Mineral Spectroscopy Visible Infrared Mssbauer Raman NMR Properties

Mineral Spectroscopy Visible Infrared Mössbauer Raman NMR

Properties of Light • Light is conducted through materials on the valence electrons. • Light travels more slowly in materials. • Electrically conducting materials are opaque

Electromagnetic Spectrum Visible Light: 7700 - 3900Å

Properties of Light • Light is conducted through materials on the valence electrons. • Light travels more slowly in materials. • Electrically conducting materials are opaque.

Behavior of Light in Materials • Absorption (light is absorbed by materials) • Color (absorption is a function of wavelength) • Pleochroism (absorption is a function of direction) • Refraction (light travels slowly in some materials) • Dispersion (velocity is a function of wavelength) • Birefringence (velocity is a function of direction)

Absorption • • • Light is attenuated on entering any material. The attenuation is a function of distance. I is intensity at some point t I 0 is initial intensity. k is absorption coefficient in cm-1. Lambert’s Law:
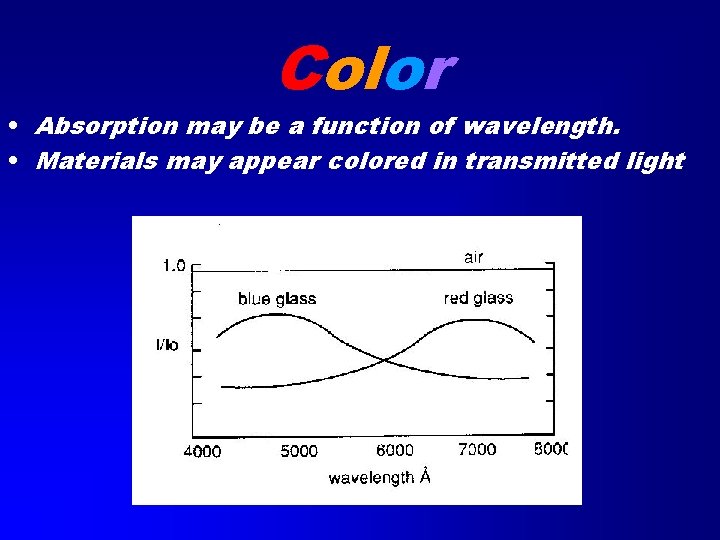
Color • Absorption may be a function of wavelength. • Materials may appear colored in transmitted light
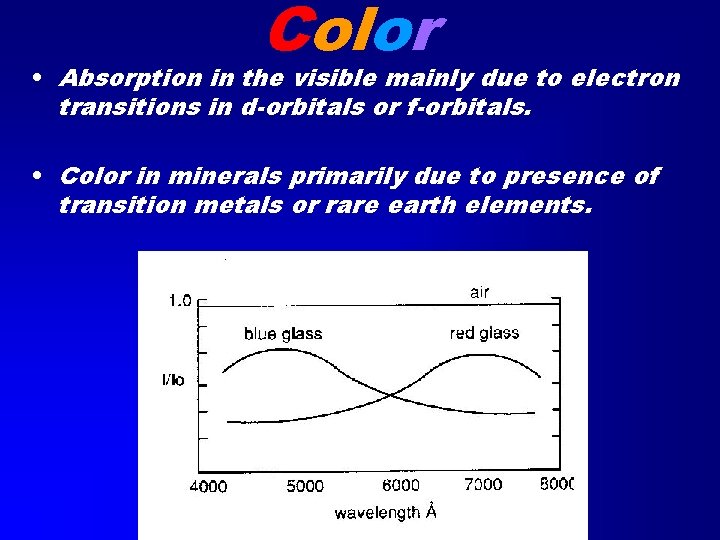
Color • Absorption in the visible mainly due to electron transitions in d-orbitals or f-orbitals. • Color in minerals primarily due to presence of transition metals or rare earth elements.
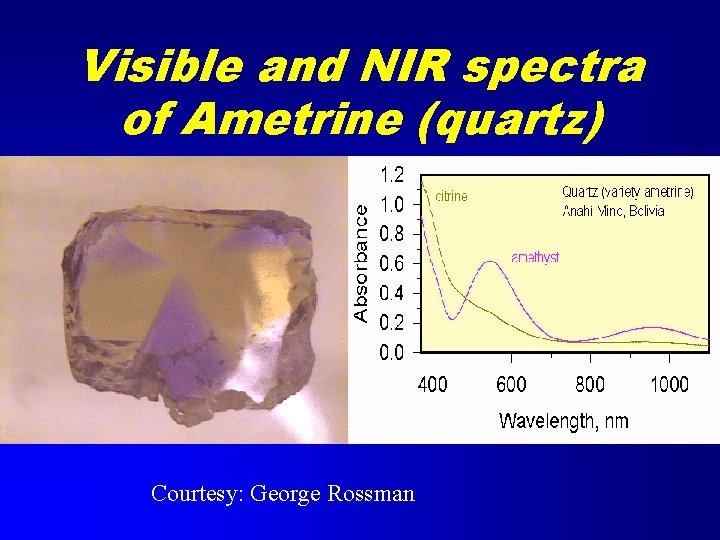
Visible and NIR spectra of Ametrine (quartz) Courtesy: George Rossman

