Mineral resources are Minerals or rocks mined from
























- Slides: 28

Mineral resources are: • Minerals or rocks mined from the earth and used in the products we use daily. • Coal, oil, and natural gas are also mined, but these energy resources will be considered separately.

The use of minerals and rocks in products • Sometimes actual minerals and rocks are used in products or to make things. The rock granite is mined to make countertops, and the mineral halite is mined, crushed, and sold as table salt. • Other times, minerals and brines are processed to extract one specific element, and these individual elements are often called mineral commodities. For example, the commodity aluminum is extracted from the rock bauxite, which contains aluminum-bearing minerals like gibbsite. • The process of extracting the desired mineral or element from an ore is called beneficiation.

Minerals are: • solid • inorganic (or identical to an inorganic mineral) • natural (or made in a way that mimics nature) • chemically homogeneous (that is, the mineral contains the same chemicals throughout) • crystalline (the atoms in a mineral are arranged in an orderly and repeating pattern)

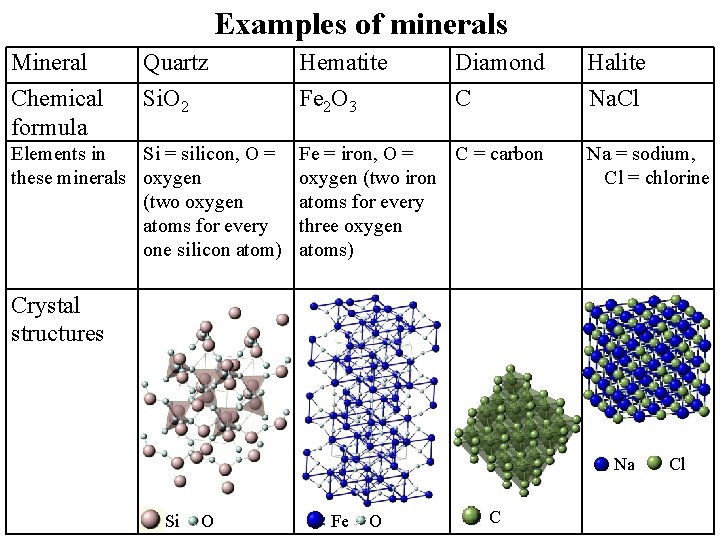
Examples of minerals Mineral Chemical formula Quartz Si. O 2 Hematite Fe 2 O 3 Diamond C Elements in Si = silicon, O = Fe = iron, O = C = carbon these minerals oxygen (two iron (two oxygen atoms for every three oxygen one silicon atom) atoms) Crystal structures Halite Na. Cl Na = sodium, Cl = chlorine Na Si O Fe O C Cl

NOT minerals The "minerals" in these “vitamins and minerals” are not real minerals. These are elements that may have been extracted from minerals.

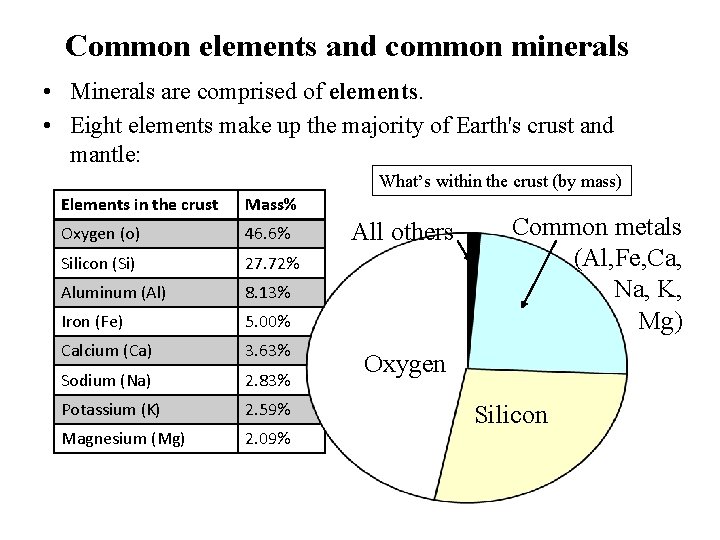
Common elements and common minerals • Minerals are comprised of elements. • Eight elements make up the majority of Earth's crust and mantle: What’s within the crust (by mass) Elements in the crust Mass% Oxygen (o) 46. 6% Silicon (Si) 27. 72% Aluminum (Al) 8. 13% Iron (Fe) 5. 00% Calcium (Ca) 3. 63% Sodium (Na) 2. 83% Potassium (K) 2. 59% Magnesium (Mg) 2. 09% All others Common metals (Al, Fe, Ca, Na, K, Mg) Oxygen Silicon

