Mimicking Oxidative Metabolism Ox Met using ROXY EC

Mimicking Oxidative Metabolism “Ox. Met” using ROXY EC System ASMS 2015 St. Louis, MO, USA 1

Outline I. Introduction - Drug metabolism – what you should know - Case Study 1 – Acetaminophen - Case Study 2 – Amodiaquine - Examples from Industry II. Conclusions 2

Drug/Xenobiotic Metabolism • Many drugs metabolize in the liver by Cytochrome P 450 enzymes • Drug metabolism consist of two phases: Phase 1: oxidation, reduction, hydrolysis Phase 2: conjugation (adduct formation) 3

Techniques used in Drug Metabolism 1. Test animals (rodents) in-vivo 2. Microsomes in-vitro Rat Liver Microsomes (RLM) Human Liver Microsomes (HLM) 3. Electrochemical mimicking (= simulation) in-electro 4
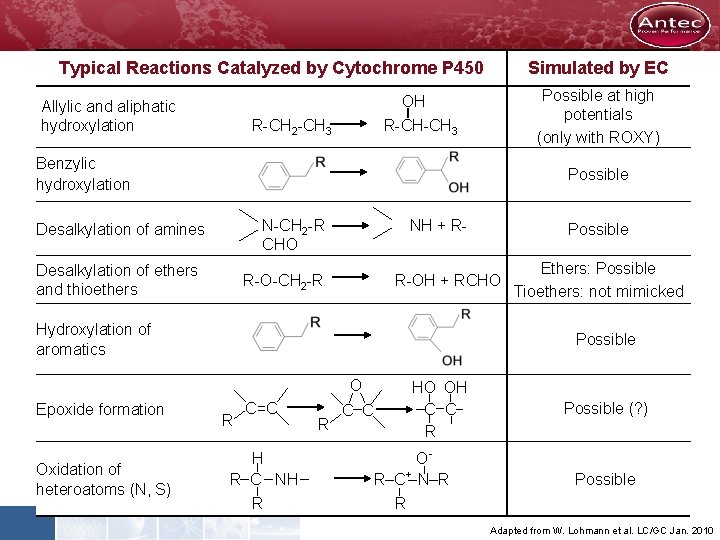
Typical Reactions Catalyzed by Cytochrome P 450 Simulated by EC Possible at high potentials (only with ROXY) OH Allylic and aliphatic hydroxylation R-CH 2 -CH 3 R-CH-CH 3 Benzylic hydroxylation Possible N-CH 2 -R CHO Desalkylation of amines Desalkylation of ethers and thioethers NH + R- R-O-CH 2 -R R-OH + RCHO Hydroxylation of aromatics Oxidation of heteroatoms (N, S) Ethers: Possible Tioethers: not mimicked Possible O Epoxide formation Possible R C=C H R C NH R R C C HO OH C C R OR C+ N R R Possible (? ) Possible Adapted from W. Lohmann et al. LC/GC Jan. 2010

