Midterm exam location Odette Building room 104 Feb

• Midterm exam location Odette Building, room 104 Feb. 3 rd from 1 - 2: 20 p. m. March 10 th from 1 - 2: 20 p. m

Solutions contain more than one types of electrolytes • The ionic strength of the solution equals the sum of the ionic strength of each individual compound. Example: Calculate the ionic strength of a solution that contains 0. 050 mol kg-1 K 3[Fe(CN)6](aq), 0. 040 mol kg-1 Na. Cl(aq), and 0. 03 mol kg-1 Ce(SO 4)2 (aq). Solution: I (K 3[Fe(CN)6]) = ½( 12*(0. 05*3) + (-3)2*0. 05) = ½ (0. 15 + 0. 45) = 0. 3; I (Na. Cl) = ½(12*0. 04 + (-1)2*0. 04) = 0. 04; I (Ce(SO 4)2) = ½(42*0. 03 + (-2)2*(2*0. 03)) =0. 36; So, I = I(K 3(Fe(CN)6]) + I(Na. Cl) + I(Ce(SO 4)2) = 0. 3 + 0. 04 + 0. 36 = 0. 7

Calculating the mean activity coefficient Example: Calculate the ionic strength and the mean activity coefficient of 2. 0 m mol kg-1 Ca(NO 3)2 at 25 o. C. Solution: In order to calculate the mean activity coefficient with the eq. 10. 3, one needs to know the ionic strength of the solution. Thus, the right approach is first to get I and then plug I into the equation 10. 3. I = ½(22*0. 002 + (-1)2*(2*0. 002)) = ½ *6*0. 002 = 0. 006; From Debye-Huckel limiting equation, log(γ±) = - |2*1|*A*(0. 006)1/2; = - 2*0. 509*0. 0775; = -0. 0789; γ ± = 0. 834;
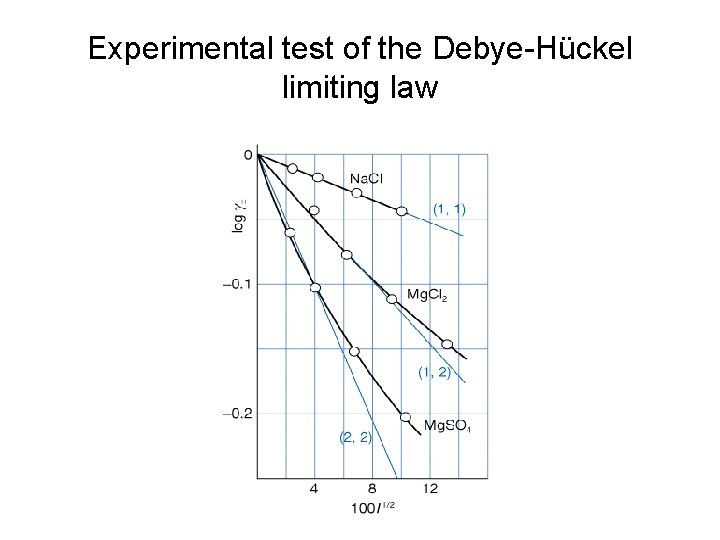
Experimental test of the Debye-Hückel limiting law

Accuracy of the Debye-Hückel limiting law Example: The mean activity coefficient in a 0. 100 mol kg-1 Mn. Cl 2(aq) solution is 0. 47 at 25 o. C. What is the percentage error in the value predicted by the Debye-Huckel limiting law? Solution: First, calculate the ionic strength I = ½[22*0. 1 + (-1)2*(2*0. 1)] = 0. 3 to calculate the mean activity coefficient. log(γ) = -|2*(-1)| A* (0. 3)1/2; = - 2*0. 509*0. 5477 = - 0. 5576 so γ = 0. 277 Error = (0. 47 -0. 277)/0. 47 * 100% = 41%
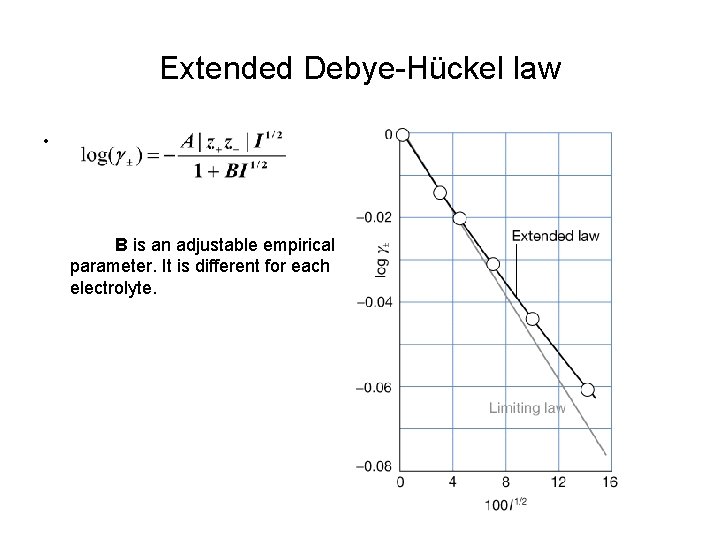
Extended Debye-Hückel law • B is an adjustable empirical parameter. It is different for each electrolyte.

