Minimal invasive surgery for pancreatic insulinoma Current evidence































- Slides: 54

Minimal invasive surgery for pancreatic insulinoma: Current evidence Dr. HO Man-fung Prince of Wales Hospital

CURRENT TREATMENT

Medical therapy Dietary modification n Diazoxide n Somatostatin analogue n Minimal effect on disease progression (especially for non-responder) n Pre-operative symptoms control n

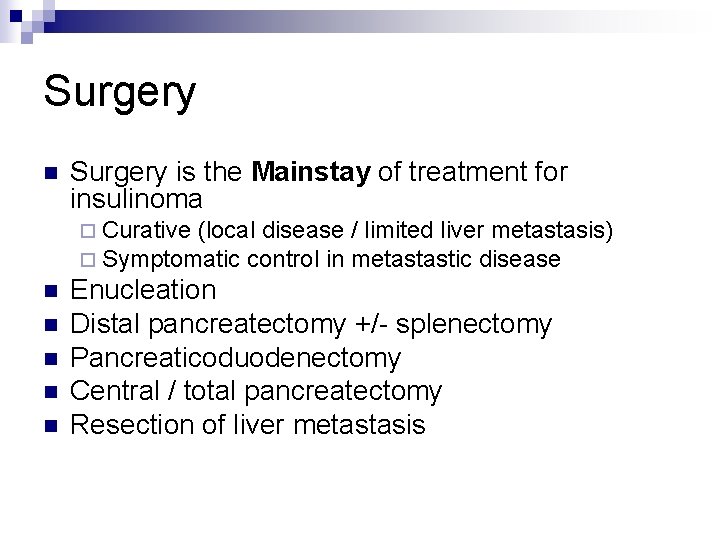
Surgery n Surgery is the Mainstay of treatment for insulinoma ¨ Curative (local disease ¨ Symptomatic control in n n / limited liver metastasis) metastastic disease Enucleation Distal pancreatectomy +/- splenectomy Pancreaticoduodenectomy Central / total pancreatectomy Resection of liver metastasis

Special concerns of insulinoma n Small size < 2 cm in ~ 80% ¨ n 90% benign and solitary ¨ ¨ n Difficulty in localization Resection strategy Room for minimal invasive surgery Overt symptoms, poorly controlled by drugs ¨ ¨ Pre-operative control of symptoms Even palliative resection in metastatic disease wound be beneficial

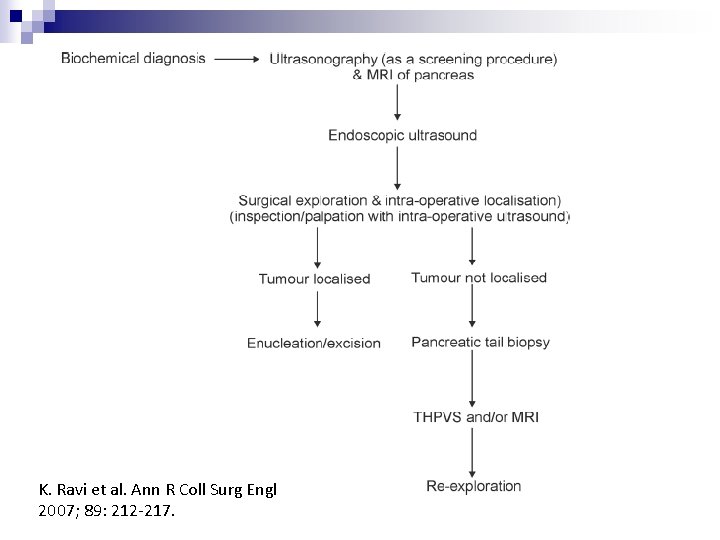
Surgical approach Pre-operative localization n IOUS n Enucleation or Pancreatectomy n ? Blind distal pancreatectomy n

n Lo et al. Surgical Endoscopy (2004) 18: 297 -302 ¨ n K. Ravi et al. Ann R Coll Surg Engl 2007; 89: 212217. ¨ n 60% with CT, 80% with EUS, 100% with THPVS 67% (incl. USG, CT, MRI, THPVS) Mehrdad Nikfarjam et al. Annals of Surgery • Volume 247, Number 1, January 2008 ¨ 29 -80% with non invasive investigation, 85 -100% with invasive investigations

Surgical approach Pre-operative localization n IOUS n Enucleation or Pancreatectomy n ? Blind distal pancreatectomy n

