Mineralized Tissues Hard Macrostructure of Bones Long Bones
Mineralized Tissues Hard Macrostructure of Bones Long Bones Short Bones Flat Bones Irregular Bones Bone Composition - Ultrastructure Cortical Bone Woven Bone Lamellar Bone Circumferential Lamellar Bone Primary Osteonal Bone Secondary Osteonal Bone Trabecular Bone Teeth Material Properties of Hard Tissues Structural Properties of Hard Tissues Mechanical Properties of Trabecular Tissue Contribution of Components to Whole Bone Strength Viscoelastic Properties of Bone Viscoelastic Model of Bone Properties Bone as a Composite Material - Model 1 Bone as a Compostie Material - Model 2 Fatigue of Bone Mechanical Properties of Whole Bones Wolff's Law Biomateriali - Prof. O. Sbaizero Tissues
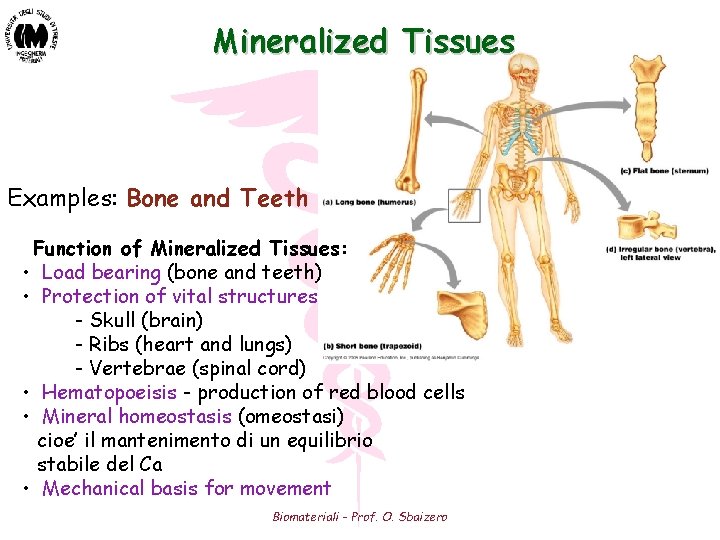
Mineralized Tissues Examples: Bone and Teeth Function of Mineralized Tissues: • Load bearing (bone and teeth) • Protection of vital structures - Skull (brain) - Ribs (heart and lungs) - Vertebrae (spinal cord) • Hematopoeisis - production of red blood cells • Mineral homeostasis (omeostasi) cioe’ il mantenimento di un equilibrio stabile del Ca • Mechanical basis for movement Biomateriali - Prof. O. Sbaizero

L’osso e’ una forma di tessuto connettivo specializzato in cui le componenti extracellulari sono mineralizzate; cio’ conferisce all’osso notevoli proprieta’: • notevole rigidita’ e resistenza • un certo grado di elasticita’ La struttura delle singole ossa fornisce la massima resistenza alla tensione meccanica pur mantenendo la minor massa possibile. Per permettere modificazioni meccaniche le ossa sono in uno stato di crescita dinamica e di riassorbimento per tutta la vita. Biomateriali - Prof. O. Sbaizero

Struttura dell’ osso Il tessuto osseo è un tessuto connettivo di sostegno. Circa un terzo della matrice dell’osso è costituito da fibre collagene; il resto è una miscellanea di sali di Ca, principalmente fosfato di Calcio ed in minor misura carbonato di Calcio. Questa combinazione conferisce all’osso eccezionali proprietà meccaniche: i cristalli di fosfato di Calcio sono molto resistenti ma anelatici e piuttosto friabili, infatti resistono bene a compressione ma si rompono facilmente se vengono piegati o sottoposti a forze improvvise; le fibre collagene sono estremamente flessibili e possono essere facilmente allungate, ripiegate o torte, ma non sopportano la compressione. Biomateriali - Prof. O. Sbaizero

Struttura dell’ osso Nell’osso i minerali sono organizzati attorno alle fibre collagene, che costituiscono un’impalcatura organica per la formazione dei cristalli. Questi cristalli minerali sono composti da un tipo particolare di fosfato di Calcio, chiamato Idrossiapatite. Il risultato è una combinazione di proteine e cristalli che possiede proprietà intermedie fra quelle del collagene e quelle dei cristalli minerali: si ottiene una combinazione dura ma in qualche modo flessibile e molto resistente alla frantumazione, essendo inoltre molto leggera. Se diminuisce il contenuto minerale del tessuto osseo, pur conservando le dimensioni degli elementi scheletrici, si va incontro a rammollimento delle ossa (osteomalacia). Biomateriali - Prof. O. Sbaizero

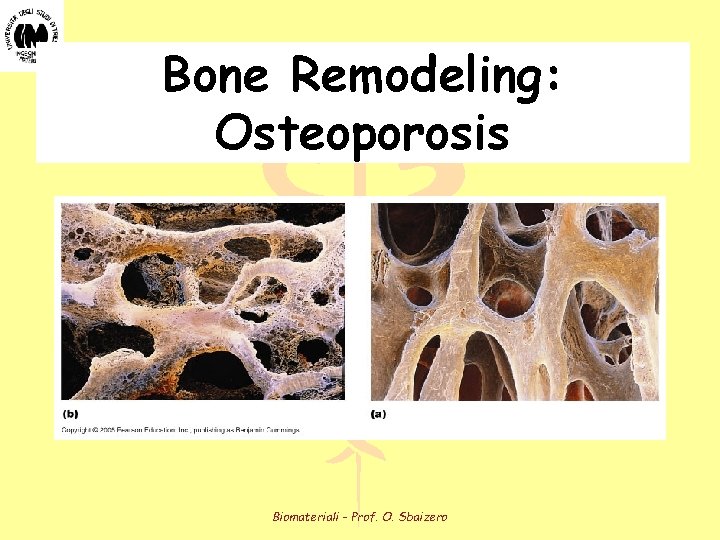
Struttura dell’ osso Il tessuto osseo cresce e si rigenera grazie all’attività di due classi di cellule: osteoclasti ed osteoblasti. Gli osteoclasti producono acidi che dissolvono la matrice ossea, liberando i sali minerali ivi contenuti. Gli osteoblasti sono responsabili dell’osteogenesi: sintetizzano le componenti organiche della matrice ossea, producendo l’osteoide (miscela risultante di fibre collagene, proteoglicani e glicoproteine). Esso viene successivamente mineralizzato, mediante deposito di cristalli di sali di Calcio, arrivando così alla formazione di osso nuovo. Il punto in cui comincia il processo di ossificazione è chiamato centro di ossificazione; da questo l’osso si espande in direzione centrifuga. Man mano che l’ossificazione continua, alcuni osteoblasti rimangono intrappolati all’interno di minuscole tasche ossee (lacune), differenziandosi in osteociti, che secernono il collagene. E’ necessario che vi sia equilibrio tra i due processi di rimozione e sintesi della matrice; se prevale l’azione di una delle due famiglie, si hanno alterazioni: si hanno osteoporosi o scorbuto nel caso in cui predominino gli osteoclasti, in tal caso le ossa diventano deboli e fragili. Biomateriali - Prof. O. Sbaizero

L’osso e’ composto da cellule e da una matrice organica extracellulare La componente minerale dell’osso e’ l’idrossiapatite, idrossiapatite mentre la parte fibrosa e’ collagene Le cellule dell’osso sono di 3 tipi: • osteoblasti : cellule immature, responsabili della sintesi e della secrezione della sostanza osteoide, che si mineralizza per formare l’osso. • Gli osteoblasti sono confinati nell’osso e diventano osteociti e sono responsabili del mantenimento della matrice dell’osso. – 90% of cells in mature skeleton –Maintain structural integrity of bone –Serve to maintain bone and help control extracellular concentrations of Ca and Phos. • osteoclasti : sono cellule multinucleate e attivamente coinvolte in processi di riassorbimento associati al continuo rimodellamento dell’osso Biomateriali - Prof. O. Sbaizero