Ringwoodite is Blue • (g-Mg 1. 63 Fe 0. 22 H 0. 4 Si 0. 95 O 4) • ~10 % of Fe present as ferric (Mössbauer)

Pleochroism • Pleochroism is the variation of absorption with direction in a crystal. • Pleochroism is observed as a color change on rotation in plane-polarized light (not crossed polars). • Pleochroism only occurs in non-cubic crystals. • Pleochroism indicates the presence of transition metals (esp Fe, also Mn, Cr, V, etc). • Biotite, tourmaline, amphibole.

Refraction and Reflection • When light strikes a polished surface of a material it is split into two rays. • One is reflected and the other refracted

Infrared spectroscopy • Near IR 5000 - 13000 cm-1 – orbital transitions • Mid-IR 2500 - 5000 cm-1 – N-H and O-H bond vibrations • Far IR 500 - 2500 cm-1 – Cation-Oxygen bond vibrations – Structural phonons.
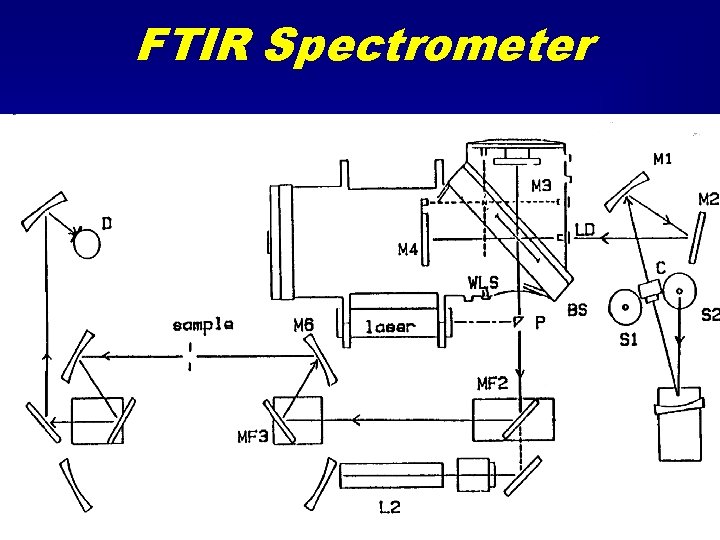
FTIR Spectrometer
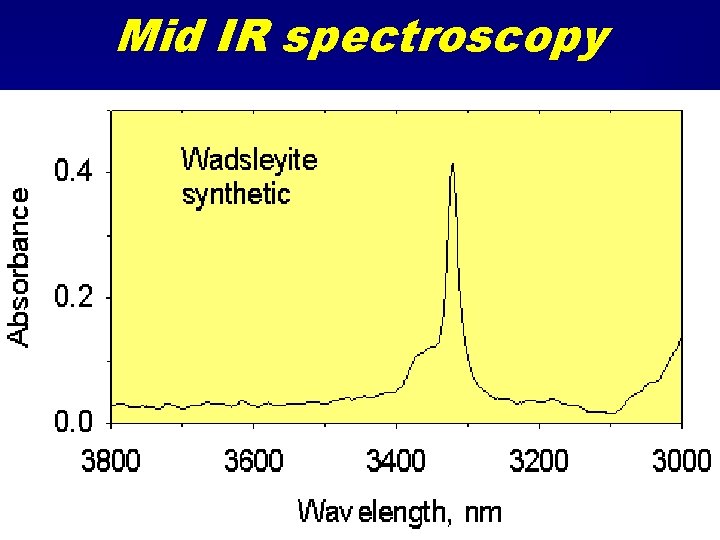
Mid IR spectroscopy
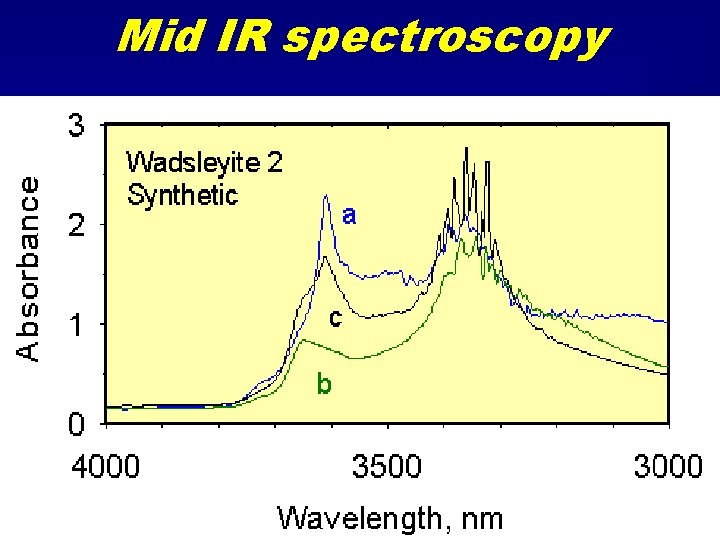
Mid IR spectroscopy