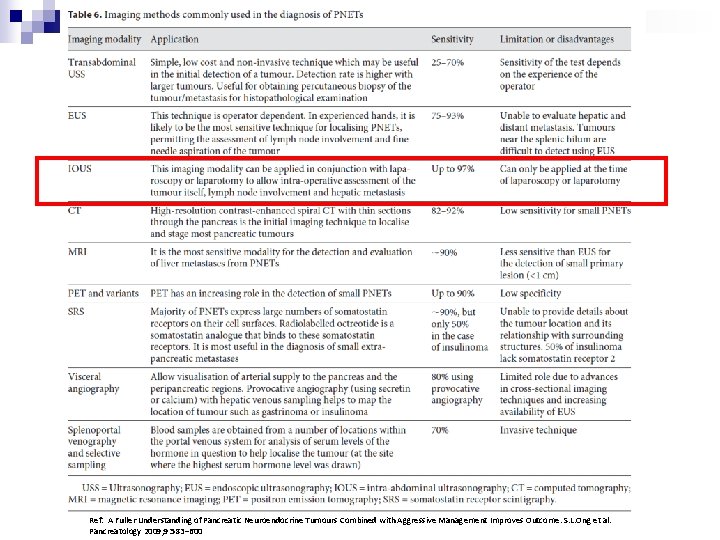
Ref: A Fuller Understanding of Pancreatic Neuroendocrine Tumours Combined with Aggressive Management Improves Outcome. S. L. Ong et al. Pancreatology 2009; 9: 583– 600

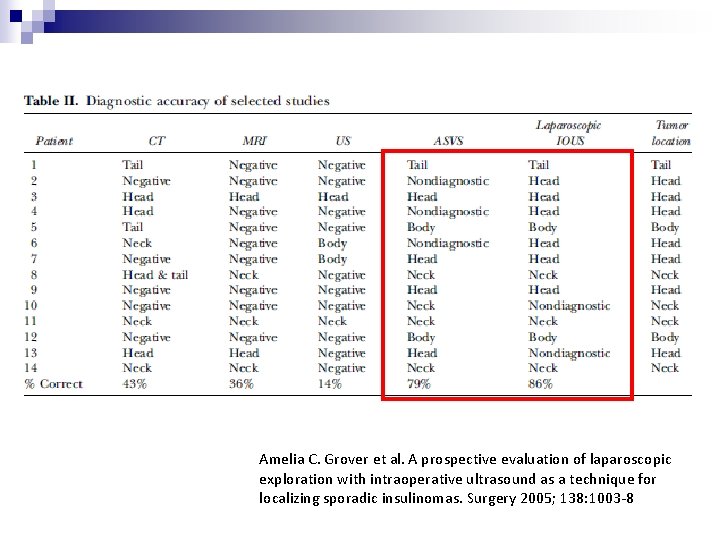
Amelia C. Grover et al. A prospective evaluation of laparoscopic exploration with intraoperative ultrasound as a technique for localizing sporadic insulinomas. Surgery 2005; 138: 1003 -8

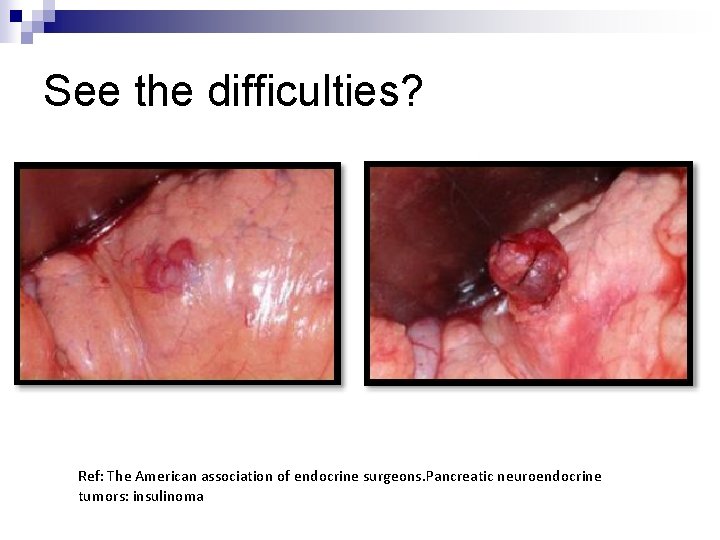
See the difficulties? Ref: The American association of endocrine surgeons. Pancreatic neuroendocrine tumors: insulinoma

Surgical approach Pre-operative localization n IOUS n Enucleation or Pancreatectomy n ? Blind distal pancreatectomy n

n Enucleation ¨ Solitary lesion ¨ Size < 2 cm ¨ Away from major vessels / pancreatic ducts n Pancreatectomy ¨ Multifocal lesions ¨ Size > 2 cm ¨ Close to major vessels / pancreatic ducts ¨ MEN 1