I processi di edificazione sono dovuti alla proliferazione degli osteoblasti, quelli di demolizione agli osteoclasti che esplicano attraverso il meccanismo del riassorbimento lacunare. Gli osteoclasti hanno la capacità di consumare lentamente il tessuto osseo grazie alla secrezione di acido lattico, che scioglie i minerali di Ca e di Mg depositati sull’osso, e di uno speciale enzima proteolitico che scompone e digerisce la sostanza organica del tessuto osseo in cui i sali sono contenuti. Biomateriali - Prof. O. Sbaizero
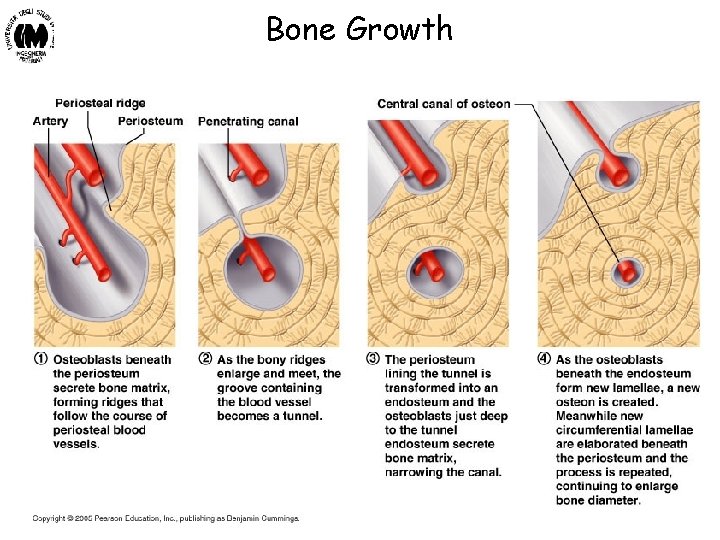
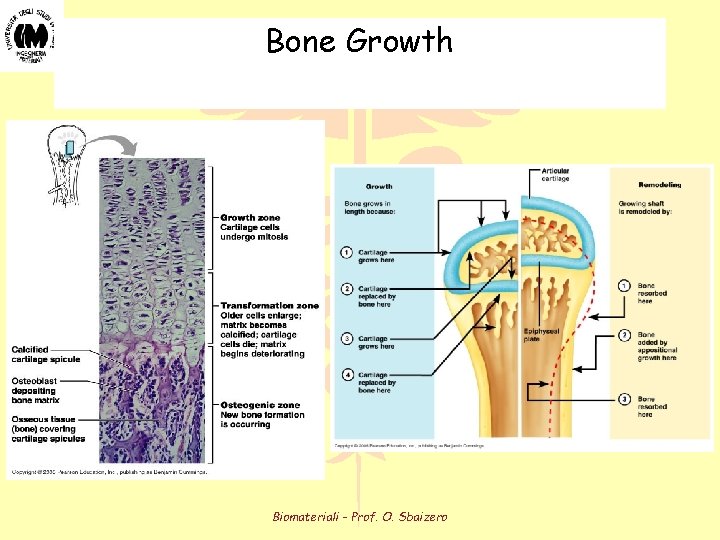
Bone Growth Biomateriali - Prof. O. Sbaizero
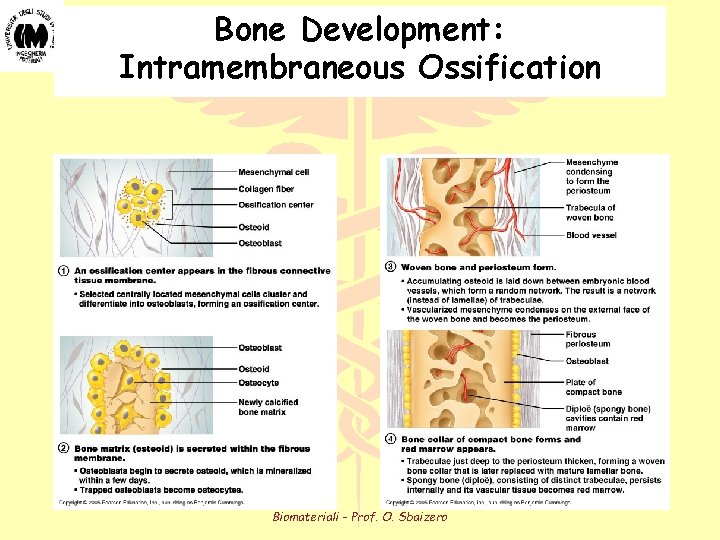
Bone Development: Intramembraneous Ossification Biomateriali - Prof. O. Sbaizero
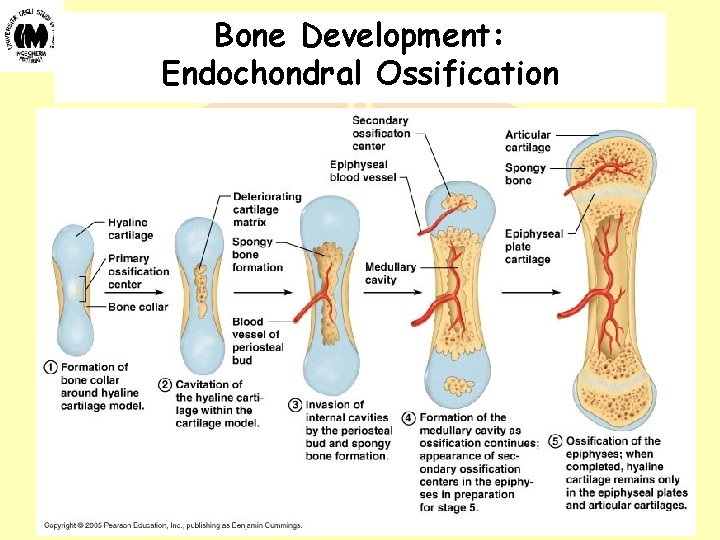
Bone Development: Endochondral Ossification Biomateriali - Prof. O. Sbaizero
Bone Growth Biomateriali - Prof. O. Sbaizero
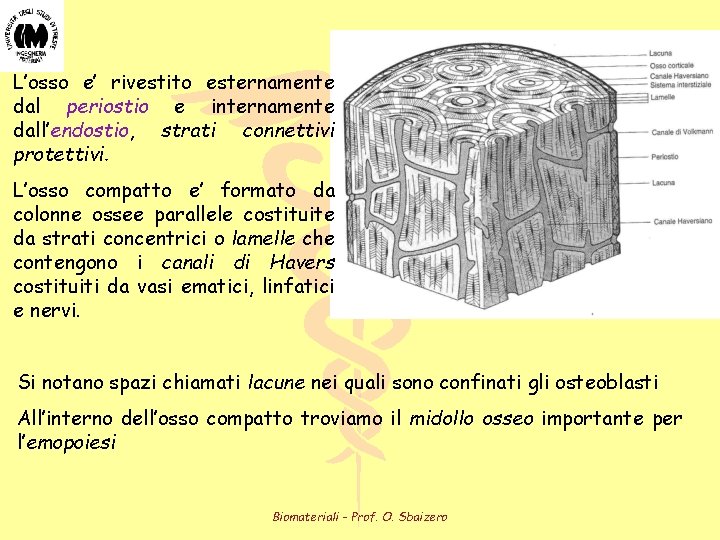
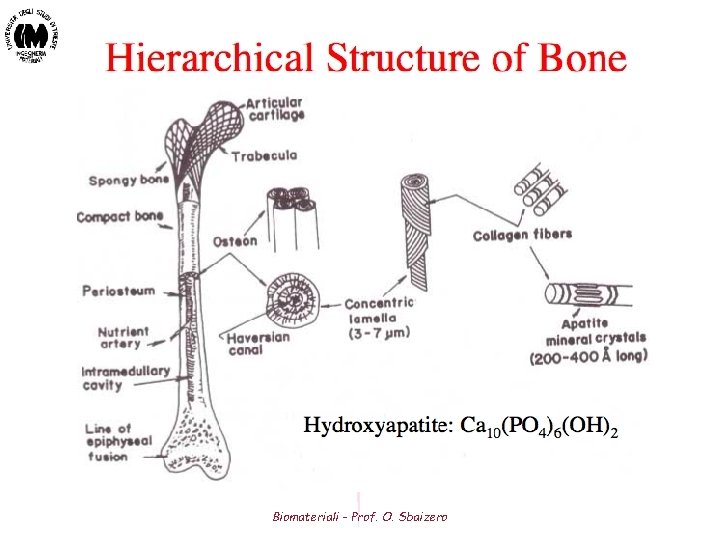
L’osso e’ rivestito esternamente dal periostio e internamente dall’endostio, strati connettivi protettivi. le ossa L’osso compatto e’ formato da colonne ossee parallele costituite da strati concentrici o lamelle che contengono i canali di Havers costituiti da vasi ematici, linfatici e nervi. Si notano spazi chiamati lacune nei quali sono confinati gli osteoblasti All’interno dell’osso compatto troviamo il midollo osseo importante per l’emopoiesi Biomateriali - Prof. O. Sbaizero
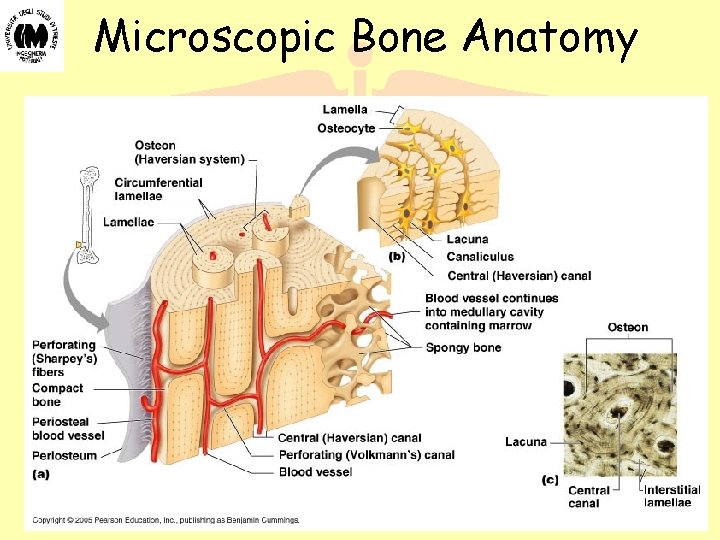
Microscopic Bone Anatomy Biomateriali - Prof. O. Sbaizero
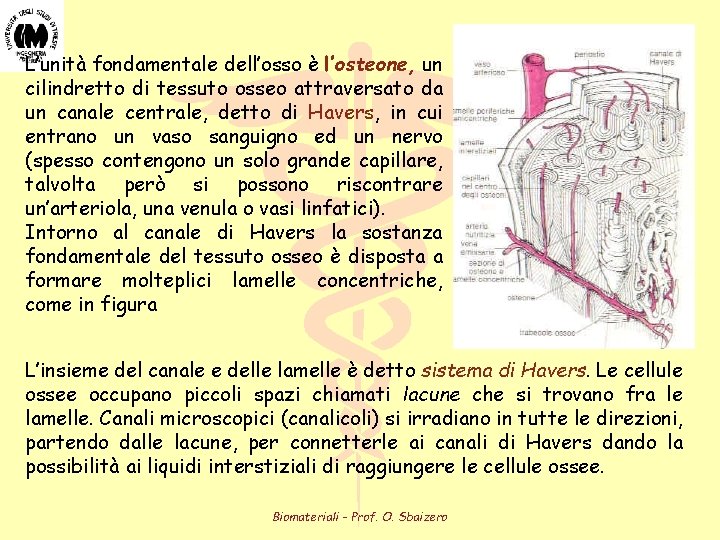
L’unità fondamentale dell’osso è l’osteone, un cilindretto di tessuto osseo attraversato da un canale centrale, detto di Havers, in cui entrano un vaso sanguigno ed un nervo (spesso contengono un solo grande capillare, talvolta però si possono riscontrare un’arteriola, una venula o vasi linfatici). Intorno al canale di Havers la sostanza fondamentale del tessuto osseo è disposta a formare molteplici lamelle concentriche, come in figura L’insieme del canale e delle lamelle è detto sistema di Havers. Le cellule ossee occupano piccoli spazi chiamati lacune che si trovano fra le lamelle. Canali microscopici (canalicoli) si irradiano in tutte le direzioni, partendo dalle lacune, per connetterle ai canali di Havers dando la possibilità ai liquidi interstiziali di raggiungere le cellule ossee. Biomateriali - Prof. O. Sbaizero