Raman Spectroscopy • Looks at wavelength shifts in scattered light. • Shifts are in atomic vibrational part of spectrum • 0 - 5000 cm-1. (same as mid to far IR) • Excitation is usually by a monochromatic source in the visible region (commonly a laser).
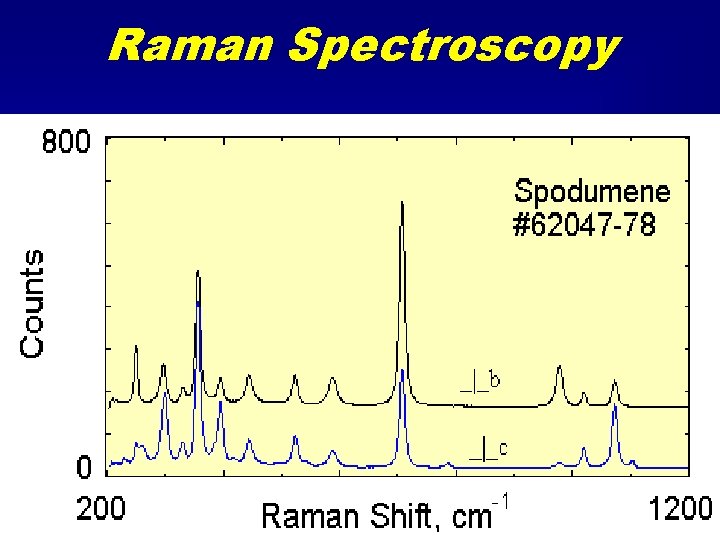
Raman Spectroscopy