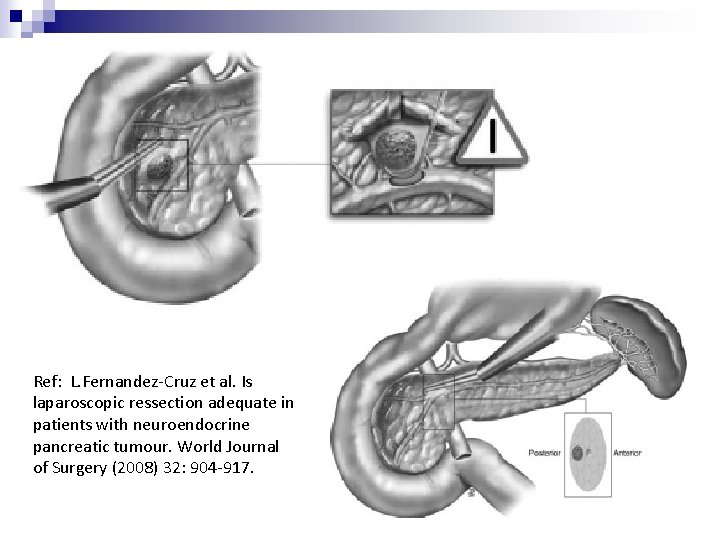
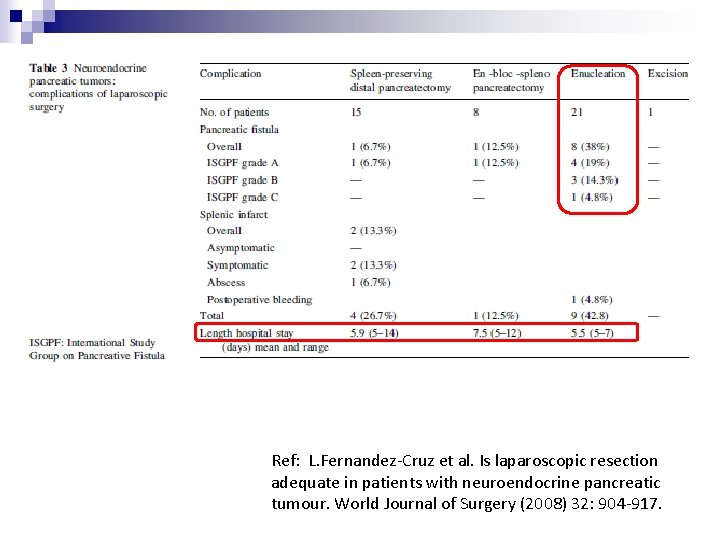
Ref: L. Fernandez-Cruz et al. Is laparoscopic ressection adequate in patients with neuroendocrine pancreatic tumour. World Journal of Surgery (2008) 32: 904 -917.

Surgical approach Pre-operative localization n IOUS n Enucleation or Pancreatectomy n ? Blind distal pancreatectomy n

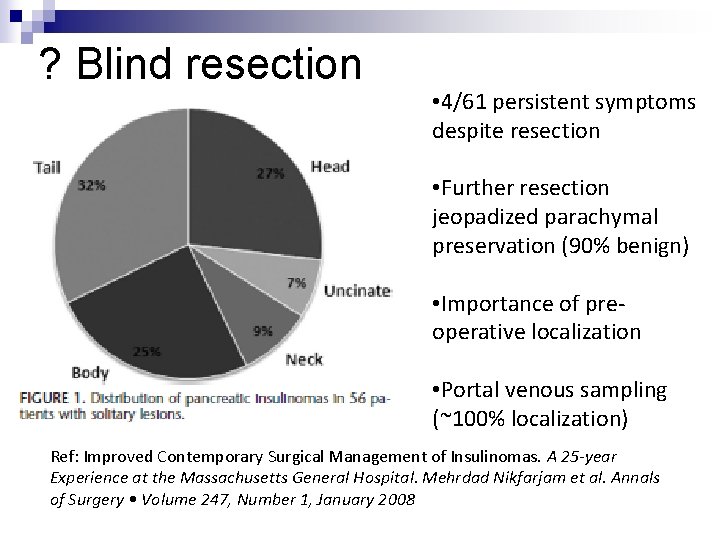
? Blind resection • 4/61 persistent symptoms despite resection • Further resection jeopadized parachymal preservation (90% benign) • Importance of preoperative localization • Portal venous sampling (~100% localization) Ref: Improved Contemporary Surgical Management of Insulinomas. A 25 -year Experience at the Massachusetts General Hospital. Mehrdad Nikfarjam et al. Annals of Surgery • Volume 247, Number 1, January 2008

K. Ravi et al. Ann R Coll Surg Engl 2007; 89: 212 -217.

Management of liver metastasis Resection n Transarterial chemoembolization n Ablation n Systemic chemotherapy n Targeted therapy(e. g. Sunitinib, everolimus) n Liver transplantation n

