Mineral NUTRITION The study of how plants obtain

Mineral NUTRITION The study of how plants obtain and use mineral nutrients Retno Mastuti Biology Department Brawijaya University 2014

Mineral Nutrition How plants acquire and use mineral nutrients 1. Why is mineral nutrition important? In most natural soils, the availability of mineral nutrients limits plant growth and primary productivity. 2. What are the essential mineral nutrients? • classification systems 3. Mineral nutrients in the soil • nutrient availability • adsorption to soil particles • effects of p. H 4. Roots and mineral nutrient acquisition • root structure • depletion zones 5. Mycorrhizae 6. Nitrogen - the most limiting soil nutrient

Nutritional needs of plants Plant tissues contain > 60 kinds of elements are all of these elements essential for growth ? why they are essential ? how the plant absorb them ? how they are utilized ? what effects if it is lacking ?

Essential elements: are required for normal growth and reproduction. Their absence inhibits plant life cycle. Classification according to: Have a clear physiological role, have a direct or indirect action in plant metabolism are part of an essential molecule (macromolecule, metabolite) inside the plant No other element can replace it and correct the deficiency relative concentration in plant tissue : 17 elements are classified as 9 macronutrients (present at > 10 mmol / kg dry wt. ) 8 micronutrients (< 10 mmol / kg dry wt. ) biochemical role and physiological function
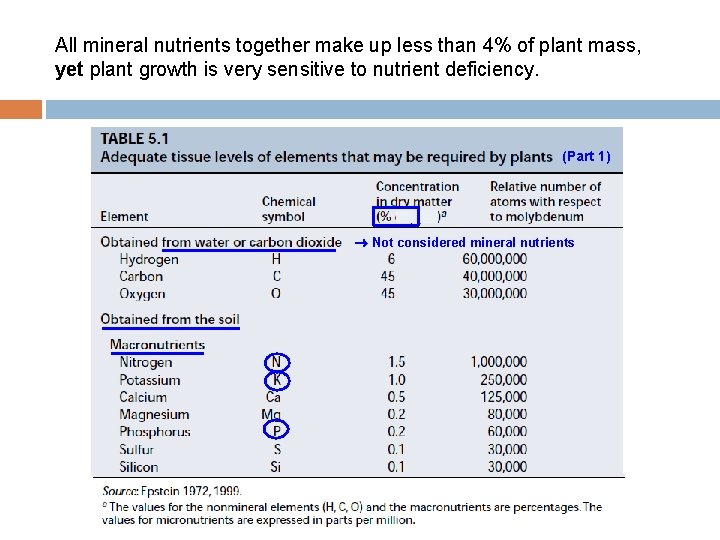
All mineral nutrients together make up less than 4% of plant mass, yet plant growth is very sensitive to nutrient deficiency. (Part 1) Not considered mineral nutrients
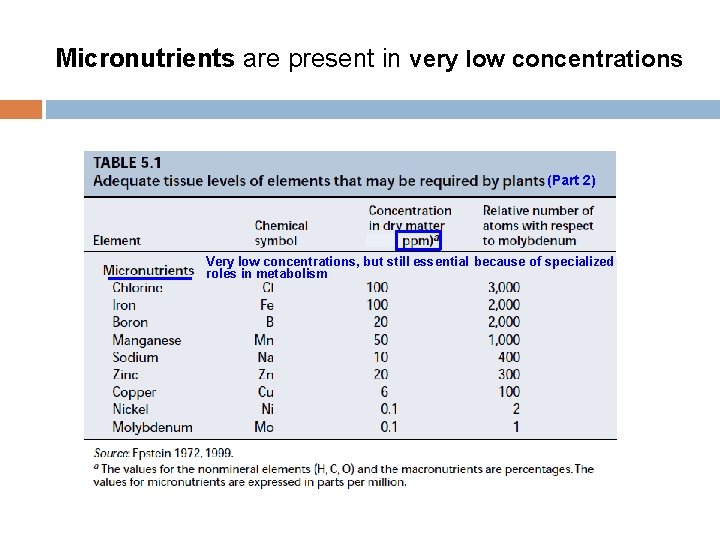
Micronutrients are present in very low concentrations (Part 2) Very low concentrations, but still essential because of specialized roles in metabolism
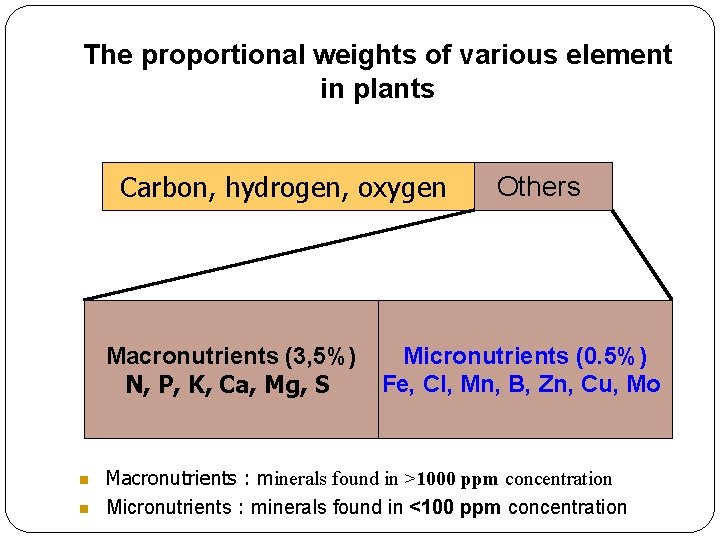
The proportional weights of various element in plants Carbon, hydrogen, oxygen Macronutrients (3, 5%) N, P, K, Ca, Mg, S Others Micronutrients (0. 5%) Fe, Cl, Mn, B, Zn, Cu, Mo Macronutrients : minerals found in >1000 ppm concentration Micronutrients : minerals found in <100 ppm concentration