Case Study 1: Acetaminophen Paracetamol Panadol Tylenol APAP 6
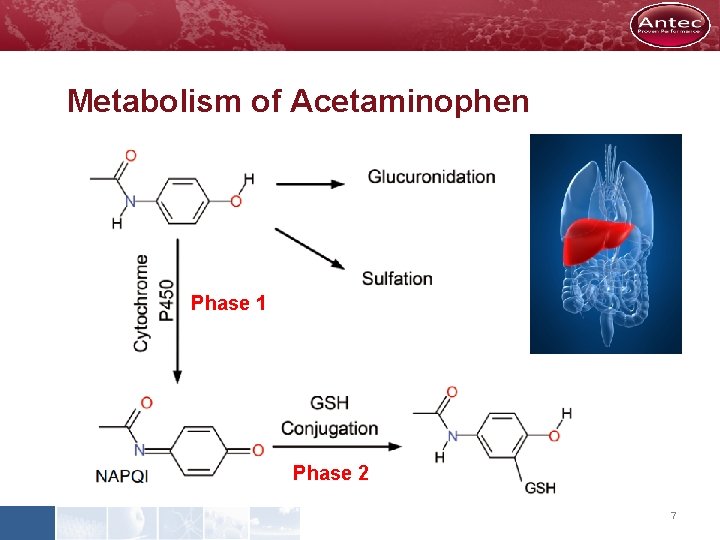
Metabolism of Acetaminophen Phase 1 Phase 2 7
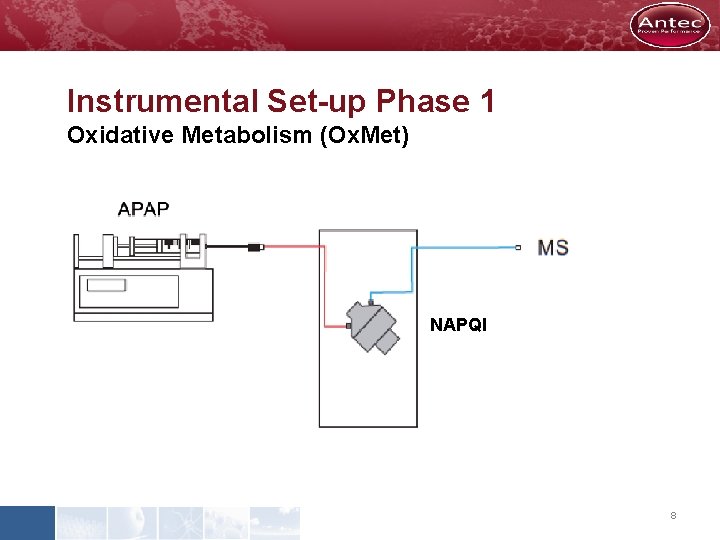
Instrumental Set-up Phase 1 Oxidative Metabolism (Ox. Met) NAPQI 8