MINIMAL INVASIVE SURGERY

Lapasroscpic pancreatic resection n 1 st laparoscopic pancreatic resection -1992 Gagner M et al (1996). J Gastrointest Surg 1: 20 -26 ¨ Cushieri A. et al (1996). Ann Surg 223: 280 -285 ¨ n 1 st laparoscopic resection of insulinoma – 1992 n Low incidence and difficult anatomical location, laparoscopic experience published relatively late compared to other laparoscopic surgery

n n 2 enucleations and 2 distal pancreatectomies done laparoscopically 100% pre-operative localization Only for lesions over body and tail 1 patient with post operative pancreatic leakage

n n n 1 st comparative study of laparoscopic vs open approach (12 vs 9) Comparison with historical cohort No significant difference in morbidty, mortality, intraoperative variables Only 1 patient used intra-operative USG Denied use of intra-operative USG to be necessary

Laparoscopic USG n Only way to replace palpation in laparoscopic surgery n Localization of lesion(s) Sensitivity 83 -98%a ¨ Comparable to THPVS b ¨ n a) b) Define anatomical relationship with major vessels, main pancreatic duct Mehrdad Nikfarjam et al. Improved Contemporary Surgical Management of Insulinomas. A 25 -year Experience at the Massachusetts General Hospital. . Annals of Surgery • Volume 247, Number 1, January 2008 Amelia C. Grover et al. A prospective evaluation of laparoscopic exploration with intraoperative ultrasound as a technique for localizing sporadic insulinomas. Surgery 2005; 138: 1003 -8

n n n p. NETs with pancreatic resection, 20 patients with insulinoma Pre-op localization 100% 1/20 conversion to open Mean follow up of 36 months, no recurrence Significant less blood loss and operative time for laparoscopic enucleation

n n n n 21 patients Pre-operative localization - unknown 1/21 converted open IOUS: localization, intraoperative decision, marking of transection line 3 patients with pancreatic fistula All except 1 discharged in 1 week No recurrence

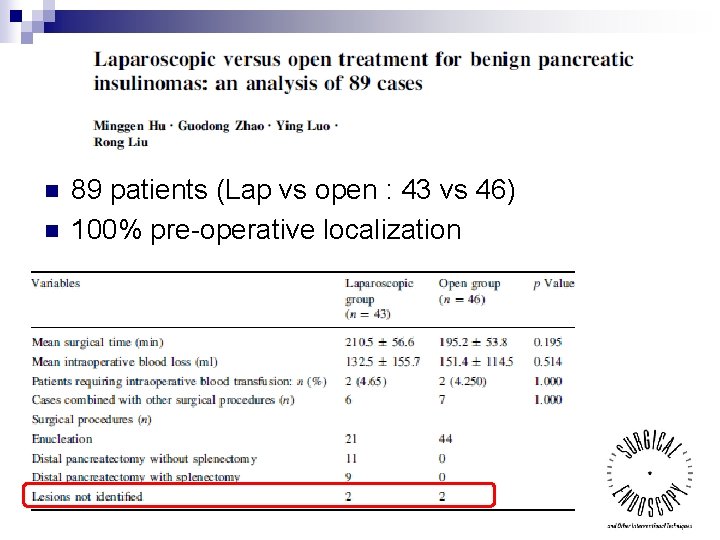
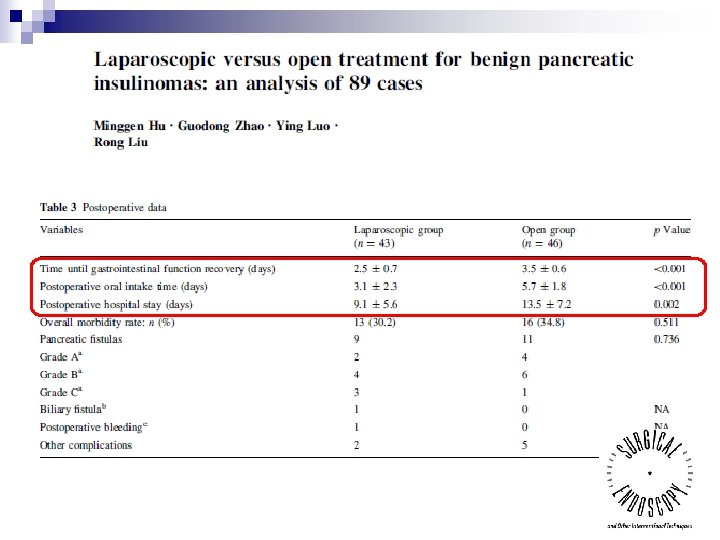
n n 89 patients (Lap vs open : 43 vs 46) 100% pre-operative localization