Generalizations of Nutrition Requirement : plants require different amounts of different element (ex. H and Mo) different elements are absorbed in different forms (ex. cation – anion) Plants can accumulate an element although there is no specific requirement for the element Plants can accumulate an element although it is not considered to be an essential element most elements have several functions

General categories of function of essential elements : Can be parts of structural unit (ex. C, N) Can be parts of compounds involved in important metabolism (ex. Mg, P) Can be function as enzyme activators or inhibitor Can alter the osmotic potential of a cell help to maintain the osmotic balance of a cell (ex. K) Part of carbon compound (N, S) Important in energy storage or structural integrity (P, Si, B) Remain in ionic form (K, Ca, Mg, Cl, Mn, Na) Involved in redox reaction (Fe, Zn, Cu, Ni, Mo)

Inadequate supply - Mineral element deficiencies produce visible symptoms When minerals are deficient the growth of the plant is stunted or the plant shows other symptoms. The combination of symptoms can be traced to the roles that mineral plays in metabolism or physiology.
Mineral defficiensies disrupt metabolism and function Inadequate supply Nutritional disorders Related the roles played by essential elements in normal metabolims Characteristics defficiencies symptoms

Mobility and retranslocate

Mobility of Mineral Elements in Phloem Mobile Nutrients – deficiencies typically appear on older growth first. Nutrient has moved to the younger parts of the plants Immobile nutrients – deficiencies typically appear on newer growth and shoot tips first. Initially sequestered in younger leaves which are now the oldest ones. When the soil is exhausted of mineral the younger zones suffer the symptoms because the minerals are held by the older leaves

Some common symptoms Stunted growth : stem ~ N deficiency root ~ P deficiency Chlorosis (Mg, N, and Fe deficiencies) : chlorophyll synthesis chlorophyll degradation Necrosis : dead spots or zones (Mg, K or Mn deficiency) Color changes : ex. excessive anthocyanin production in stems ~ P deficiency

K deficiency P deficiency Zn deficiency Mg deficiency N deficiency

The absence of essential elements causes deficiency symptoms Essential because of their metabolic functions Characteristic deficiency symptoms shown because of these roles Typical deficiency responses are Chlorosis: yellowing; precursor to Necrosis: tissue death Expressed when a supply of an essential metabolite becomes limiting in the environment Element concentrations are limiting for growth when they are below the critical concentraion This is the concentration of nutrient in the tissue just below the level giving maximum growth