Mössbauer Spectroscopy Resonant Gamma Ray spectroscopy Uses 57 Fe gamma decay at 14. 4 Me. V Source is 57 Co Source is accelerated mechanically to produce ultra-fine relativistic energy shifts • Absorption as a function of source velocity • Looks at electric field effects at nucleus due to d-orbital occupancy and perturbations from local coordination effects • •
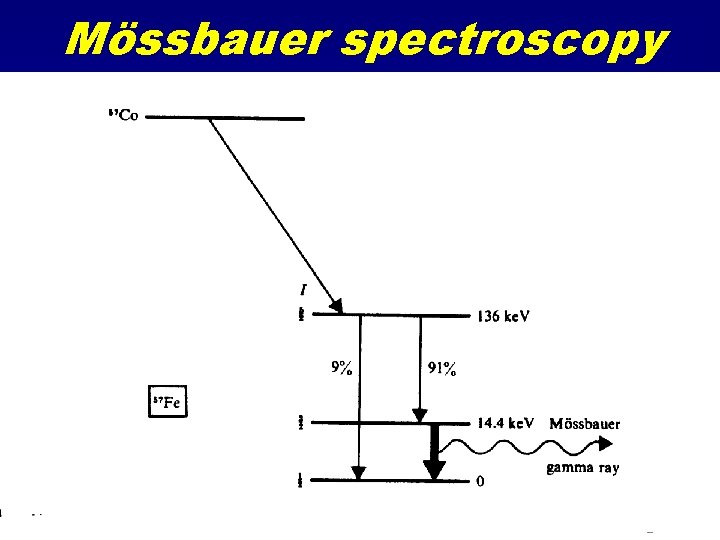
Mössbauer spectroscopy
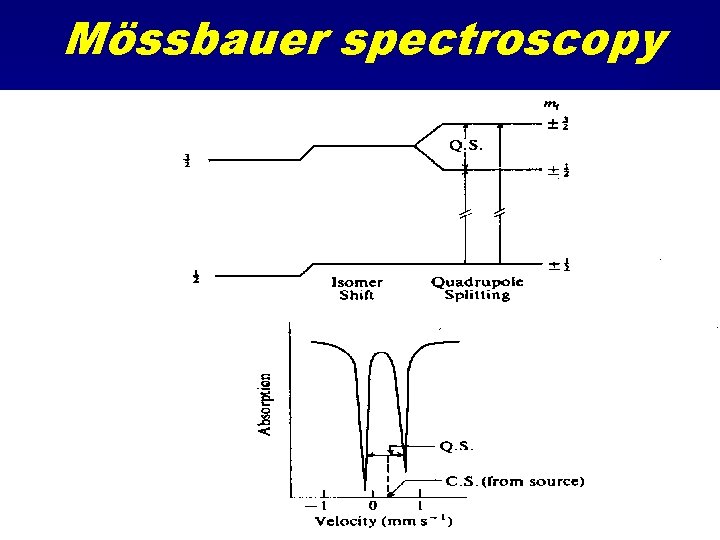
Mössbauer spectroscopy
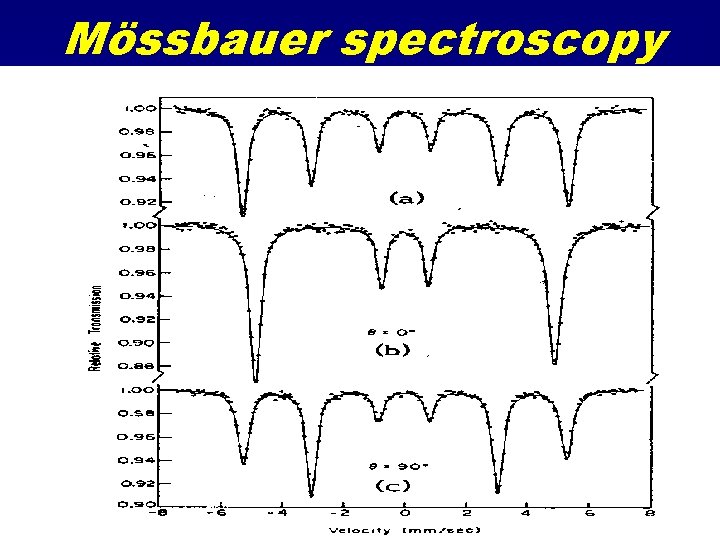
Mössbauer spectroscopy