Nell’ osso compatto i sistemi haversiani sono fittamente addensati l’uno all’altro ed i piccoli spazi interposti sono riempiti da lamelle interstiziali. Nell’ osso spugnoso invece, vi sono parecchi spazi vuoti compresi tra esili processi ossei (trabecole) che sono uniti gli uni agli altri quasi come le travi di una impalcatura. Biomateriali - Prof. O. Sbaizero

L’osso esiste in due forme principali: osso trabecolare : immaturo caratterizzato da organizzazione casuale del collagene; con lo sviluppo diviene: osso lamellare : costituisce la maggior parte dello scheletro maturo ed è costituito da strati successivi ; a sua volte si divide in osso compatto e osso spugnoso. • osso compatto (Cortical/Compact) – 80% of adult skeleton –More resistant to bending and torsional forces • osso spugnoso (Trabecular/Cancellous) – 20% adult skeleton –Concentrated where compressive forces predominate Biomateriali - Prof. O. Sbaizero
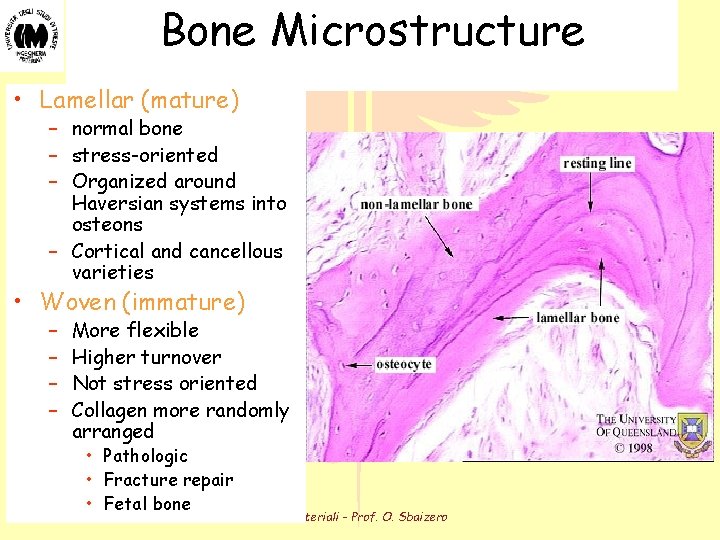
Bone Microstructure • Lamellar (mature) – normal bone – stress-oriented – Organized around Haversian systems into osteons – Cortical and cancellous varieties • Woven (immature) – – More flexible Higher turnover Not stress oriented Collagen more randomly arranged • Pathologic • Fracture repair • Fetal bone Biomateriali - Prof. O. Sbaizero
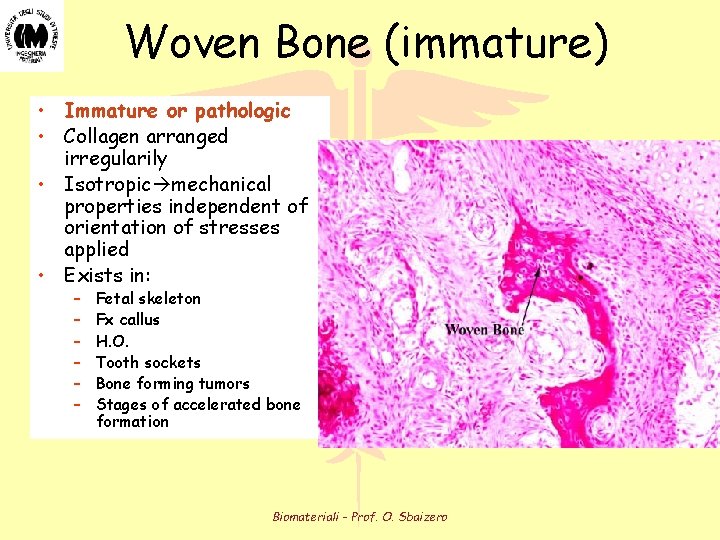
Woven Bone (immature) • Immature or pathologic • Collagen arranged irregularily • Isotropic mechanical properties independent of orientation of stresses applied • Exists in: – – – Fetal skeleton Fx callus H. O. Tooth sockets Bone forming tumors Stages of accelerated bone formation Biomateriali - Prof. O. Sbaizero
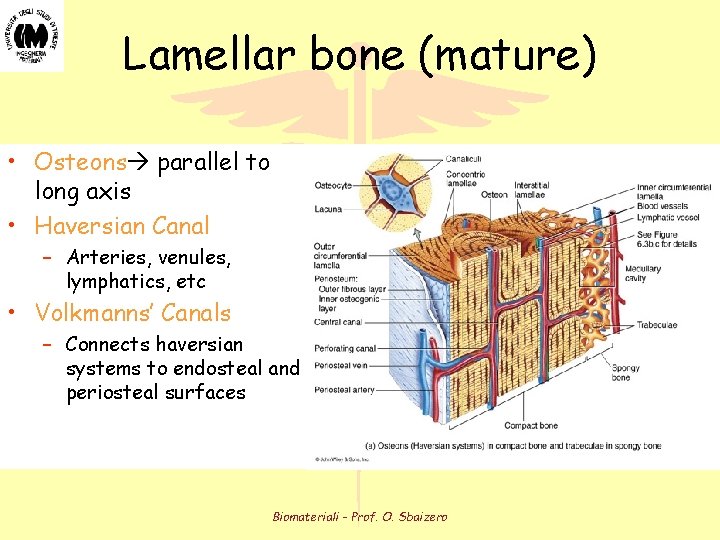
Lamellar bone (mature) • Osteons parallel to long axis • Haversian Canal – Arteries, venules, lymphatics, etc • Volkmanns’ Canals – Connects haversian systems to endosteal and periosteal surfaces Biomateriali - Prof. O. Sbaizero
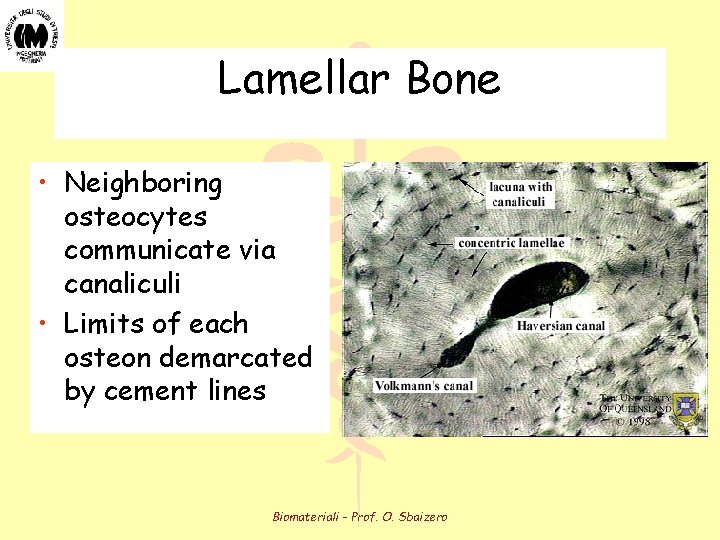
Lamellar Bone • Neighboring osteocytes communicate via canaliculi • Limits of each osteon demarcated by cement lines Biomateriali - Prof. O. Sbaizero
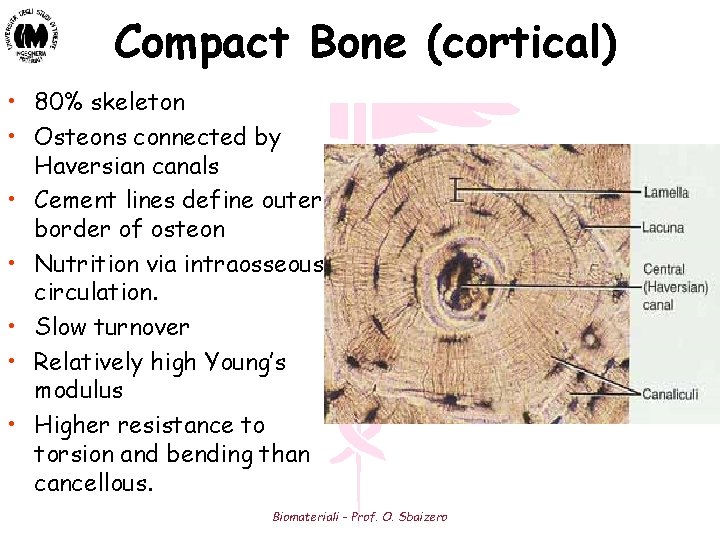
Compact Bone (cortical) • 80% skeleton • Osteons connected by Haversian canals • Cement lines define outer border of osteon • Nutrition via intraosseous circulation. • Slow turnover • Relatively high Young’s modulus • Higher resistance to torsion and bending than cancellous. Biomateriali - Prof. O. Sbaizero
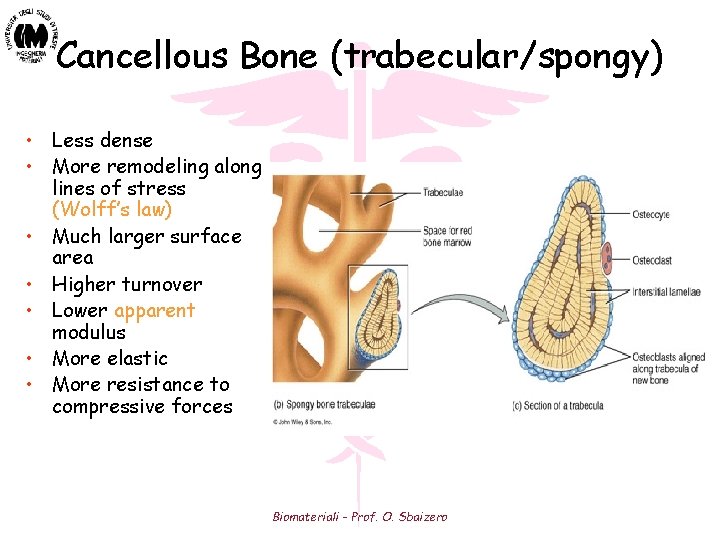
Cancellous Bone (trabecular/spongy) • Less dense • More remodeling along lines of stress (Wolff’s law) • Much larger surface area • Higher turnover • Lower apparent modulus • More elastic • More resistance to compressive forces Biomateriali - Prof. O. Sbaizero