Positive vs. Negative ESI-MS of NAPQI negative ESI positive ESI D. Tsikas, J. of Chrom. B, 879 (2011) 1476 -1484 9
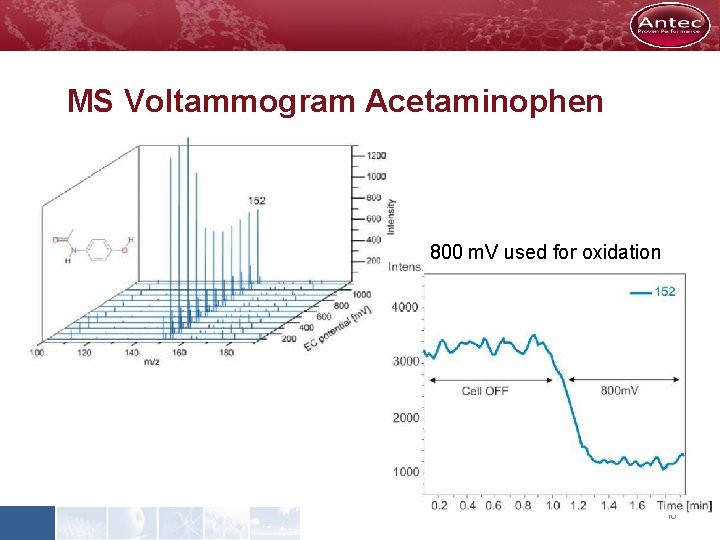
MS Voltammogram Acetaminophen 800 m. V used for oxidation 10
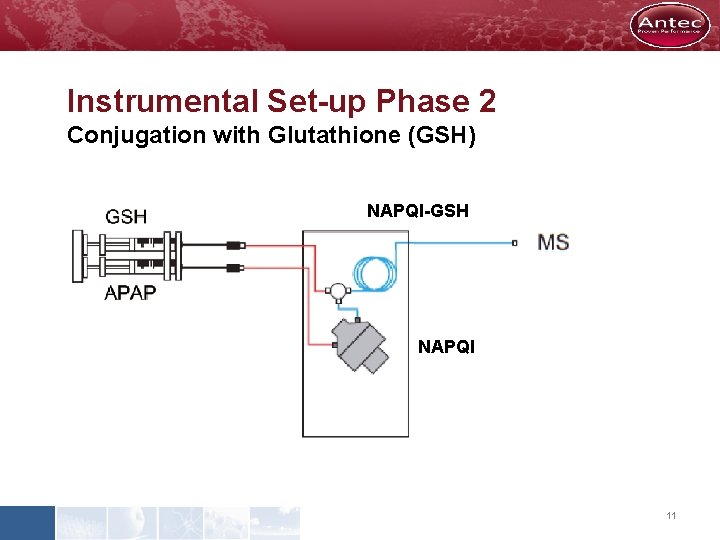
Instrumental Set-up Phase 2 Conjugation with Glutathione (GSH) NAPQI-GSH NAPQI 11
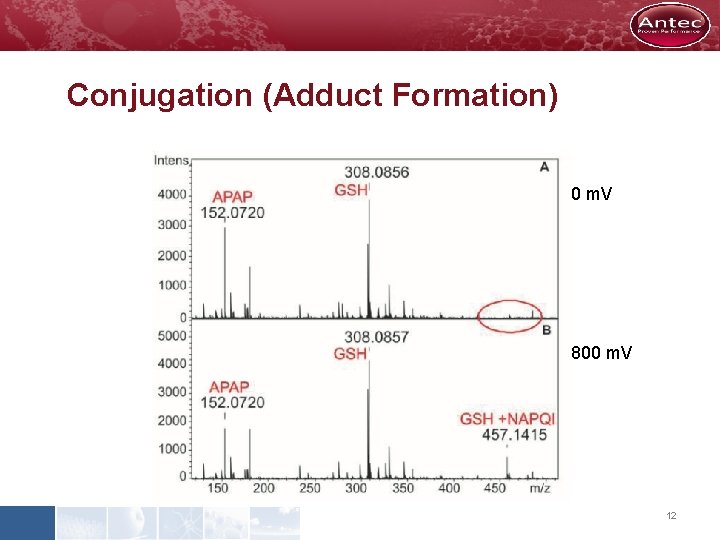
Conjugation (Adduct Formation) NAPQI-GSH NAPQI 0 m. V 800 m. V 12
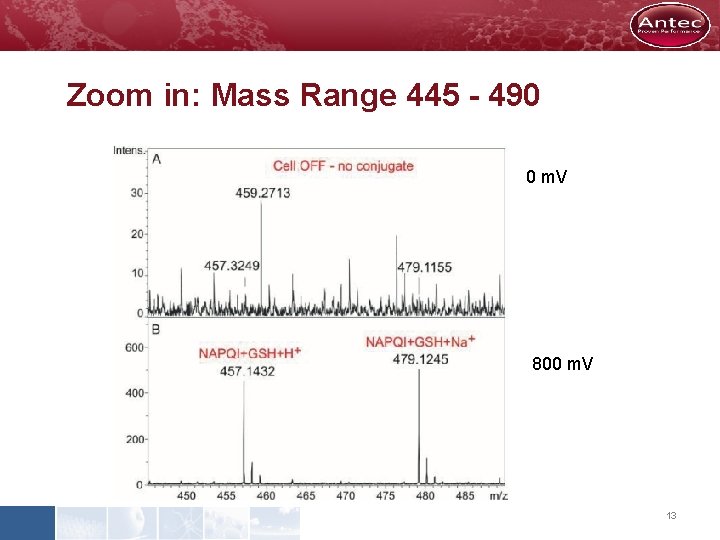
Zoom in: Mass Range 445 - 490 0 m. V 800 m. V 13

Case Study 2: Amodiaquine Camoquin Flavoquine 14
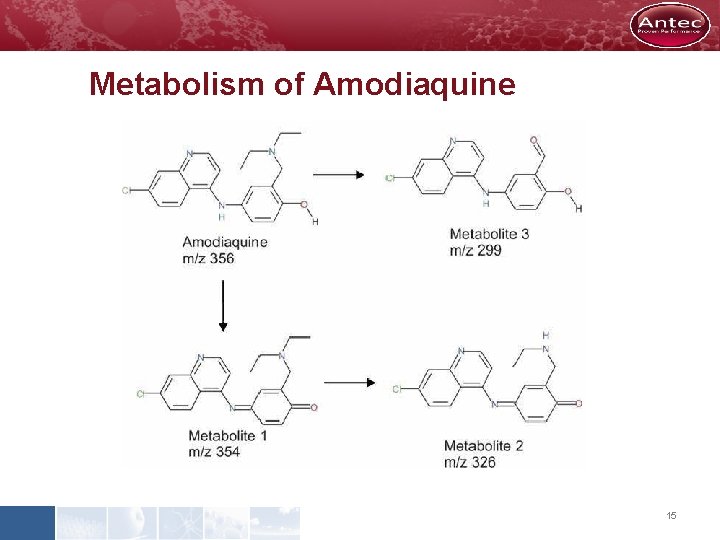
Metabolism of Amodiaquine 15