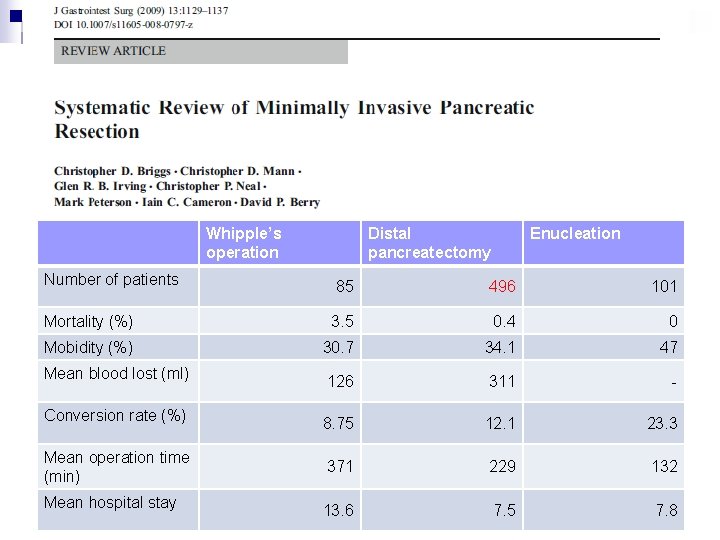
Whipple’s operation Distal pancreatectomy Enucleation Number of patients 85 496 101 Mortality (%) 3. 5 0. 4 0 Mobidity (%) 30. 7 34. 1 47 Mean blood lost (ml) 126 311 - Conversion rate (%) 8. 75 12. 1 23. 3 Mean operation time (min) 371 229 132 Mean hospital stay 13. 6 7. 5 7. 8

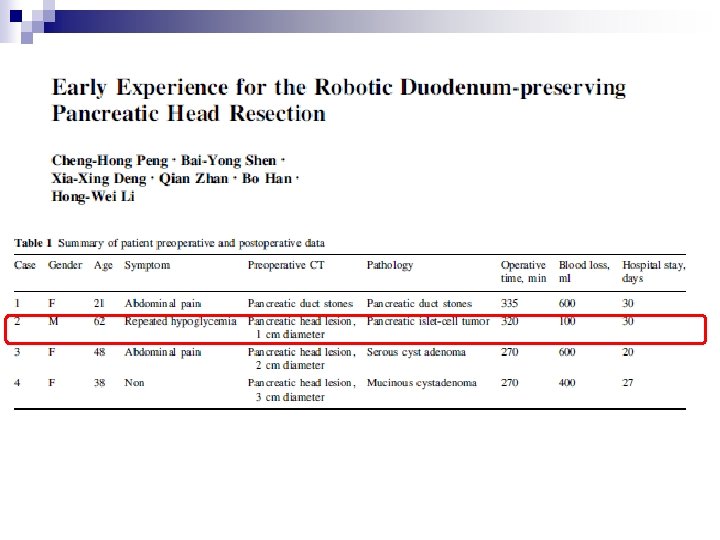
Represent early experience n Highly selected cases n Indicating minimal invasive surgery is feasible n Pancreatic fistula is still the most prevalent complication n Lacking of long term results, e. g. survival, recurrence n Heterogeneous disease n

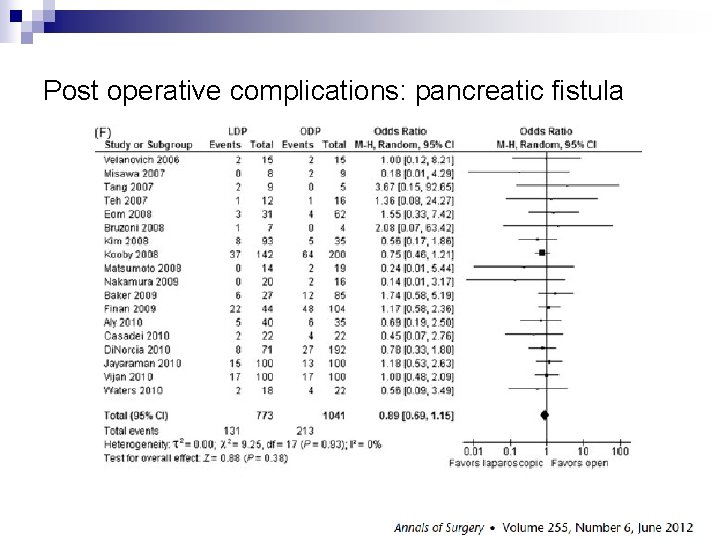
n Total n= 1814 (18 studies) n n Laparoscopic arm (LDP): 773 (43%) Open arm : 1041 (57%) n Conversion rate : 0 – 30% (not reported in 4 studies)