Trabecular Bone • Also known as cancellous bone or spongy bone • Structural matrix of bony plates, beams, and struts with marrow in intervening spaces - Structure of beams and struts depends on location and loading situation - Trabecular struts typically 150 -300 µm in diameter - Form a 3 -D lattice structure with optimized strength to weight ratio in healthy bone • Microstructure of Trabecular Bone - Trabeculae formed from lamellar bone Biomateriali - Prof. O. Sbaizero

Trabecular Bone • Macrostructure of Trabecular Bone - Dependent on anatomic location - Generally develops in response to loading situation at site - Vertebrae Young adults: Vertical plates and horizontal struts Older adults: Vertical beams and horizontal struts - Femoral Neck - Trabeculae form tensile and compressive bands which are directed along the force trajectories experienced by the femoral neck - Femoral-Tibial Joint (Knee) - Trabeculae aligned along direction of long bones, with connecting horizontal struts Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero

Cancellous Bone Biomateriali - Prof. O. Sbaizero
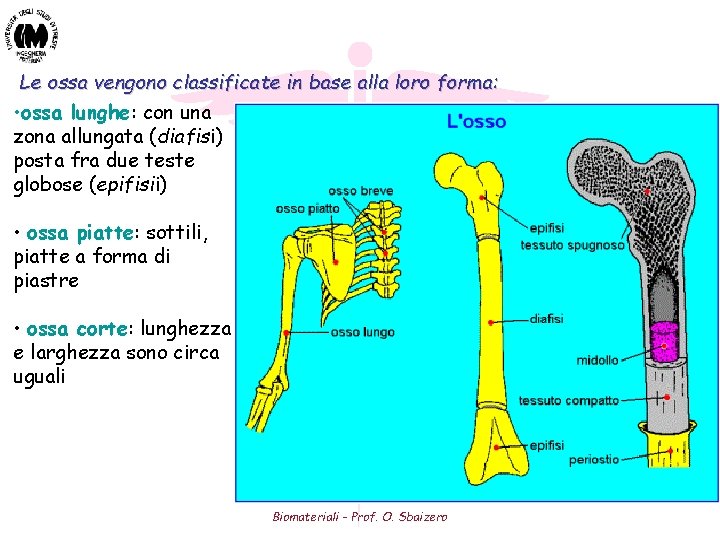
Le ossa vengono classificate in base alla loro forma: • ossa lunghe: con una zona allungata (diafisi) posta fra due teste globose (epifisii) • ossa piatte: sottili, piatte a forma di piastre • ossa corte: lunghezza e larghezza sono circa uguali Biomateriali - Prof. O. Sbaizero

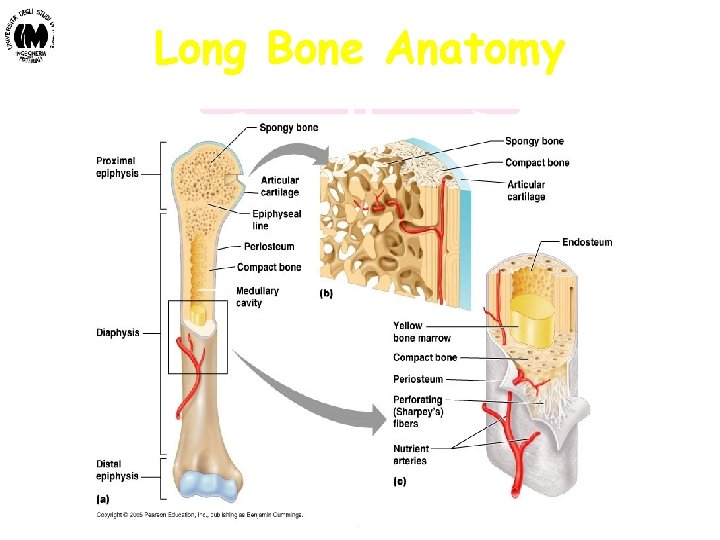
Macrostructure of Bones (1) • Consist of cortical bone, trabecular bone, and marrow • Distribution of each depends on anatomical location • Long bones - Tubular in shape - Length of bone greater than breadth #Some long bones may actually be short - Consist of a shaft and two enlarged, curved ends - Shaft of bone consists of cortical bone surrounding medullary cavity (filled with marrow) - diaphysis - Ends of bone consist of cortical shell of bone surrounding trabecular bone Examples: Femur (thigh) Tibia and fibula (calf) Humerus (upper arm) Radius and ulna (lower arm) Biomateriali - Prof. O. Sbaizero
Long Bone Anatomy Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero

Macrostructure of Bones (2) • After birth, longitudinal growth of long bones occurs in two regions Epiphysis - Highly trabecular region at most proximal and distal ends of long bone Metaphysis - Highly trabecular region at proximal and distal end of diaphysis - The epiphysis and methaphysis are separated by epiphyseal cartilage plate (growth plate) that ossifies when growth has stopped • Throughout life, radial growth can occur at two surfaces Periosteum - Outer surface of bone, through which blood supply reaches the bone Endosteum - Inner surface of bone, in contact with medullary canal - Bone can be deposited or resorbed at these two surfaces - Resorbtion can occur in any region of the bone, typically followed by deposition of bone in the same region -- termed remodeling Biomateriali - Prof. O. Sbaizero

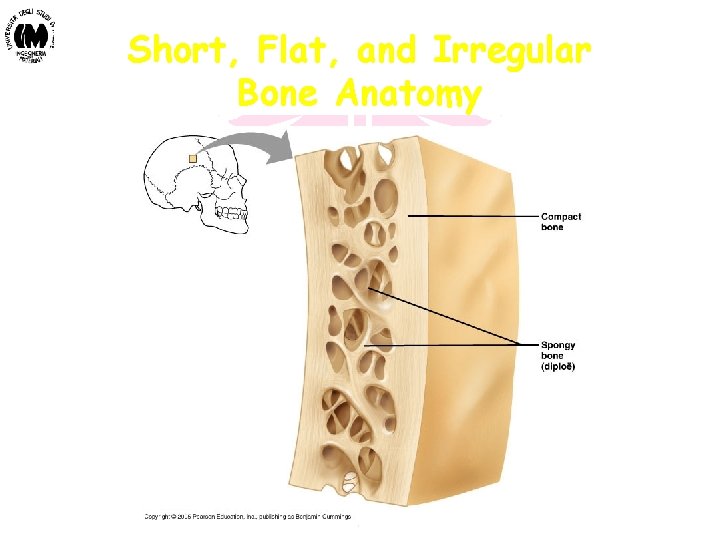
Macrostructure of Bones (3) • Short bones (See Figure 2 A)Cuboidal in shape Consist of cortical shell with inner trabecular core Exist only in the wrist and foot - Carpal and tarsal bones • Flat bones (See Figure 2 B)Consist of two plates of cortical bone with trabecular tissue in between Generally curved rather than flat - Calvaria (top of skull) - Sternum (breast bone) - Scapula (shoulder blade) - Ribs • Irregular bones (See Figure 2 B)Consist of thin cortical shell surrounding trabecular core Various shapes Composition depends on bone - Facial bones - Vertebrae (bones of spine) Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

FIGURE 2 B Biomateriali - Prof. O. Sbaizero
Short, Flat, and Irregular Bone Anatomy Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero
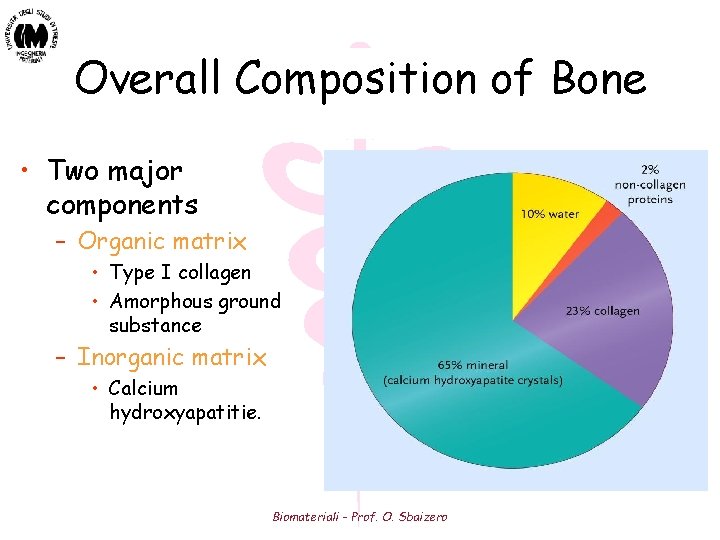
Overall Composition of Bone • Two major components – Organic matrix • Type I collagen • Amorphous ground substance – Inorganic matrix • Calcium hydroxyapatitie. Biomateriali - Prof. O. Sbaizero

Bone Composition - Ultrastructure • Combination of: - Mineral phase (65 wt%): Majority is hydroxyapatite [HA] (calcium phosphate) Also citrate, carbonate, fluoride, and hydroxyl ions - Organic phase (22 wt%) Collagen (90 -96 wt%) Cellular components (osteoclasts, osteoblasts, osteocytes) - Water (9 wt%) - Hydroxyapatite crystals form slender needles in the collagen fiber matrix - The resulting mineral containing fibrils form lamellar sheets Biomateriali - Prof. O. Sbaizero