Mimicking Amodiaquine Metabolism by EC/MS Cell OFF 400 m. V 1200 m. V Metabolites 2 & 3 Metabolite 1 Amodiaquine 16
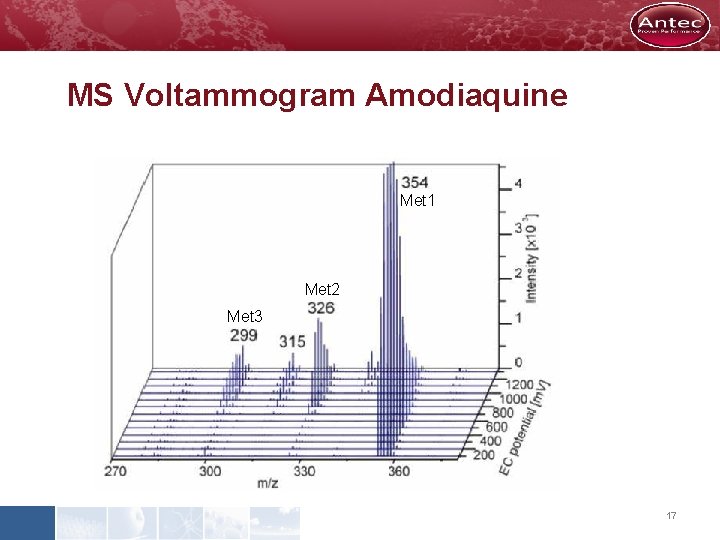
MS Voltammogram Amodiaquine Met 1 Met 2 Met 3 17
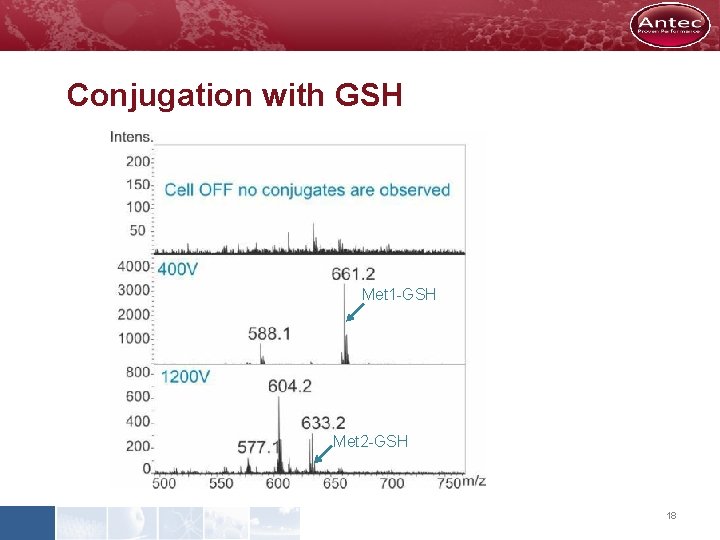
Conjugation with GSH Met 1 -GSH Met 2 -GSH 18

Examples form Industry 19
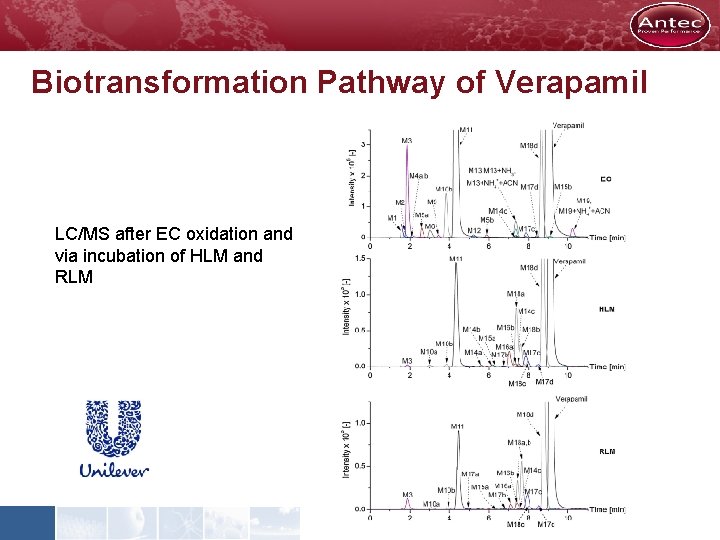
Biotransformation Pathway of Verapamil LC/MS after EC oxidation and via incubation of HLM and RLM