Calculating parameter B Example : The mean activity coefficient of Na. Cl in a diluted aqueous solution at 25 o. C is 0. 907 (at 10. 0 mmol kg-1). Estimate the value of B in the extended Debye-Huckel law. Solution: First calculate the ionic strength I = ½[12*0. 01 + (-1)2*0. 01] = 0. 01 From equation log(0. 907) = - (0. 509|1*(-1)|*0. 011/2)/(1+ B*0. 011/2) B = - 1. 67

Half-reactions and electrodes Two types of electrochemical cells: 1. Galvanic cell: is an electrochemical cell which produces electricity as a result of the spontaneous reactions occurring inside it. 2. Electrolytic cell: is an electrochemical cell in which a non-spontaneous reaction is driven by an external source of current.

• Other important concepts include: Oxidation: the removal of electrons from a species. Reduction: the addition of electrons to a species. Redox reaction: a reaction in which there is a transfer of electrons from one species to another. Reducing agent: an electron donor in a redox reaction. Oxidizing agent: an electron acceptor in a redox reaction. • Two type of electrodes: Anode: the electrode at which oxidation occurs. Cathode: the electrode at which reduction occurs
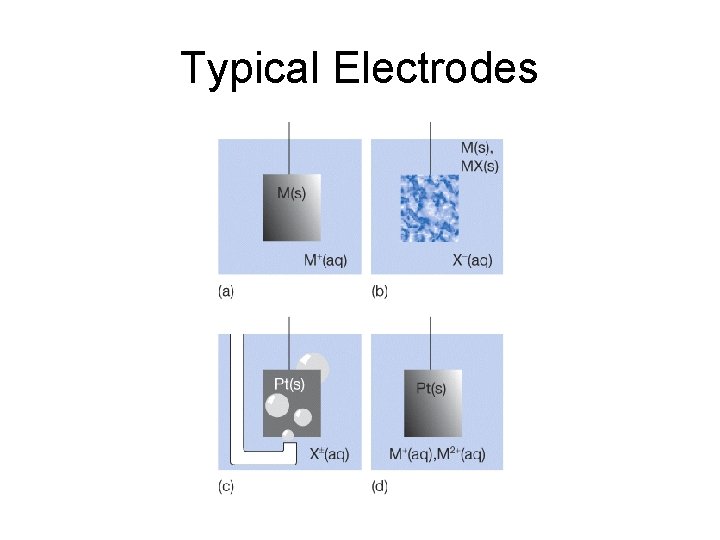
Typical Electrodes
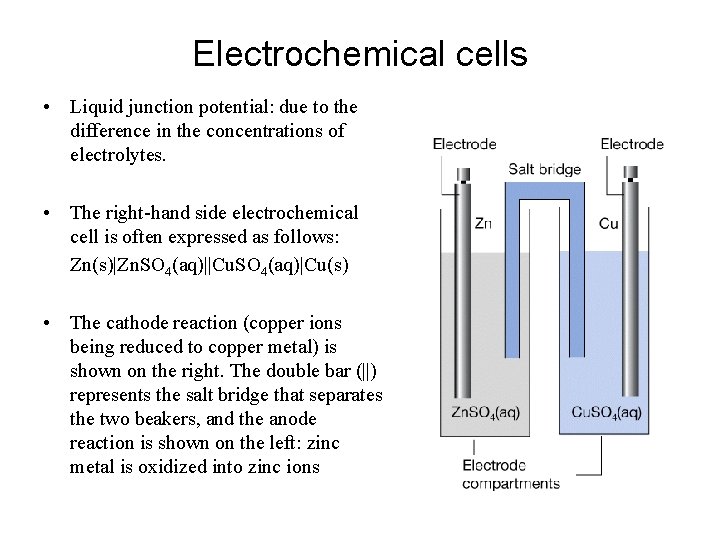
Electrochemical cells • Liquid junction potential: due to the difference in the concentrations of electrolytes. • The right-hand side electrochemical cell is often expressed as follows: Zn(s)|Zn. SO 4(aq)||Cu. SO 4(aq)|Cu(s) • The cathode reaction (copper ions being reduced to copper metal) is shown on the right. The double bar (||) represents the salt bridge that separates the two beakers, and the anode reaction is shown on the left: zinc metal is oxidized into zinc ions