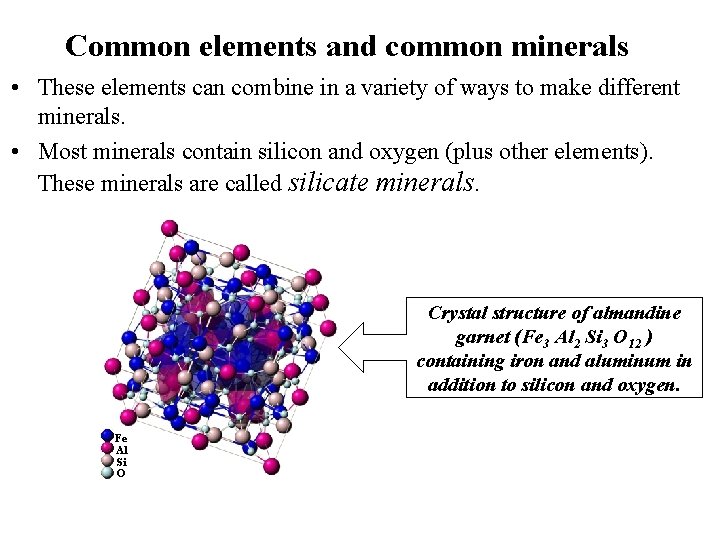
Common elements and common minerals • These elements can combine in a variety of ways to make different minerals. • Most minerals contain silicon and oxygen (plus other elements). These minerals are called silicate minerals. Crystal structure of almandine garnet (Fe 3 Al 2 Si 3 O 12 ) containing iron and aluminum in addition to silicon and oxygen. Fe Al Si O

Common elements and common minerals Why do we care about this? • Sustainability: The eight elements just listed are the most plentiful. Other elements are more rare; we find them less frequently and have a lower overall supply of them. • Ease of use: Silicate minerals tend to be refractory; they have high melting points and low solubilities, so they hold onto the elements within them. • Although the majority of Earth’s elements are found in silicate minerals, they are usually found in higher quantities in nonsilicate minerals, commonly oxide or sulfide minerals. It is more efficient to mine elements when they are found in higher concentrations.

If we want the element in the mineral (and not the mineral itself), then we seek nonsilicate minerals that contain the element, because it is easier (fewer resources are needed) to extract those elements from the mineral. For example, although the silicate mineral fayalite (Fe 2 Si. O 4) contains iron (Fe), it is easier and more efficient to extract iron from hematite (Fe 2 O 3), which is a nonsilicate mineral. Fayalite. Not an economic source for iron, though it contains iron. Hematite. Iron is commonly extracted from this mineral.

Mineral properties • A mineral's chemical and crystalline nature gives it properties that make it useful. • These properties also must be considered when determining how best to mine, process, and dispose of the mineral and mine waste.

Mineral properties: Chemistry • The elements within minerals give them distinct and useful properties. For example: Sulfur allows gunpowder to ignite at a lower temperature and provides fuel for the fire. Sulfur can be found as a mineral (native sulfur, S) Sulfur can also exist as an element within other minerals like pyrite (Fe. S).

Mineral properties: Chemistry For example: Aluminum metal is very lightweight but strong. Aluminum does not form a mineral on its own, but is beneficiated from the minerals gibbsite and bohemite. Aluminum alloys were extensively used for constructing Apollo 16 whose Command Service Module (CSM) "Casper" is shown here

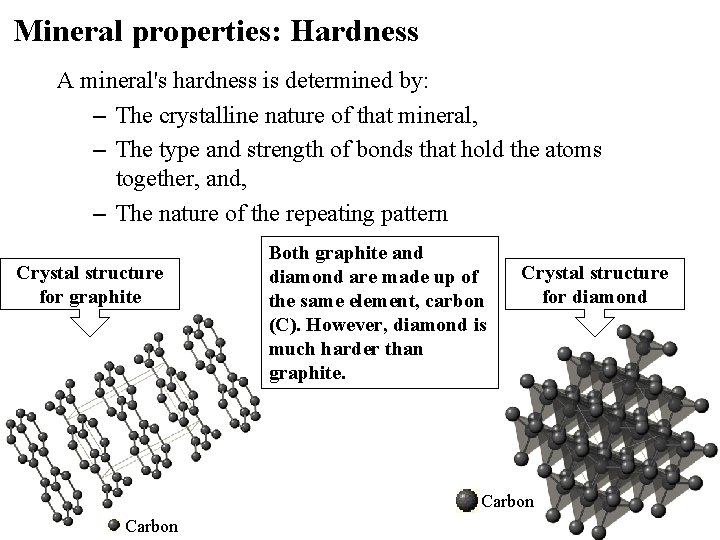
Mineral properties: Hardness A mineral's hardness is determined by: – The crystalline nature of that mineral, – The type and strength of bonds that hold the atoms together, and, – The nature of the repeating pattern Crystal structure for graphite Both graphite and diamond are made up of the same element, carbon (C). However, diamond is much harder than graphite. Crystal structure for diamond Carbon

Mineral properties: Hardness For example: Very hard minerals (like diamond, corundum, and garnet) are useful as abrasives. Saw blades impregnated with diamonds can cut rock, and sandpaper is often made with garnet sand. Almandine garnet crystals. Hardness of almandine garnet is 7– 8 On the Moh’s scale of hardness Talc (hardness = 1 on the Moh’s scale of hardness) is used in baby powder because it is a very soft mineral.