Organic Matrix • 40% dry weight collagen (90%) proteoglycans non-collagenous matrix proteins growth factors cytokines Biomateriali - Prof. O. Sbaizero

Collagen • 90% organic matrix • Provides tensile strength to bone • Primarily type I • Structure – Triple helix fibril – Hole zones b/w fibrils – Pores b/w sides of parallel molecules • Minerals laid down in holes and gaps – X-linking decreases solubility and increases the tensile strength. Biomateriali - Prof. O. Sbaizero

Proteoglycans • Composed of glycosaminoglycans complexes • Inhibit mineralization • Numerous functions ranging from growth factors to binding properties • Partially responsible for compressive strength of bone. Biomateriali - Prof. O. Sbaizero

Matrix Proteins (non-collagenous) • Promote mineralization and bone formation – Osteocalcin • • • Produced by osteoblasts Directly related to regulation of bone density Most abundant non-collagen matrix protein Inhibited by PTH Activated by 1, 25 Vitamin D Can measure in urine or serum as marker of bone turnover Biomateriali - Prof. O. Sbaizero

Matrix Proteins (non-collagenous) • Osteonectin – Secreted by platelets and osteoblasts – Possible role in regulation of calcium and/or organization of mineral within matrix. • Osteopontin – Cell binding protein Biomateriali - Prof. O. Sbaizero

Growth Factors and Cytokines • Aid in differentiation, activation, growth and turnover of bone – – – TGF-ß IGF IL-1 IL-6 VEGF BMP’s Biomateriali - Prof. O. Sbaizero
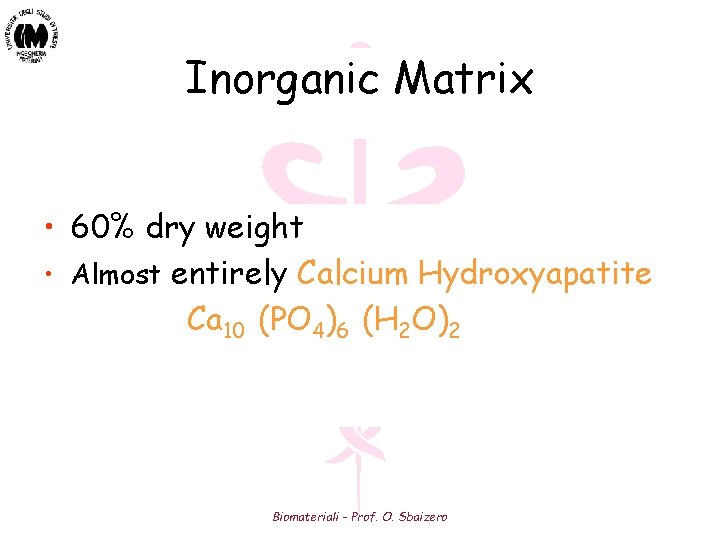
Inorganic Matrix • 60% dry weight • Almost entirely Calcium Hydroxyapatite Ca 10 (PO 4)6 (H 2 O)2 Biomateriali - Prof. O. Sbaizero

Calcium Hydroxyapatite • Provides the compressive strength of bone • Responsible for the mineralization of bone • Primary mineralization occurs in holes and pores Biomateriali - Prof. O. Sbaizero

Marrow • Red marrow – Active in hematopoiesis – Fetal skeleton – As adult only found in flatbones and vertebrae, but long bones can convert if need arises • Yellow marrow – Mainly fat cells most commonly found in adult long bones Biomateriali - Prof. O. Sbaizero
Periosteum • Condensed fibrous tissue outer layer. • Inner cambium layer • Highly vascular • Hold osteoprogenitor cells that differentiate into osteoblasts during growth or repair (in the cambium layer) • Sharpey’s fibers bind periosteum to bone • Very important in Fx repair Biomateriali - Prof. O. Sbaizero

Periosteum • NOT PRESENT at: – Articular surfaces – Sites of tendon/ligament insertions – Surfaces of sesamoid bones – Other specific regions • Subcapular femoral neck Biomateriali - Prof. O. Sbaizero
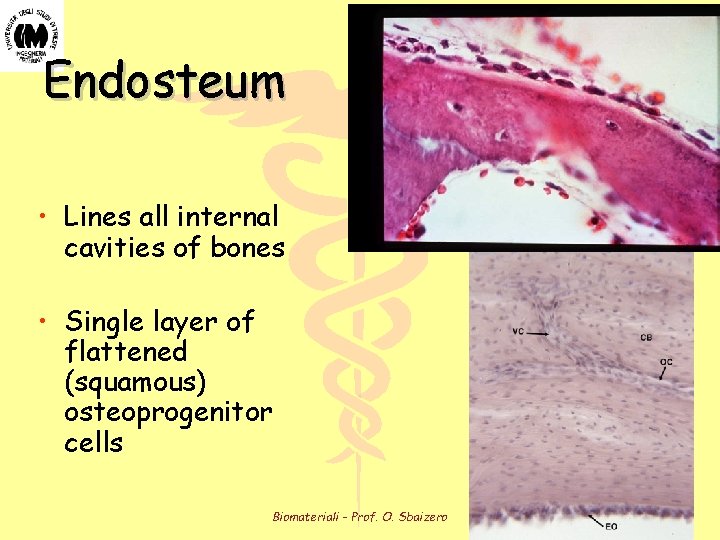
Endosteum • Lines all internal cavities of bones • Single layer of flattened (squamous) osteoprogenitor cells Biomateriali - Prof. O. Sbaizero
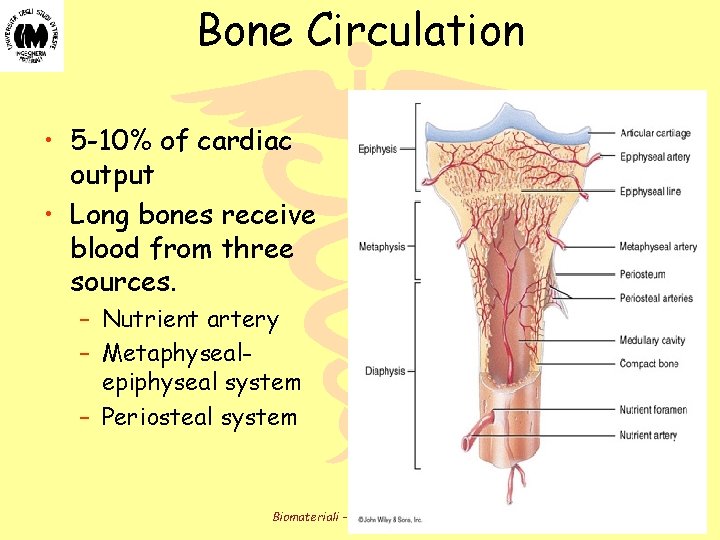
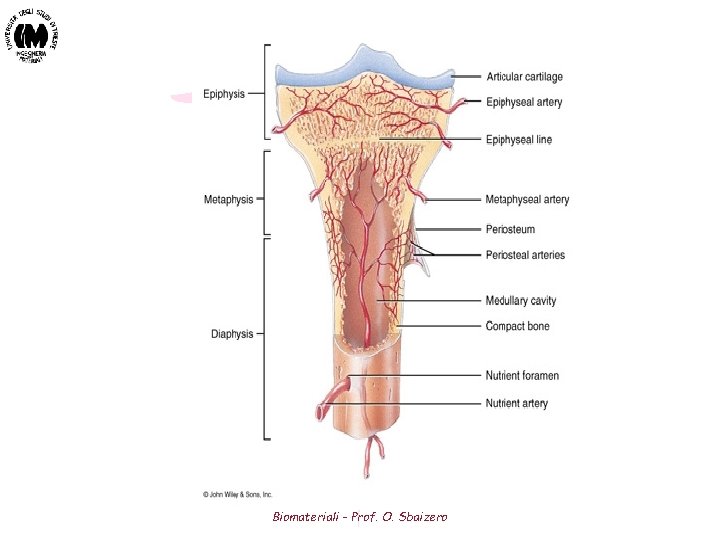
Bone Circulation • 5 -10% of cardiac output • Long bones receive blood from three sources. – Nutrient artery – Metaphysealepiphyseal system – Periosteal system Biomateriali - Prof. O. Sbaizero

STRUTTURA DEI TESSUTI Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

Osservando le curve stressstrain si puo’ dire che: • per piccole deformazioni l’osso e’ lineare, plastico per grandi deformazioni • esse sono funzione della velocita’ di deformazione, infatti sono diverse le pendenze della curva, il punto di snervamento a il punto di rottura • l’osso si irrigidisce ( aumenta il modulo di Young) al crescere della velocita’ di deformazione Biomateriali - Prof. O. Sbaizero

Viscoelastic Properties of Bone • The mechanical properties of both cortical and trabecular bone have been found to vary with strain rate • Young's mod. of trabecular tissue is estimated to have the following relationship with strain rate E = Estatic*(de/dt)0. 06 • Cortical bone has also been seen to exhibit a creep fracture response • Laboratory tests typically conducted at strain rates of 0. 01 to 0. 001 sec-1, quasi-static Biomateriali - Prof. O. Sbaizero