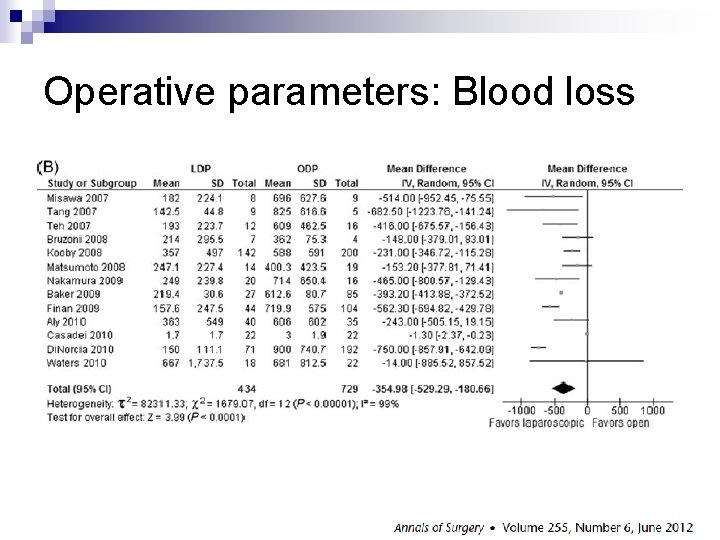
Operative parameters: Blood loss

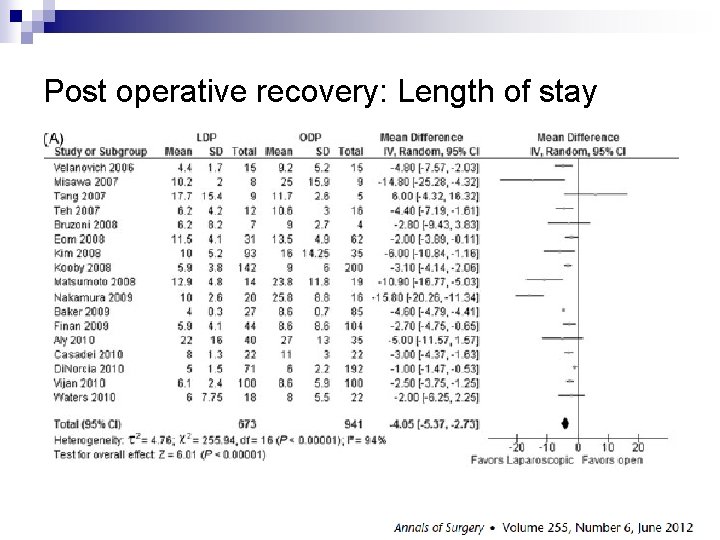
Post operative recovery: Length of stay

Post operative complications: pancreatic fistula

n Laparoscopic distal pancreatectomy has the advantage of: ¨ Lower blood loss ¨ Faster recovery ¨ Comparable complication profile with open approach n This technique is a reasonable approach in selected cancer patients

What about robots?

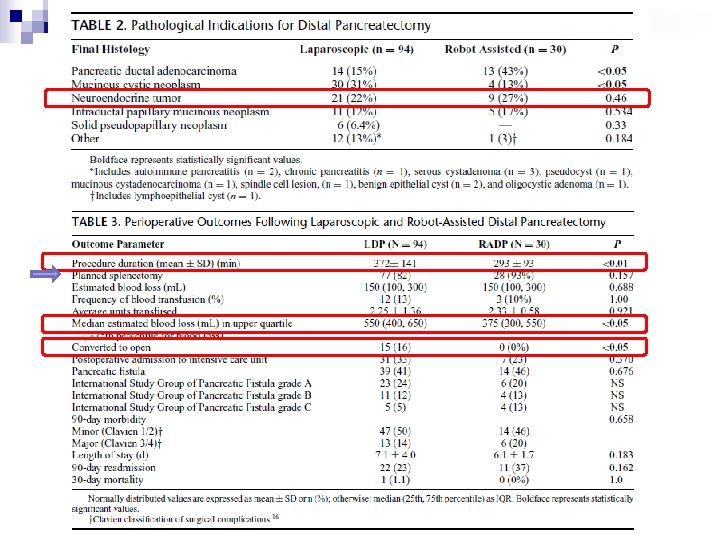
n n n 1 st 30 cases in University of Pittsburgh Compared with 94 patient with laparoscopic distal pancreatectomy 0% conversion rate 100% R 0 resection Median LN harvest (Lap vs Robot = 9 vs 19)


v n n n Better visualization, freedom of movement, stability Preservation of spleen and splenic vessels 21/22 patient with successful splenic preservation 17 patient with splenic vessels preserved 1 patient developed post op splenic infarct Further expand the advantage minimal invasive surgery by improving rate of spleen preservation