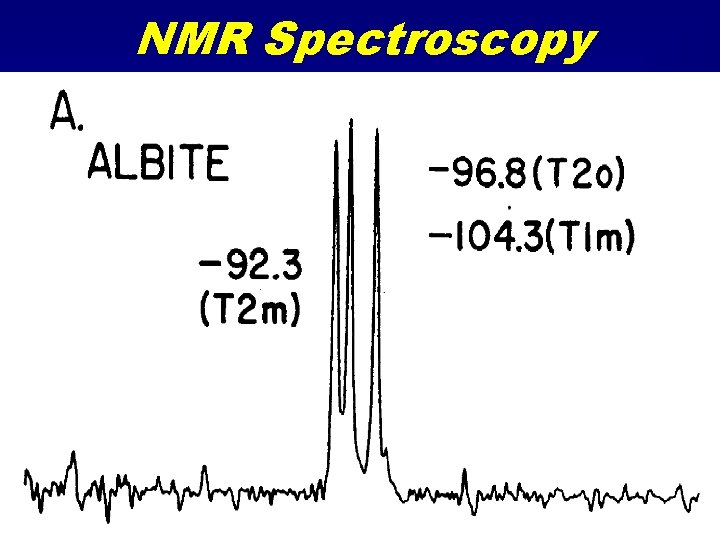
NMR Spectroscopy • Nuclear Magnetic Resonance • Similar to Mössbauer spectroscopy but many more nuclides • Radio frequency emission spectroscopy due to magnetic transitions in nucleus. • Solid samples are spun in a strong magnetic field (Magic Angle Spinning) • A RF field applied and turned off. • Sample emits RF
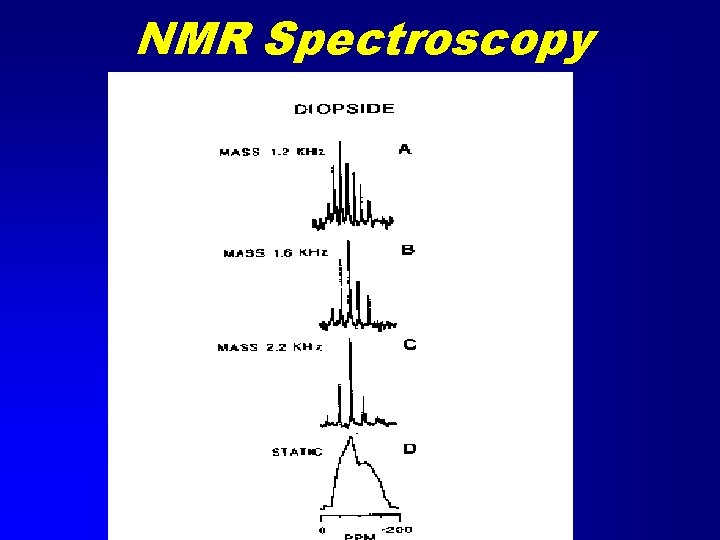
NMR Spectroscopy
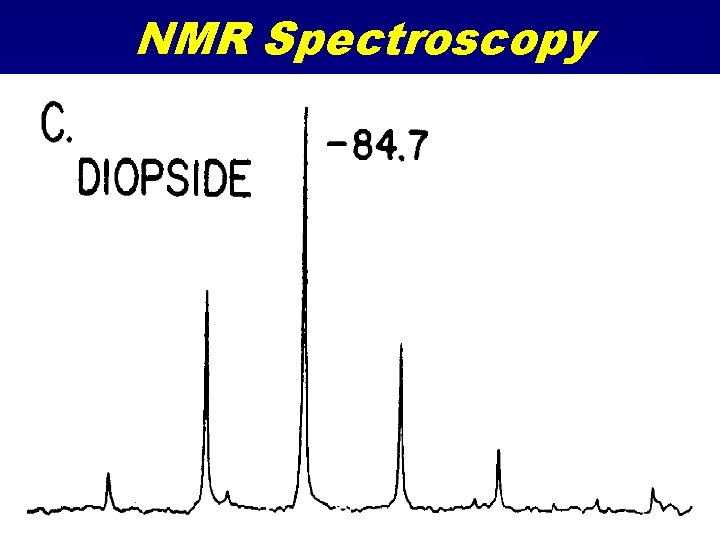
NMR Spectroscopy
NMR Spectroscopy
- Slides: 26