Viscoelastic Model of Bone Properties • Impact injuries (falls, vehicular accidents) typically involve strain rates 10 sec -1 - At higher strain rates, bone has a higher ultimate strength but fractures at a lower strain • The viscoelastic properties of bone are most likely due to a combination of: • Simplifed model is taken to be a three element spring/dashpot model (Figure 9) • Bone thus behaves in the following manner (at relatively low strain rates): - Undergoes initial, immediate deformation with application of stress - Deformation will continue to an asymptotic limit if stress is maintained - When s is removed, initial deformation will be regained (within elastic limits) and additional viscous deformation will gradually be regained (Figure 10) Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero
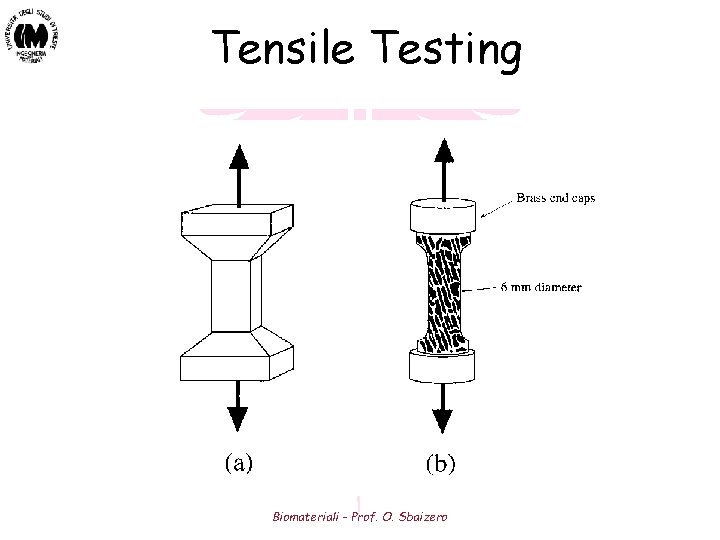
Tensile Testing Biomateriali - Prof. O. Sbaizero
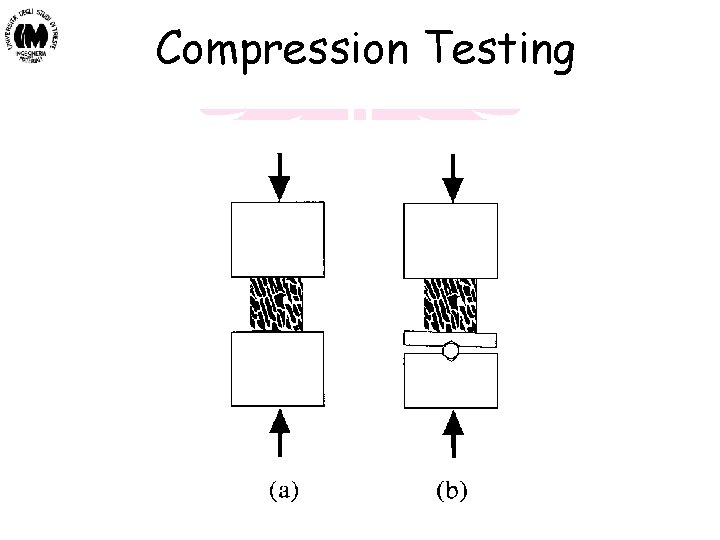
Compression Testing Biomateriali - Prof. O. Sbaizero
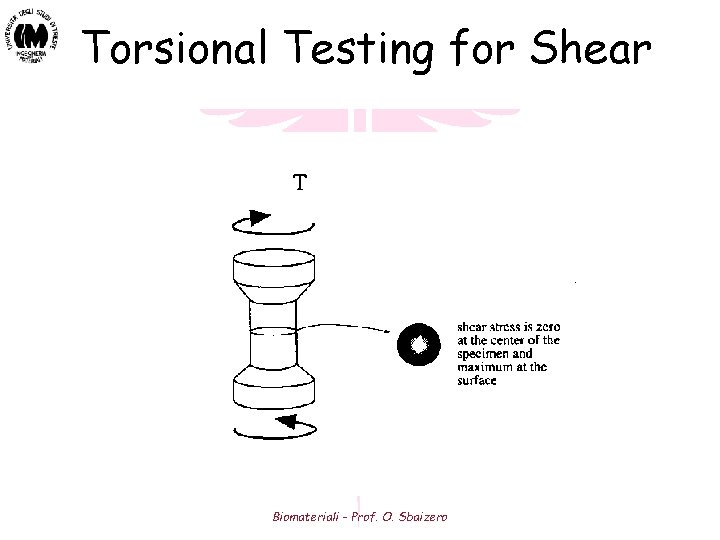
Torsional Testing for Shear Biomateriali - Prof. O. Sbaizero
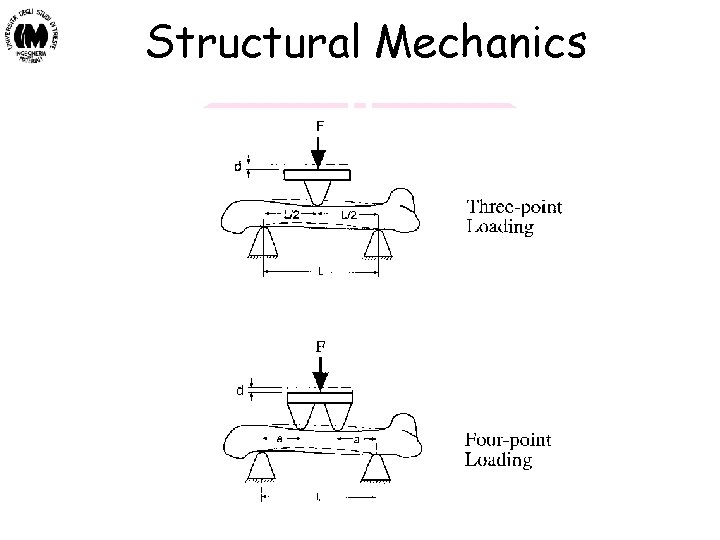
Structural Mechanics Biomateriali - Prof. O. Sbaizero

Other testing methods • Indentation • Nanoindentation • Ultrasound Biomateriali - Prof. O. Sbaizero

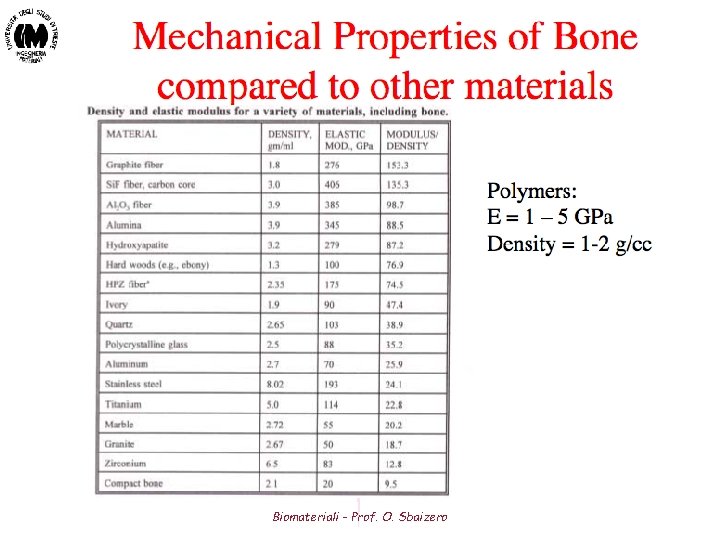
Material Properties of Hard Tissues (1) Biomateriali - Prof. O. Sbaizero
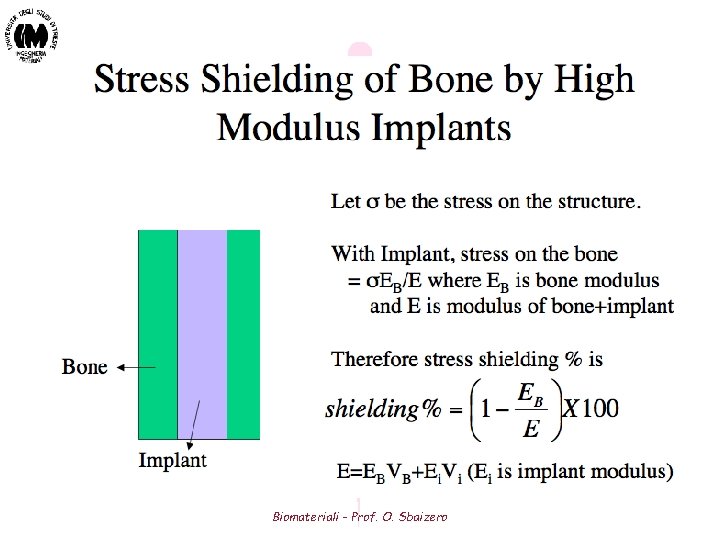
Biomateriali - Prof. O. Sbaizero

Structural Properties of Hard Tissues • Structural properties include - Mechanical properties of tissue - Structural density (or apparent density) - Equal to the mass of the bony material per unit volume of tissue (including marrow or marrow space) - Measures of trabecular structure as listed above • Material and structural properties of blocks of cortical bone, dentin, and enamel tissue considered equal due to compact structure • Material and structural properties of trabecular bone differ significantly - Material properties are defined for trabeculae alone - Structural properties are defined for tissue including bone and marrow - Trabecular bone is anisotropic, with degree of anisotropy varying between anatomic location - Properties (no density) are thus directionally dependent - Properties vary greatly between individuals and anatomic locations Biomateriali - Prof. O. Sbaizero

Material Properties of Hard Tissues (2) • Properties of tissues vary between individuals and with anatomic location • Material properties are significantly affected by the degree of mineralization (and therefore density) of the bone - The density of bone is not an intrinsic property and can change with time Mineral stores within the bone can change based on physiological demand Pathological processes can affect the degree of mineralization - In normal, healthy bone, the range of bone mineralization is small • Cortical bone - Cortical bone is anisotropic Typically modelled as orthotropic or transversely isotropic Longitudinal direction (along axis of osteons) has highest mechanical properties Weaker and lower modulus in radial and transverse directions - Cortical bone is weaker in tension and shear than in compression • Trabecular bone - Continuing debate over whether trabecular bone is merely porous cortical bone or whether an intrinsic material difference exists in the bony material Trabecular material properties are less than those of cortical bone Difficult to test individual trabeculae to determine conclusively Biomateriali - Prof. O. Sbaizero