Mineral properties: Color Some minerals have distinct, and vibrant, colors. This makes them incredibly useful as pigments in paints, cosmetics, colored plastic, etc. The rust red color of hematite (left) and rust yellow color of limonite ( a variety of goethite, right) have long been used as a pigment. Malachite’s green color has made it useful in paints

Mineral properties: Color For example: The native people in Himba Village, Namibia, powder iron oxide minerals (like hematite and goethite) to color their skin and hair.

Mineral properties: Specific gravity is a relative density, determined both by a mineral's chemistry (minerals containing heavier elements will have higher specific gravities) and how closely together the atoms are packed. Galena (Pb. S) contains the heavy element lead, giving it a high specific gravity

Mineral properties: Behavior of light in the crystal The crystalline structure determines if and how light passes through a mineral. For example, light reflects inside of diamond, which gives diamond ring an exquisite sparkle. Other minerals (like rutile) are quite opaque, which makes titanium oxide (the chemical name of rutile) an important additive in things that need to be opaque, like paint. Some minerals are also useful in blocking other wavelengths of light; barite and lead (from the mineral galena) block X-rays, for example. The crystalline structure of calcite (Iceland spar variety) can split light rays passing through it, making the horizontal lines seen through the calcite crystal appear to double up in this photo. Iceland Spar crystals were once used in polarizing microscopes and telescopes because of this property.

Mineral properties: Luster describes how light interacts with the surface of a mineral. For example, the mineral hematite can have both metallic or nonmetallic luster; hematite with metallic luster (photo on left) is used to make jewelry.

Mineral properties: Crystal shape and cleavage These are determined by the nature of the crystalline structure. The sheet-like cleavage of muscovite allows it to be broken into tiny pieces of glitter. Muscovite’s cleavage causes it to break into sheets.

Mineral properties: Solubility Different minerals dissolve differently depending on their crystalline structure (the type of bonds) and chemistry. When a mineral dissolves, it breaks into the ions that it contains. Some dissolve quickly in water (or acidic or alkali solutions), whereas others are very stable. For example, people eat tablets made of calcite; that calcite dissolves in the stomach, neutralizing some of the acid and reducing heartburn TUMS® tablet dissolving in acidic water releasing bubbles of carbon dioxide.

Mineral properties: Solubility For some applications, an insoluble mineral is preferred. For example, the Eads Bridge that crosses the Mississippi River is faced with rock made of insoluble minerals below the water line, whereas more decorative limestone (made of the more soluble mineral calcite) faces the support above the water line. If a mineral is being mined from the element it contains, then it will be easier to extract that element from a soluble mineral.

Mineral properties: Magnetism The chemistry of certain minerals allows them to store an applied magnetic field. For example, magnetic minerals in a hard drive can be programmed to store information. A bar magnet hanging from a sample of magnetite. Magnetite is an iron oxide (Fe 3 O 4), and is naturally magnetic.

Mineral properties: Electrical conductivity is mainly determined by the presence of metallic bonds. Metals have these bonds, which is why metals are favored for wires. Copper’s electrical conductivity and resistance to corrosion make it ideal for electric wiring. Copper can be found as a pure metal (native copper, Cu) Copper is often beneficiated from minerals like chalcopyrite (Cu. Fe. S 2).

Mineral properties: Electrical conductivity Minerals that have low electric conductivity will be used for insulators, which block and/or confine the electric current. Clay minerals such as kaolinite show very low electrical and thermal conductivity. Ceramic insulators made from clay minerals are used for supporting electric power lines

Mineral properties: Thermal conductivity Minerals can also be used to conduct or confine heat. Thermal conductivity is determined by both a mineral’s chemistry and crystalline structure Clay minerals such as kaolinite show very low electrical and thermal conductivity. Ceramic kitchen utensils made from clay minerals are used to confine the temperature of food and beverages

Mineral properties: Melting point Different minerals melt at different temperatures. Minerals with high melting points are used for hightemperature applications. For example, the mineral graphite is used for lining furnaces and as crucibles for melting metals

Mineral properties: Response to stress Different minerals/rocks behave differently under stress. For example, while many minerals will shatter if hit with a hammer, gold is malleable. This allowed early people to easily shape it into ornaments. Piezoelectricity is another property related to stress. An electric current is generated in piezoelectric minerals when a stress is applied. For example, a hammer hits a piezoelectric crystal, and this will generate a spark to ignite a cigarette lighter. The piezoelectricity of quartz allows it to be used to tell time (in quartz crystal watches), and piezoelectricity is also useful in transformers and motors.