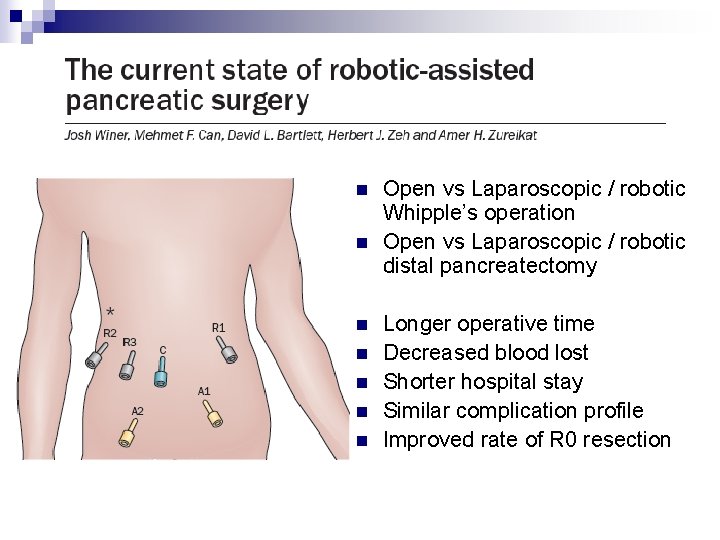
n n n n Open vs Laparoscopic / robotic Whipple’s operation Open vs Laparoscopic / robotic distal pancreatectomy Longer operative time Decreased blood lost Shorter hospital stay Similar complication profile Improved rate of R 0 resection

Robotic pancreatic surgery n n n Literature in its infancy Small cohort available even for high volume centres Case selection bias Learning curve Long term results ?

Local ablative therapy n Percutaneous RFA ablation ¨ Stephan Limer et al. European Journal of Gastroenterology and Hepatology 2009, 21: 10971101 n EUS Guided ablation of insulinoma: a new treatment option ¨ Michael J. Levy et al. gastrointestinal Endoscopy, Vol 75, No. 1; 200 -206

Conclusion n n Insulinoma is benign most of the time, but causing significant biochemical disturbance that require surgical treatment Localization is of utmost importance in surgical success Laparoscopic surgery offers treatment with less trauma and similar safety profile Novel treatment for surgically unfit individuals

Conclusion n Insulinoma is an ideal entity for minimal invasive pancreatic surgery ¨ Lesion are small and benign most of the time ¨ no concern for involved margin, lymphatic dissection n Laparoscopic and robotic pancreatic surgery is feasible for management of pancreatic insulinoma


INSULINOMA

Insulinoma Subgroup of pancreatic neuroendocrine tumours (p. NETs) n Commonest functional p. NETs (25%) n Incidence : 4 in 1, 000 n Unsuppressed production of endogenous insulin n As part of genetic syndromes (5 -8%) n ¨ MEN I, VHL, NF I, TS

Presentation n Whipple’s triad: ¨ Fasting hypoglycaemia (< 2. 2 mmol/L) ¨ Symptomatic hypoglycaemia (autonomic and neuroglycaemic symptoms) ¨ Relieve of symptoms after administration of glucose Weight gain n Other related syndromes n

Biochemical diagnosis Ref: A Fuller Understanding of Pancreatic Neuroendocrine Tumours Combined with Aggressive Management Improves Outcome. S. L. Ong et al. Pancreatology 2009; 9: 583– 600

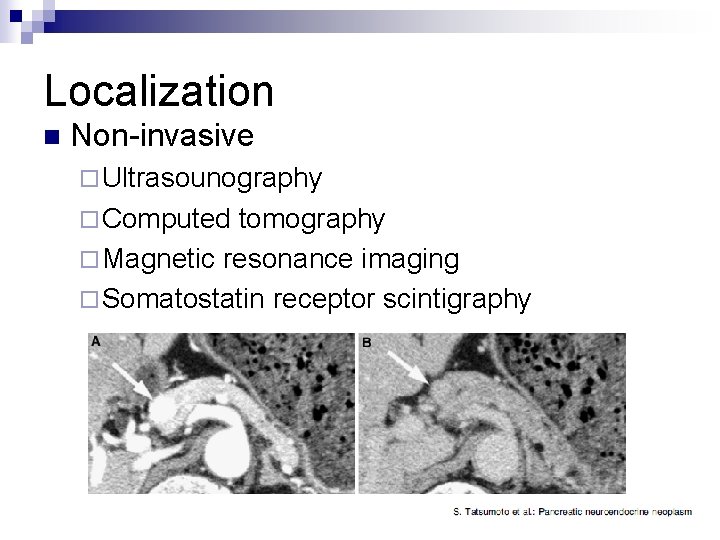
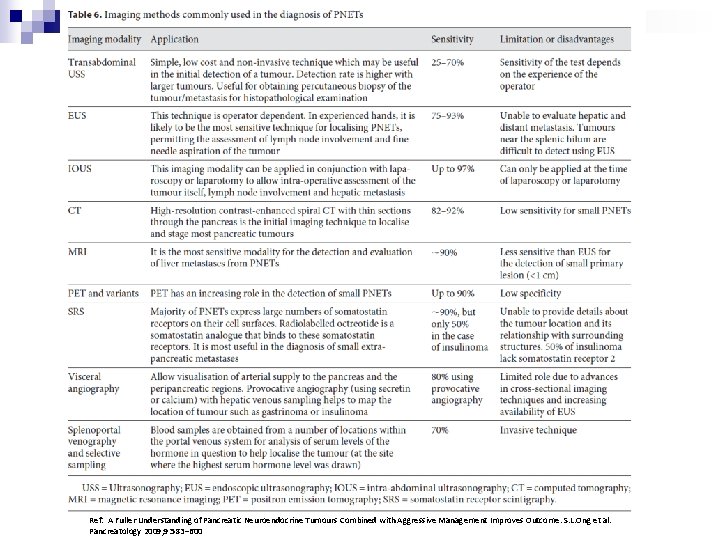
Localization n Non-invasive ¨ Ultrasounography ¨ Computed tomography ¨ Magnetic resonance imaging ¨ Somatostatin receptor scintigraphy