Mechanical Properties of Trabecular Tissue Measured mechanical properties for trabecular tissue are in the following range Young's modulus: Elastic Modulus of trabecular bone ranges between 1 and 150 MPa Compressive strength: Compressive Strength of trabecular Bone ranges between 2 and 19 MPa • For trabecular tissue, compressive strength has been foundto be linearly related to Young's modulus • Young's mod. , for a given direction of testing, can be related to the density (ra) of the material by a power-law relationship with n ranging from about 1 to 3 depending on the trabecular structure E = b + m * ra n Biomateriali - Prof. O. Sbaizero

Contribution of Components to Whole Bone Strength • The contribution of the trabecular and cortical components of whole bone to overall structural strength will vary with anatomical location - Variable amounts of cortical and trabecular bone are present at every site • In highly trabecular sites, the trabecular bone can be presumed to provide a substantial portion of the mechanical integrity of the structure - In the femoral neck, bending strength was reduced by approximately 40% when the trabecular bone was removed - In vertebral bodies, removal of the cortical shell was determined to reduce the compressive strength by about 10 percent - In both cases, the contribution of trabecular bone to overall strength is greater than what would be proportional to its mass or mineral density Biomateriali - Prof. O. Sbaizero

Bone as a Composite Material - Model 1 • Compound bar with isostrain condition - Bone acts as reinforced bar with collagen matrix and oriented HA fibres - In normally mineralized bone, HA and collagen occupy about equal amounts of load-bearing volume (the water is not a load-bearing component) - Similar to a reinforced concrete beam (Figure 11) - If the components are assumed to undergo the same strain, then the fraction of the load borne by each when the fibres are alligned with the loading direction can be estimated by: Total load is the sum of loads on each component Pt = P m + P c s = P/A = E e A is cross-sectional area, E is modulus Pm = Am* Em* em and Pc = Ac* Ec*ec Since em = ec Pc = Pm *(Ac* Ec/Am* Em) Therefore Pm = Pt*Am* Em/(Am* Em + Ac * Ec ) and Pc = Pt*Ac* Ec/(Am* Em + Ac* Ec) This model is likely to overestimate the load borne by the HA "fibres" or crystals Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

Bone as a Composite Material - Model 2 • Two-phase model - Bone acts a two-phase composite similar to fibreglass • This type of material typically has a modulus intermediate to the two components but a higher strength than either component tested separately • Fracture will occur if a crack, typically initiated in the high modulus material, is able to propagate and run through the whole structure • HA crystals are so small that the occurrence of random flaws is also small • When cracks do occur they quickly enter the more ductile collagen phase and energy is dissipated in deformation instead of crack propagation • Calculation of load borne by each phase is more complicated in this model due to the possiblity of multiple orientations of the collagen fibre/HA crystal compound within the structure Biomateriali - Prof. O. Sbaizero

Fatigue of Bone • Fatique microdamage occurs in bone just as it does in other materials under cyclic loading • Bone undergoes cyclic loading throughout life, with frequency, duration, and load varying with time and between individuals • Fatigue microdamage reduces a bone's elastic modulus • The benefit of bone as a living structure is that it can repair microdamage before it progresses to failure - Some pathological processes interfere with this repair mechanism which may be one cause of increased fracture occurance - Stress fractures are most common, in healthy individuals, in cases of both increased loading and increased number of cycles over normal use and typically occur in a matter of hours - Example: Forced marches in boot camp Biomateriali - Prof. O. Sbaizero

Mechanical Properties of Whole Bones The load at failure of a whole bone will depend on: Geometry - bone size and shape Distribution of cortical and trabecular bone Mechanical properties of cortical and trabecular bone Loading rate Mode of loading (compression, tension, shear) Direcion of loading and point of application • Failure generally occurs due to tensile or shear loading (Fig 12) - There is a always a bending moment on bones loaded physiologically inducing both compressive and tensile forces - Failure in bending begins on the tensile surface of the bone - Failure due to shear results in a spiral fracture of the bone • Exception is crush fractures (at sites like the vertebrae) - Failure is actually a result of successive bending-induced fractures of the trabeculae and finally bending of the cortical shell • Failure of bone in vivo under impact is also dependent on the energy absorbed by the surrounding soft tissue Biomateriali - Prof. O. Sbaizero

FIGURE 12 -1 Biomateriali - Prof. O. Sbaizero

FIGURE 12 -2 Spiral fractures result from bone which fails in shear. Biomateriali - Prof. O. Sbaizero

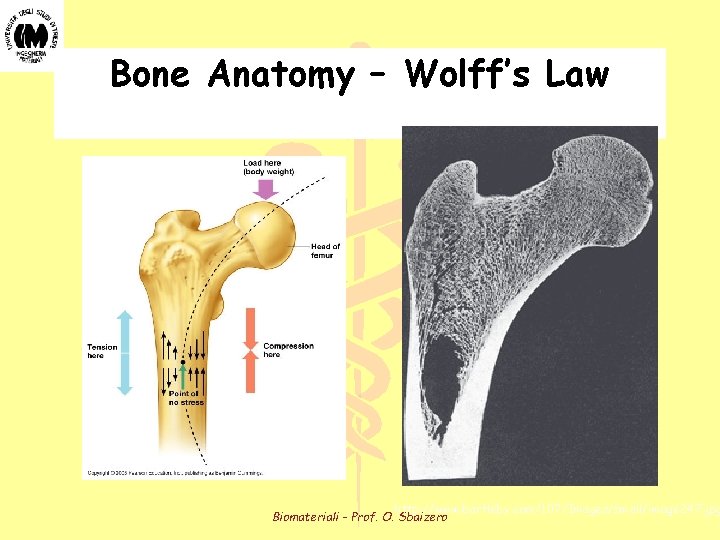
ll rimodellamento osseo E’ un fenomeno per il quale l’osso e’ in grado di ottimizzare la sua forma in funzione del carico che deve sopportare Quindi grandi capacita’ adattative in risposta alle sollecitazioni meccaniche La fenomenologia del rimodellamento osseo va sotto il nome di legge di Wolff che si riassume: • il rimodellamento osseo e’ governato da sollecitazioni flessionali, non dagli sforzi principali • il rimodellamento osseo e’ stimolato da carichi dinamici ciclici, non da carichi statici • la flessione dinamica produce una crescita ossea nella zone in cui la flessione causa la concavita’ Biomateriali - Prof. O. Sbaizero

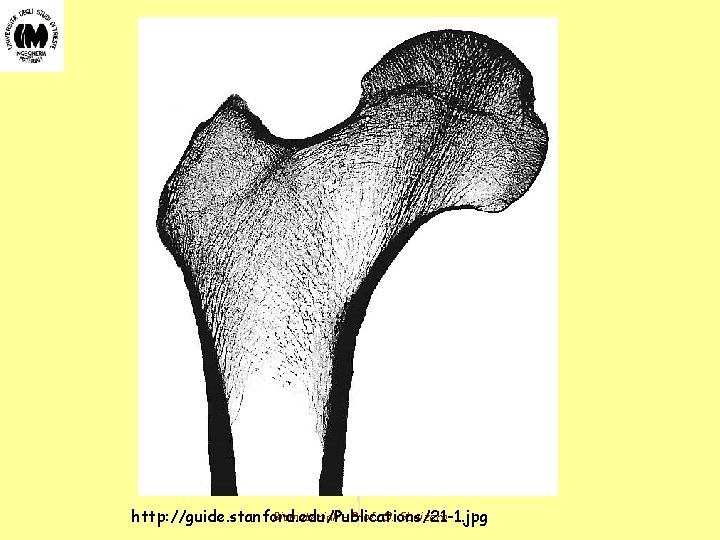
Wolff's Law (1) • Bone is a living tissue which is constantly undergoing modeling and remodeling • In 1892, Wolff presented the first evidence that bone remodels in response to mechanical forces acting upon it - The trabecular bone structure of the femoral neck follows the principal stress trajectories at that location (See Figure 13) • Further evidence that bone remodeling is stress or strain dependent - Bone is lost as a result of extended immobilization or time in a zero-gravity environment - Centrifuge, hypergravity experiments on rats resulted in increased bone mass - A bone graft from the tibia used to replace a metacarpal remodels to resemble the original bone structure - Bone in the anterior and posterior aspects of the femur is weaker and less stiff than that of the medial and lateral aspects - Corresponds to amount of stress at each location due to natural bending - In pigs, the increased strain on the radius noted after removal of the ulna was gradually reduced to normal levels by an increase in radial diameter Biomateriali - Prof. O. Sbaizero
Bone Anatomy – Wolff’s Law http: //www. bartleby. com/107/Images/small/image 247. jpg Biomateriali - Prof. O. Sbaizero
Biomateriali - Prof. O. Sbaizero http: //guide. stanford. edu/Publications/21 -1. jpg

Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

Biomateriali - Prof. O. Sbaizero

Wolff's Law (2) • There is continued debate on two general areas of this phenomena #1: What is the driving force behind remodeling? #2: What is the mechanism by which mechanical data is transferred to the bone cells to produce remodeling? Hypotheses on point #1: - Time averaged strain over a threshold limit acts as the stimulus for bone remod, with a goal of maintaining an "ideal" level of strain at every location - Disruption of communication channels (through canaliculi) between the periostem and the internal osteocytes result in bone hypertrophy - Fatigue induced micro-cracks are necessary to trigger any bone remodeling which will result in a change of bone mass - Increased strain rate, which often parallels increases in strain levels, is the stimulus behind increased bone deposition Biomateriali - Prof. O. Sbaizero

Wolff's Law (3) Hypotheses on point #2: - Bone is a piezolectric material which generates electrical signals in response to (dynamic) mechanical loading. These electrical signals then trigger the bone cells to control remodeling. - Mechanical deformation results in changes in fluid flow within bone canals, the fluid flow stimulates bone cells (osteocytes or osteoblasts) : (1) Changes in the fluid-induced shear stress observed by cells (2) Changes in the hydrostatic pressure observed by the cells (3) Changes in cell deformation/strain caused by fluid flow past cells (4) Electrokinetic phenomena (streaming potential) induced when fluid flows past the charged surface of bone and bone cells - These mechanisms may act individually, or most likely cooperatively, to induce a remodeling response in bone cells Biomateriali - Prof. O. Sbaizero