Limiting nutrient levels negatively affect growth Plant responses to limiting nutrients usually very visible: affects yield/growth! Again, chlorosis and necrosis of leaves is typical Sometimes straightforward relationship e. g. , in chlorosis (lack of green color), N: chlorophyll component Mg: cofactor in chlorophyll synthesis Ctrl -P - Ca -N - Fe
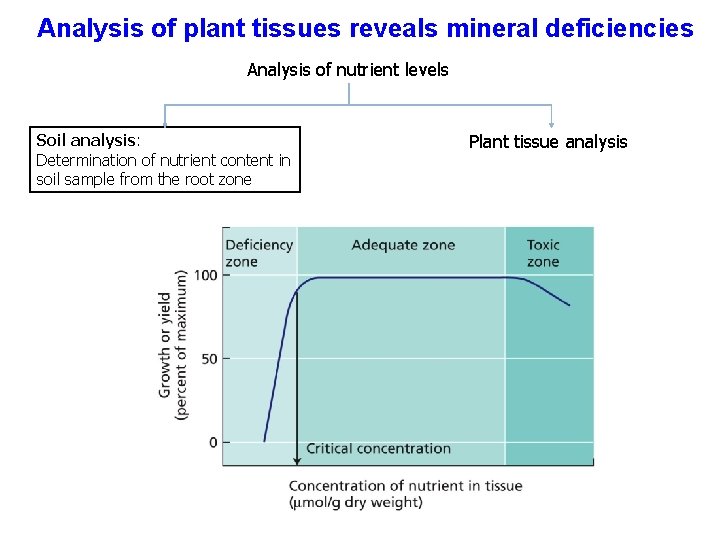
Analysis of plant tissues reveals mineral deficiencies Analysis of nutrient levels Soil analysis: Determination of nutrient content in soil sample from the root zone Plant tissue analysis
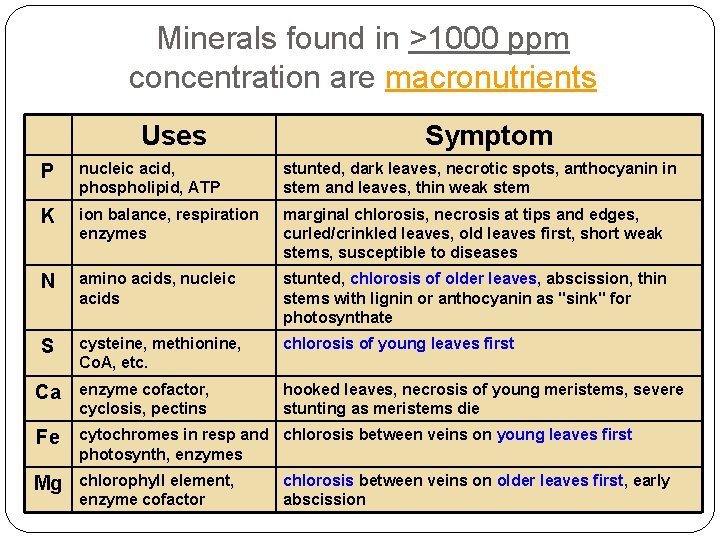
Minerals found in >1000 ppm concentration are macronutrients Uses Symptom P nucleic acid, phospholipid, ATP stunted, dark leaves, necrotic spots, anthocyanin in stem and leaves, thin weak stem K ion balance, respiration enzymes marginal chlorosis, necrosis at tips and edges, curled/crinkled leaves, old leaves first, short weak stems, susceptible to diseases N amino acids, nucleic acids stunted, chlorosis of older leaves, abscission, thin stems with lignin or anthocyanin as "sink" for photosynthate S cysteine, methionine, Co. A, etc. chlorosis of young leaves first Ca enzyme cofactor, cyclosis, pectins hooked leaves, necrosis of young meristems, severe stunting as meristems die Fe cytochromes in resp and chlorosis between veins on young leaves first photosynth, enzymes Mg chlorophyll element, enzyme cofactor chlorosis between veins on older leaves first, early abscission
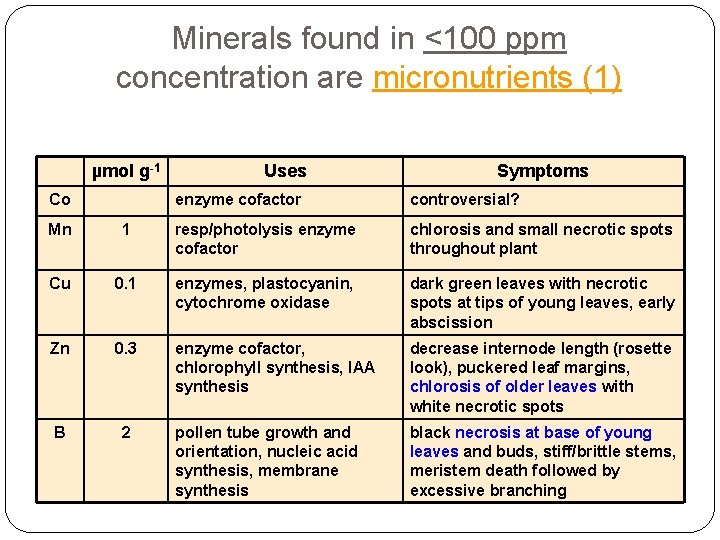
