Minerals Minerals the building blocks of rocks Definition

Minerals

Minerals: the building blocks of rocks • Definition of a Mineral: ü naturally occurring ü inorganic ü solid ü characteristic crystalline structure ü definite chemical composition

How do we identify minerals? • Physical properties: ü ü ü ü Color Luster Hardness Crystal shape Cleavage Specific gravity Other

Physical Properties of Minerals • Color: – Most obvious, but often misleading – Different colors may result from impurities Example: Quartz

Physical Properties of Minerals • Color: Streak – color of a mineral in powdered form (used for metallic minerals) Obtained by scratching a mineral on a piece of unglazed porcelain (Streak Plate). Example: Hematite

Physical Properties of Minerals • Luster: – How a mineral surface reflects light – Two major types: • Metallic luster • Non-metallic luster Metallic example: Galena Non-metallic example: Orthoclase

Physical Properties of Minerals • Hardness: – How easy it is to scratch a mineral – Mohs Scale of Hardness • relative scale • consists of 10 minerals, ranked 1 (softest) to 10 (hardest)
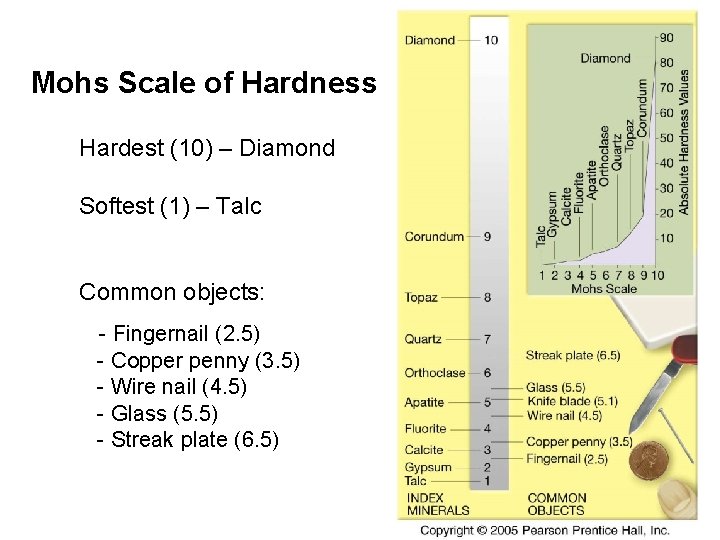
Mohs Scale of Hardness Hardest (10) – Diamond Softest (1) – Talc Common objects: - Fingernail (2. 5) - Copper penny (3. 5) - Wire nail (4. 5) - Glass (5. 5) - Streak plate (6. 5)

Physical Properties of Minerals • Crystal shape (or form): – external expression of a mineral’s internal atomic structure – planar surfaces are called crystal faces – angles between crystal faces are constant for any particular mineral Quartz Pyrite

Physical Properties of Minerals • Cleavage vs. Fracture: – The way a mineral breaks – Cleavage: tendency of a mineral to break along planes of weakness – Minerals that do not exhibit cleavage are said to fracture Do not confuse cleavage planes with crystal faces! Crystal faces are just on the surface and may not repeat when the mineral is broken.

Physical Properties of Minerals • Cleavage is described by: – Number of planes – Angles between adjacent planes – These are constant for a particular mineral

Physical Properties of Minerals • Cleavage (1 direction): Example: mica

Physical Properties of Minerals • Cleavage (2 directions): orthoclase amphibole

Physical Properties of Minerals • Cleavage (3 directions): halite calcite

Physical Properties of Minerals • Cleavage (4 directions): fluorite

Physical Properties of Minerals • Fracture: – minerals that do not exhibit cleavage are said to fracture – smooth, curved surfaces when minerals break in a glass-like manner: conchoidal fracture Quartz

Physical Properties of Minerals • Specific gravity: – weight of a mineral divided by weight of an equal volume of water – metallic minerals tend to have higher specific gravity than non-metallic minerals Galena SG=7. 5 Quartz SG=2. 67

Physical Properties of Minerals • Other properties: – reaction with hydrochloric acid (calcite fizzes) – taste (halite tastes salty) – feel (talc feels soapy, graphite feels greasy) – magnetism (magnetite attracts a magnet)

Mineral Groups • Rock-forming minerals – ~30 common minerals make up most rocks in Earth’s crust – Composed mainly of the 8 elements that make up over 98% of the crust

Mineral Groups Element Abundances SILICATES Silica (Si. O 4)4 - Common cations that bond with silica anions All others: 1. 5%