Wolff's Law (4) • General phenomenological theories regarding bone remodeling - The objective of bone remodeling, with changes in bone mass, is to minimize the flexural deformity of the bone through a drift in the mass of bone towards the concavity created by the bending. (Fig 12) - Cyclic loading is required to induce bone apposition both to maintain and to increase bone mass. • A number of bone remodeling theories have been included in finite element mod. in an attempt to see which produces the most natural type of bone remodeling • Very possibly a combination of mechanisms which result in the overall remodeling of bone in response to mechanical loading (or lack thereof) The "ideal" level of s or e must be site dependent to allow for the fact that minimally loaded bones, such as the calvaria (top of skull) and ossicles (inner ear bones) do not atrophy In any case, the necessary strain to maintain bone is small Maintenance - 1000 to 3000 microstrain (e = 0. 001 to 0. 003) Biomateriali - Prof. O. Sbaizero
Bone Remodeling: Osteoporosis Biomateriali - Prof. O. Sbaizero

Teeth • Multiphasic structure consisting of 4 basic materials plus the gingiva (gums) and alveolar bone of the jaw (See Figure 7) Enamel: - 97% calcium phosphate salts in the form of large HA crystals - Hardest substance in the body Dentin: - Similar distribution of collagen and matrix to cortical bone - Properties similar to cortical bone Pulp: - Marrow-like substance through which blood supply is provided to dentin also contains nerve cells and thin collagenous fibres Cementum: - Coarsely fibred bone-like substance that does not contain canaliculi, Haversian systems, or blood vessels Biomateriali - Prof. O. Sbaizero
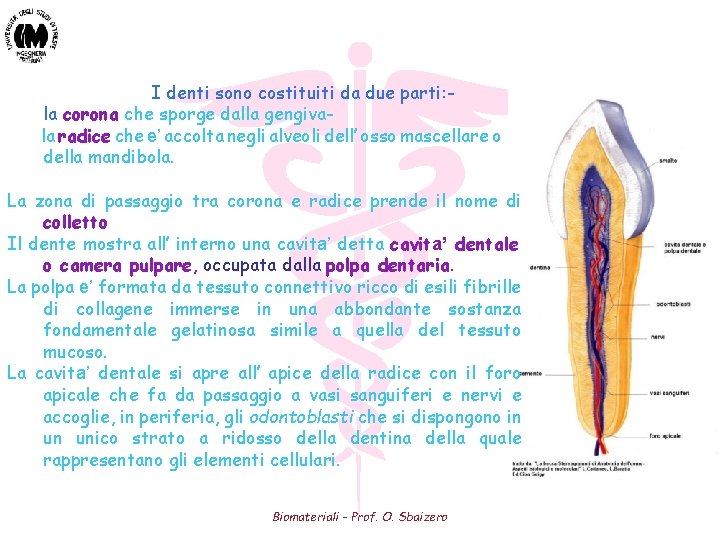
I denti sono costituiti da due parti: la corona che sporge dalla gengivala radice che e’ accolta negli alveoli dell’ osso mascellare o della mandibola. La zona di passaggio tra corona e radice prende il nome di colletto Il dente mostra all’ interno una cavita’ detta cavita’ dentale o camera pulpare, occupata dalla polpa dentaria. La polpa e’ formata da tessuto connettivo ricco di esili fibrille di collagene immerse in una abbondante sostanza fondamentale gelatinosa simile a quella del tessuto mucoso. La cavita’ dentale si apre all’ apice della radice con il foro apicale che fa da passaggio a vasi sanguiferi e nervi e accoglie, in periferia, gli odontoblasti che si dispongono in un unico strato a ridosso della dentina della quale rappresentano gli elementi cellulari. Biomateriali - Prof. O. Sbaizero
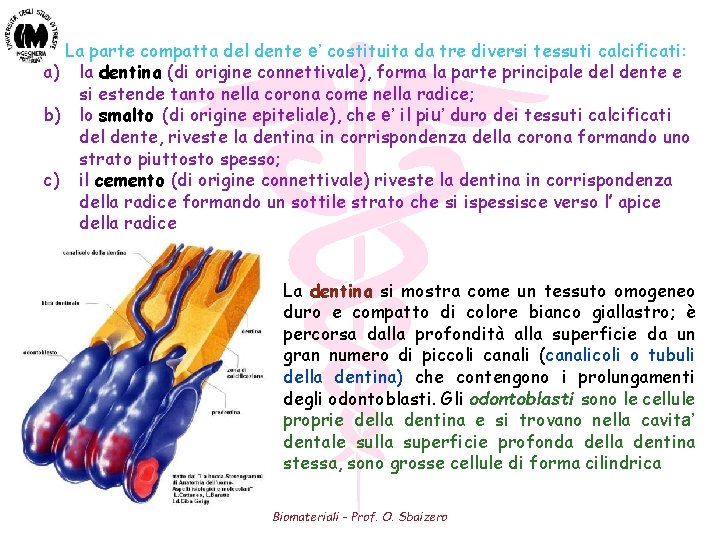
La parte compatta del dente e’ costituita da tre diversi tessuti calcificati: a) la dentina (di origine connettivale), forma la parte principale del dente e si estende tanto nella corona come nella radice; b) lo smalto (di origine epiteliale), che e’ il piu’ duro dei tessuti calcificati del dente, riveste la dentina in corrispondenza della corona formando uno strato piuttosto spesso; c) il cemento (di origine connettivale) riveste la dentina in corrispondenza della radice formando un sottile strato che si ispessisce verso l’ apice della radice La dentina si mostra come un tessuto omogeneo duro e compatto di colore bianco giallastro; è percorsa dalla profondità alla superficie da un gran numero di piccoli canali (canalicoli o tubuli della dentina) che contengono i prolungamenti degli odontoblasti. Gli odontoblasti sono le cellule proprie della dentina e si trovano nella cavita’ dentale sulla superficie profonda della dentina stessa, sono grosse cellule di forma cilindrica Biomateriali - Prof. O. Sbaizero
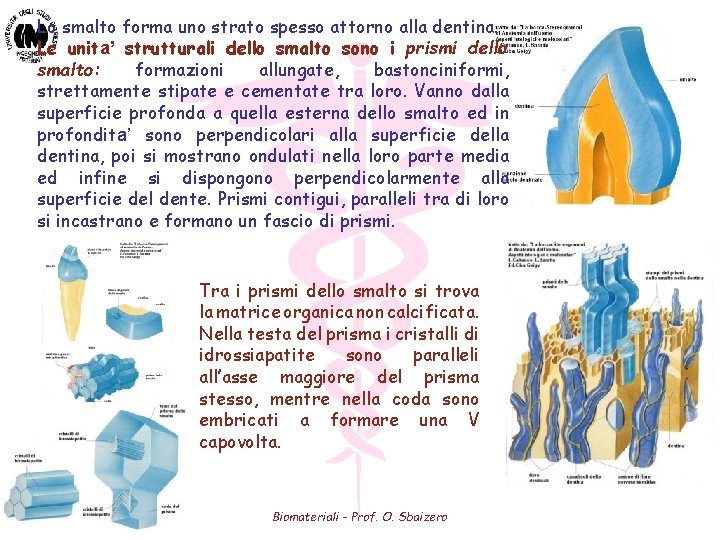
Lo smalto forma uno strato spesso attorno alla dentina. Le unita’ strutturali dello smalto sono i prismi dello smalto: formazioni allungate, bastonciniformi, strettamente stipate e cementate tra loro. Vanno dalla superficie profonda a quella esterna dello smalto ed in profondita’ sono perpendicolari alla superficie della dentina, poi si mostrano ondulati nella loro parte media ed infine si dispongono perpendicolarmente alla superficie del dente. Prismi contigui, paralleli tra di loro si incastrano e formano un fascio di prismi. Tra i prismi dello smalto si trova la matrice organica non calcificata. Nella testa del prisma i cristalli di idrossiapatite sono paralleli all’asse maggiore del prisma stesso, mentre nella coda sono embricati a formare una V capovolta. Biomateriali - Prof. O. Sbaizero
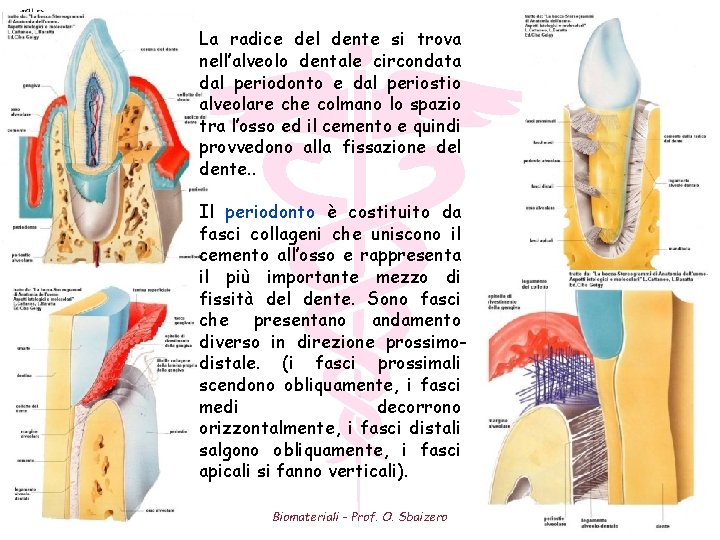
La radice del dente si trova nell’alveolo dentale circondata dal periodonto e dal periostio alveolare che colmano lo spazio tra l’osso ed il cemento e quindi provvedono alla fissazione del dente. . Il periodonto è costituito da fasci collageni che uniscono il cemento all’osso e rappresenta il più importante mezzo di fissità del dente. Sono fasci che presentano andamento diverso in direzione prossimodistale. (i fasci prossimali scendono obliquamente, i fasci medi decorrono orizzontalmente, i fasci distali salgono obliquamente, i fasci apicali si fanno verticali). Biomateriali - Prof. O. Sbaizero
- Slides: 96