Minerals found in <100 ppm concentration are micronutrients (1) µmol g-1 Uses Symptoms Co enzyme cofactor controversial? Mn 1 resp/photolysis enzyme cofactor chlorosis and small necrotic spots throughout plant Cu 0. 1 enzymes, plastocyanin, cytochrome oxidase dark green leaves with necrotic spots at tips of young leaves, early abscission Zn 0. 3 enzyme cofactor, chlorophyll synthesis, IAA synthesis decrease internode length (rosette look), puckered leaf margins, chlorosis of older leaves with white necrotic spots B 2 pollen tube growth and orientation, nucleic acid synthesis, membrane synthesis black necrosis at base of young leaves and buds, stiff/brittle stems, meristem death followed by excessive branching
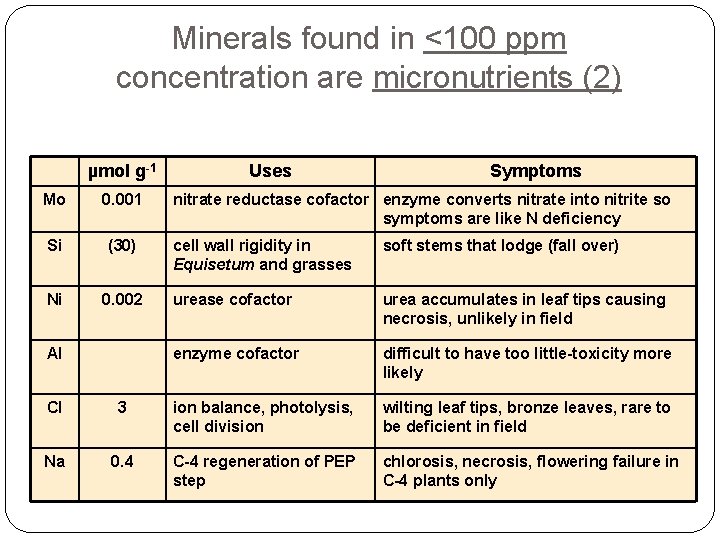
Minerals found in <100 ppm concentration are micronutrients (2) µmol g-1 Mo 0. 001 Si (30) Ni Uses Symptoms nitrate reductase cofactor enzyme converts nitrate into nitrite so symptoms are like N deficiency cell wall rigidity in Equisetum and grasses soft stems that lodge (fall over) 0. 002 urease cofactor urea accumulates in leaf tips causing necrosis, unlikely in field Al enzyme cofactor difficult to have too little-toxicity more likely Cl 3 ion balance, photolysis, cell division wilting leaf tips, bronze leaves, rare to be deficient in field Na 0. 4 C-4 regeneration of PEP step chlorosis, necrosis, flowering failure in C-4 plants only
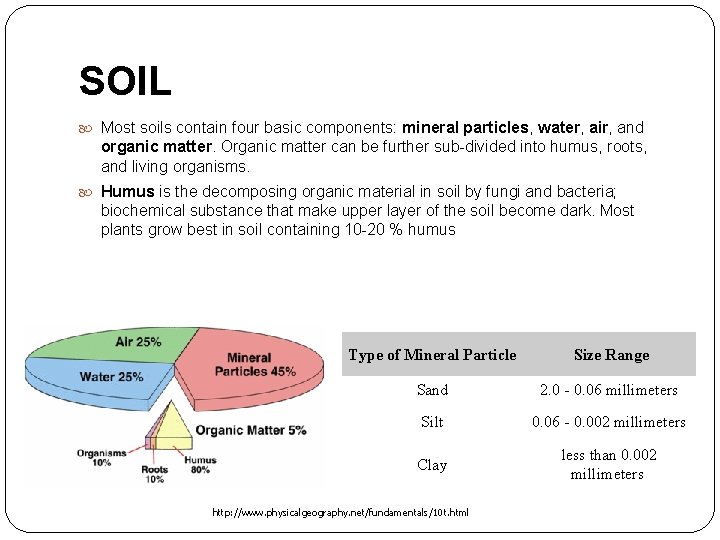
SOIL Most soils contain four basic components: mineral particles, water, air, and organic matter. Organic matter can be further sub-divided into humus, roots, and living organisms. Humus is the decomposing organic material in soil by fungi and bacteria; biochemical substance that make upper layer of the soil become dark. Most plants grow best in soil containing 10 -20 % humus Type of Mineral Particle Size Range Sand 2. 0 - 0. 06 millimeters Silt 0. 06 - 0. 002 millimeters Clay less than 0. 002 millimeters http: //www. physicalgeography. net/fundamentals/10 t. html