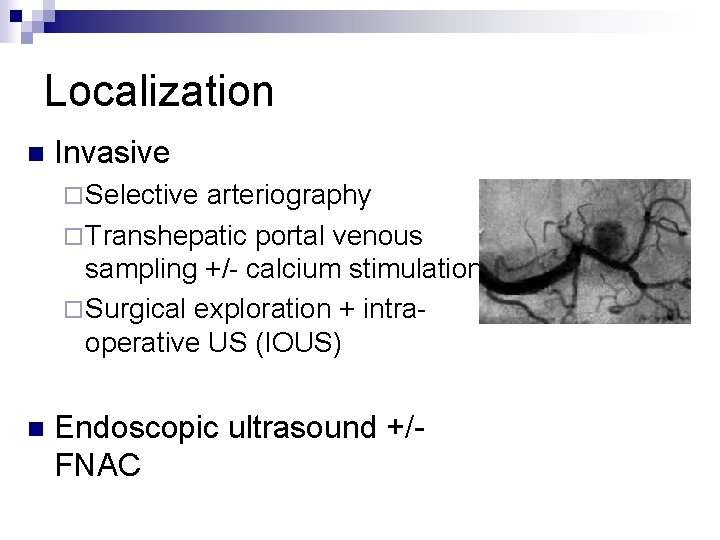
Localization n Invasive ¨ Selective arteriography ¨ Transhepatic portal venous sampling +/- calcium stimulation ¨ Surgical exploration + intraoperative US (IOUS) n Endoscopic ultrasound +/FNAC

Ref: A Fuller Understanding of Pancreatic Neuroendocrine Tumours Combined with Aggressive Management Improves Outcome. S. L. Ong et al. Pancreatology 2009; 9: 583– 600

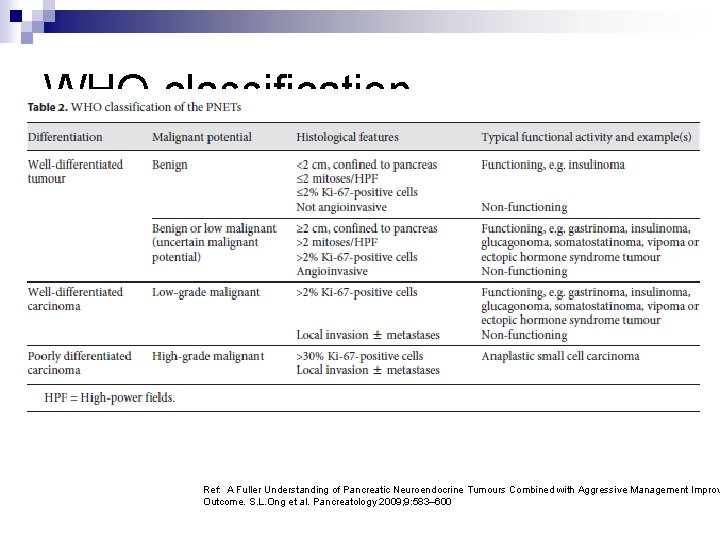
WHO classification Ref: A Fuller Understanding of Pancreatic Neuroendocrine Tumours Combined with Aggressive Management Improv Outcome. S. L. Ong et al. Pancreatology 2009; 9: 583– 600

Ref: L. Fernandez-Cruz et al. Is laparoscopic resection adequate in patients with neuroendocrine pancreatic tumour. World Journal of Surgery (2008) 32: 904 -917.