Biotransformation Pathway of Verapamil Molecular formula of Verapamil and it metabolites after EC oxidation and incubation with HLM and RLM

Biotransformation Pathway of Verapamil Conclusions • Structure of electrochemically generated metabolites were elucidated on basis of accurate mass data and MS/MS experiments • All of the metabolic products of Verapamil incl. Norverapamil can be easily simulated using this purely instrumental technique • Furthermore newly reported metabolic reactions products like carbinolamines or imine methides become accessible • EC/LC/MS is a powerful tool in drug discovery and development providing faster and often more comprehensive results than in vivo approaches S. Jahn et al. , J. of Chroamtography A, 1218 (2011) 9210 -9220
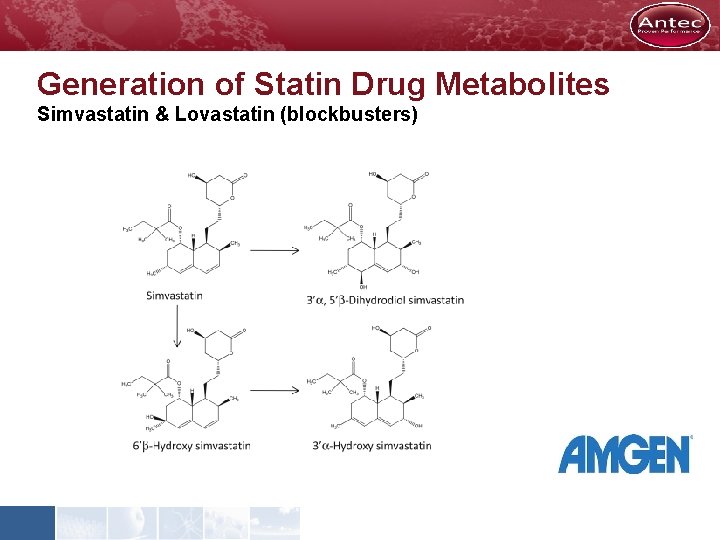
Generation of Statin Drug Metabolites Simvastatin & Lovastatin (blockbusters)
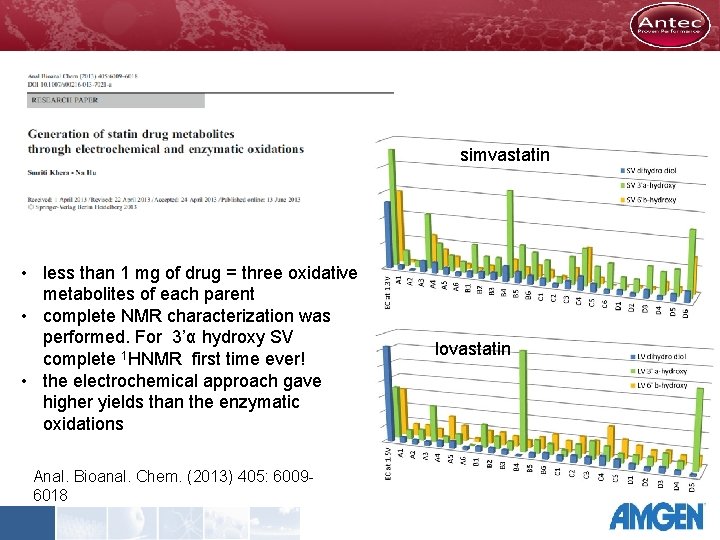
simvastatin • less than 1 mg of drug = three oxidative metabolites of each parent • complete NMR characterization was performed. For 3’α hydroxy SV complete 1 HNMR first time ever! • the electrochemical approach gave higher yields than the enzymatic oxidations Anal. Bioanal. Chem. (2013) 405: 60096018 lovastatin

Conclusions EC/MS in Drug Metabolism § ionization techniques: ESI (+/-) API, APCI § weeks vs. seconds § no isolation/clean-up § phase I and II metabolism § complementary to in vivo and in vitro § saving rodents (mice, rats) § safety (no exposure to biohazard samples) EC/MS powerful tool for biomimetic oxidation
- Slides: 25