Components of soil Humus Air Living organism • Is the decomposing organic material in soil by fungi and bacteria • Biochemical substance that make upper layer of the soil become dark • Most plants grow best in soil containing 10 -20 % humus • About 23 -30 % of the volume of most soils is air • Critical role: provides oxygens for root • Compare: clay soil and sandy soil • Organism mix and refine the soil. • Organism add humus to the soil • Respiration by these organism increase the amount of CO 2 in the soil • Many organism affect the availability of nutirents in the soil
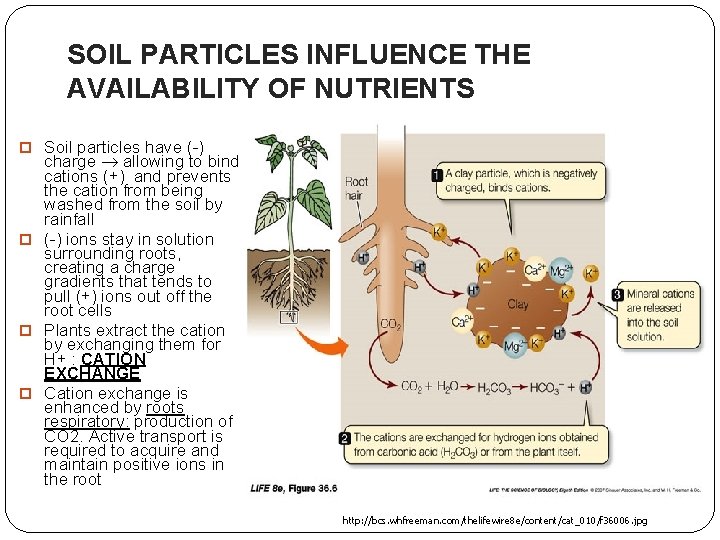
SOIL PARTICLES INFLUENCE THE AVAILABILITY OF NUTRIENTS Soil particles have (-) charge allowing to bind cations (+) and prevents the cation from being washed from the soil by rainfall (-) ions stay in solution surrounding roots, creating a charge gradients that tends to pull (+) ions out off the root cells Plants extract the cation by exchanging them for H+ : CATION EXCHANGE Cation exchange is enhanced by roots respiratory: production of CO 2. Active transport is required to acquire and maintain positive ions in the root http: //bcs. whfreeman. com/thelifewire 8 e/content/cat_010/f 36006. jpg
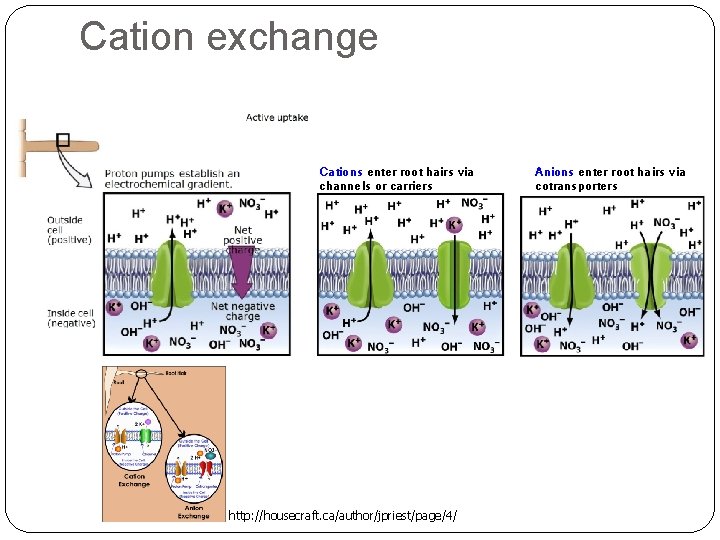
Cation exchange Cations enter root hairs via channels or carriers http: //housecraft. ca/author/jpriest/page/4/ Anions enter root hairs via cotransporters

Depletion zones - regions of lower nutrient concentration -develop around roots

Cation exchange capacity is soil p. H dependent : nutrient availability soil microbes : fungi – acidic; bacteria – alkaline root growth : slightly acidic soil (p. H 5. 5 & 6. 5) Acid soil ----- rocks release K+, Mg 2+, Ca 2+, Mn 2+ solubility of SO 42 -, H 2 PO 4 -, HCO 3 So, availability to roots Soil p. H due to : - decomposition of organic matter - Rainfall decomposition of org. Matter : (microbial decomposition) : CO 2 + H 2 O H+ + HCO 3 NH 3 + O 2 HNO 3 (nitric acid) H 2 SO 4 (sulfuric acid) H+ displace K+, Mg 2+ ----- K+, Mg 2+ etc. Available --- p. H
- Slides: 27