In the above cell, we can trace the movement of charge. – – Electrons are produced at the anode as the zinc is oxidized The electrons flow though a wire, which we can use for electrical energy The electrons move to the cathode, where copper ions are reduced. The right side beaker builds up negative charge. Cl- ions flow from the salt bridge into the zinc solution and K+ ions flow into the copper solution to keep charge balanced. To write the half reaction for the above cell, Right-hand electrode: Cu 2+(aq) + 2 e- → Cu(s) Left-hand electrode: Zn 2+(aq) + 2 e- → Zn(s) The overall cell reaction can be obtained by subtracting lefthand reaction from the right-hand reaction: Cu 2+(aq) + Zn(s) → Cu(s) + Zn 2+(aq)

Expressing a reaction in terms of half-reactions Example : Express the formation of H 2 O from H 2 and O 2 in acidic solution as the difference of two reduction halfreactions. (in class discussion) Redox couple: the reduced and oxidized species in a halfreaction such as Cu 2+/Cu, Zn 2+/Zn…. Ox + v e- → Red The quotient is defined as: Q = a. Red/a. Ox Example: Write the half-reaction and the reaction quotient for a chlorine gas electrode. (in class discussion)
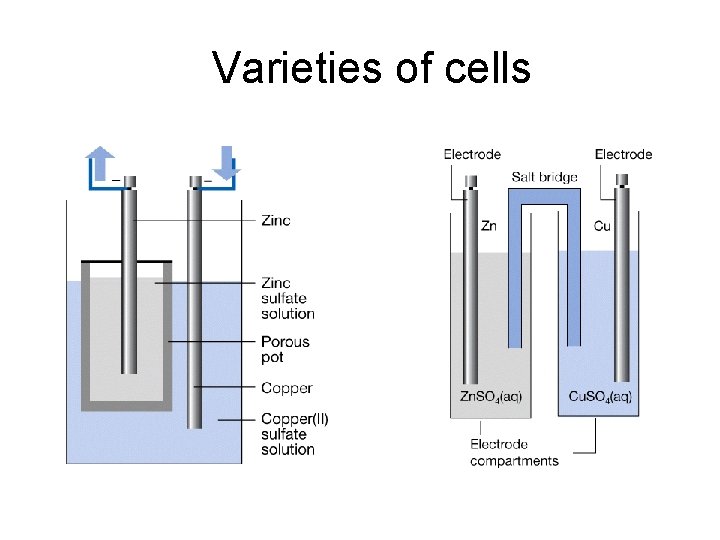
Varieties of cells
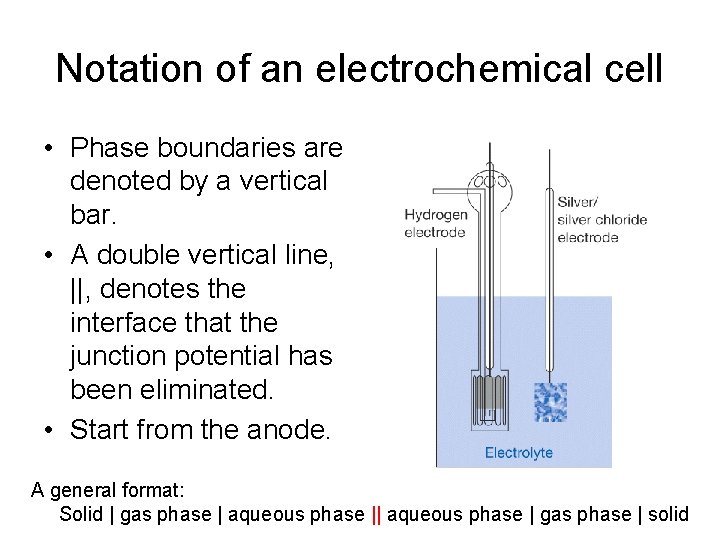
Notation of an electrochemical cell • Phase boundaries are denoted by a vertical bar. • A double vertical line, ||, denotes the interface that the junction potential has been eliminated. • Start from the anode. A general format: Solid | gas phase | aqueous phase | gas phase | solid

Cell Potential • Cell potential: the potential difference between two electrodes of a galvanic cell (measured in volts V). • Maximum electrical work : we, max = ΔG • Electromotive force, E, • Relationship between E and Δr. G: Δr. G = -νFE where ν is the number of electrons that are exchanged during the balanced redox reaction and F is the Faraday constant, F = e. NA. • At standard conditions, this equation can be written as Δr. Gθ = -νFEθ
- Slides: 16