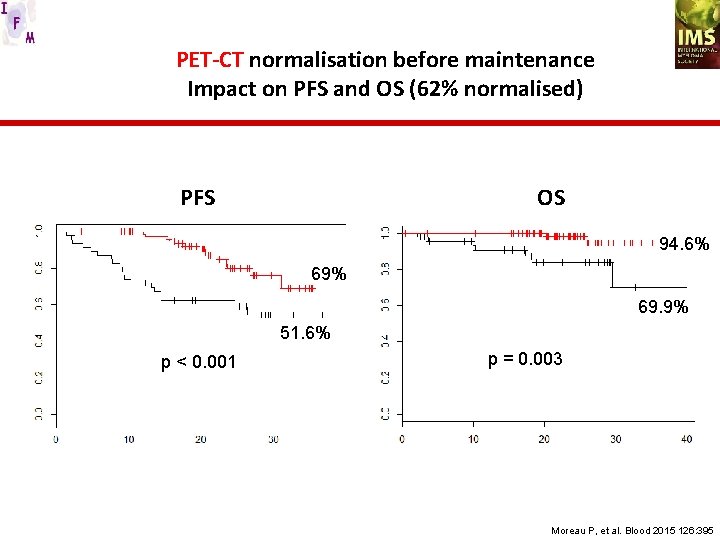
PET-CT normalisation before maintenance Impact on PFS and OS (62% normalised) OS PFS 94. 6% 69. 9% 51. 6% p < 0. 001 p = 0. 003 Moreau P, et al. Blood 2015 126: 395
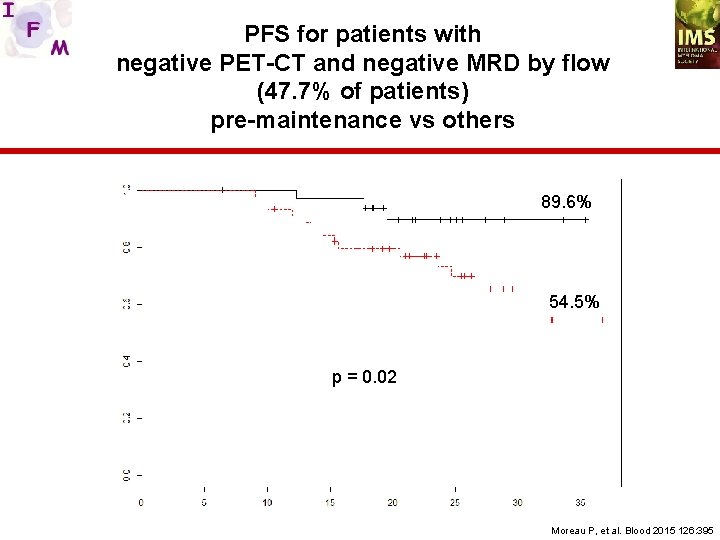
PFS for patients with negative PET-CT and negative MRD by flow (47. 7% of patients) pre-maintenance vs others 89. 6% 54. 5% p = 0. 02 Moreau P, et al. Blood 2015 126: 395

Conclusions - PET-CT and MRI are equally effective to detect bone involvement in symptomatic patients at diagnosis. - PET-CT after 3 cycles of RVD and pre-maintenance is a powerful prognostic marker for PFS - PET-CT pre-maintenance is a powerful prognostic marker for OS - PET-CT and CMF are complementary tools to evaluate minimal residual disease Moreau P, et al. Blood 2015 126: 395

Assessment of Total Lesion Glycolysis and Metabolic Tumor Volume Improve the Clinical Value of Focal Lesion Assessment By FDG PET/CT in Myeloma James E Mc. Donald, Marcus M Kessler, Michael Gardner, Amy Buros, Sarah Waheed, James Ntambi, Frits van Rhee, Maurizio Zangari, Christoph Heuck, Nathan Petty, Carolina Schinke, Sharmilan Thanendrarajan, Alan Mitchell, Antje Hoering, Bart Barlogie, Gareth J Morgan, and Faith E Davies, MD 37

Purpose and design of the study The purpose of this study was to determine whether total lesion glycolysis (TLG) and metabolic tumor volume (MTV) can predict progression free survival (PFS) and overall survival (OS), and to determine whether they are superior to traditional assessment methods (i. e. : detection and enumeration of focal lesions and standardized uptake value (SUV) using 18 F-FDG PET/CT. • 191 patients underwent whole body PET/CT in the Total Therapy 3 A trial and were evaluated using 3 dimensional region of interest analysis with TLG, MTV, and standard measurement parameters derived for all focal lesions with peak SUV above the background red marrow signal. • Survival analysis was performed using Kaplan-Meier and log-rank tests. Univariate and multivariate analyses were performed using Cox proportional hazards regression. 38 Mc. Donald JE, et al. Blood 2015 126: 724
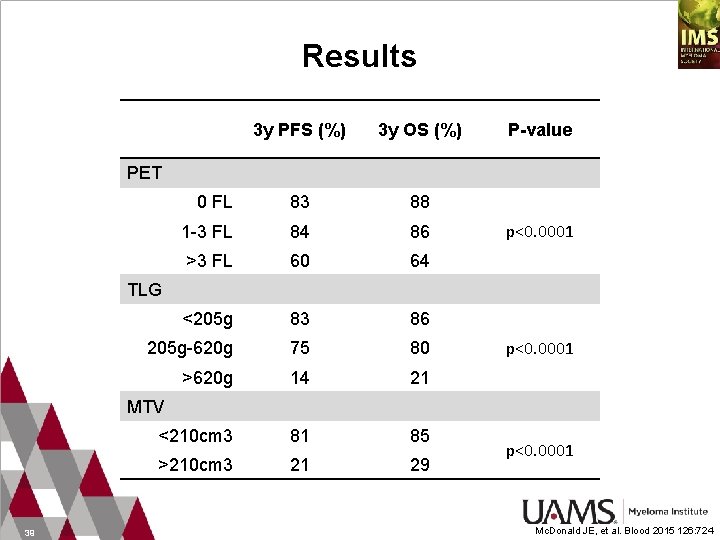
Results 3 y PFS (%) 3 y OS (%) 0 FL 83 88 1 -3 FL 84 86 >3 FL 60 64 <205 g 83 86 205 g-620 g 75 80 >620 g 14 21 <210 cm 3 81 85 >210 cm 3 21 29 P-value PET p<0. 0001 TLG p<0. 0001 MTV 39 p<0. 0001 Mc. Donald JE, et al. Blood 2015 126: 724

Results • In multivariate analysis baseline TLG>620 g retained prognostic significance for predicting PFS and OS together with high B 2 M, LDH and GEP based proliferation. • Importantly baseline TLG >620 g and baseline MTV >210 cm 3 were statistically more predictive of poor PFS and OS than baseline PET with >3 focal lesions in both univariate and multivariate models. 40 Mc. Donald JE, et al. Blood 2015 126: 724

Conclusions • TLG assessment is superior to counting the number of focal lesions. • It is the optimum method for evaluating the extent of focal lesions and to predict clinical outcome. • As this measurement can be standardized using FDA approved software, it should be utilized clinically and in trials going forward. 41 Mc. Donald JE, et al. Blood 2015 126: 724
- Slides: 41