Mineral Groups • • Silicates (most abundant) Non-silicates (~8% of Earth’s crust): – – – Oxides Carbonates Sulfides Sulfates Halides Native elements O 2(CO 3)2 S 2(SO 4)2 Cl-, F-, Br(single elements; e. g. , Au)
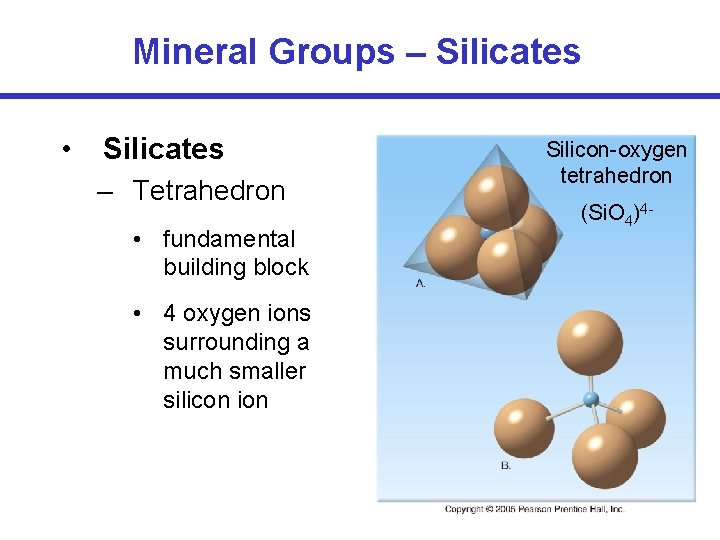
Mineral Groups – Silicates • Silicates – Tetrahedron • fundamental building block • 4 oxygen ions surrounding a much smaller silicon ion Silicon-oxygen tetrahedron (Si. O 4)4 -

Mineral Groups – Silicates • Joining Silicate Structures – How tetrahedra may be linked: • independent tetrahedra • single chains • double chains • sheets • 3 -D framework
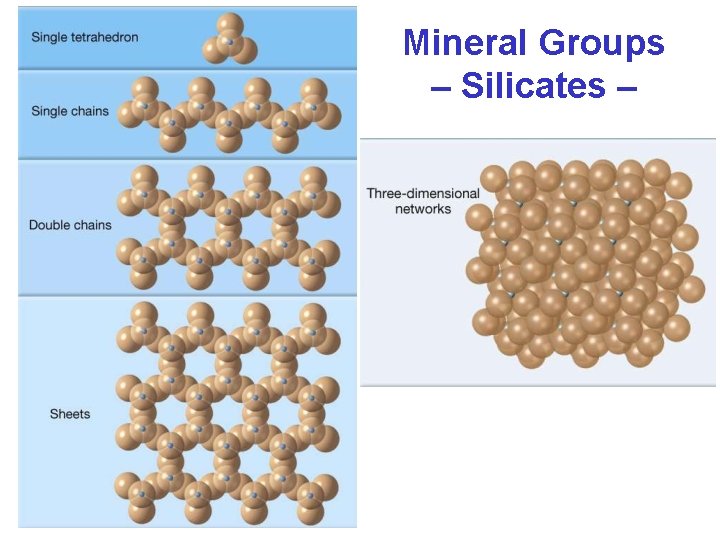
Mineral Groups – Silicates –

Mineral Groups – Silicates Olivine Group dark silicates (Fe-Mg) ferromagnesian No cleavage

Mineral Groups – Silicates Pyroxene Group Ferromagnesian / dark silicates (Fe-Mg) Augite 2 -directions of cleavage (at nearly 90 degrees)

Mineral Groups – Silicates Amphibole Group Ferromagnesian / dark silicates (Ca, Fe-Mg) Hornblende 2 -directions of cleavage (not at 90 degrees)

Mineral Groups – Silicates Mica Group and Clay Minerals light silicates (K, Al) non-ferromagnesian Muscovite 1 -direction of cleavage

Mineral Groups – Silicates Feldspar Group K-feldspar light silicates (K-Na-Ca, Al) Most common mineral group Orthoclase Plagioclase 2 -directions of cleavage (at 90 degrees) Ca/Na-feldspar

Mineral Groups – Silicates Quartz light silicates (pure Si. O 2) no cleavage (conchoidal fracture) hard, resistant to weathering Quartz
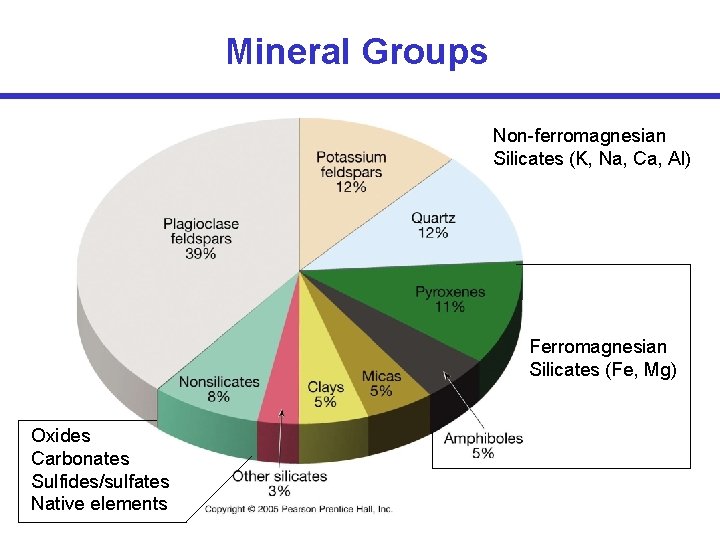
Mineral Groups Non-ferromagnesian Silicates (K, Na, Ca, Al) Ferromagnesian Silicates (Fe, Mg) Oxides Carbonates Sulfides/sulfates Native elements
- Slides: 31