MICROWAVE SPECTRUM AND AB INITIO CALCULATIONS OF metaCHLOROBENZALDEHYDE

MICROWAVE SPECTRUM AND AB INITIO CALCULATIONS OF meta-CHLOROBENZALDEHYDE Sean Arnold, Jessica Garrett, & Dr. Gordon Brown Department of Science and Mathematics Coker College 300 E College Ave. Hartsville, SC 29550 RK 15

Applications of m-chlorobenzaldehyde Its Schiff base is known to have antifungal properties Used in the synthesis of amino alcohols Common starting material in organic synthesis
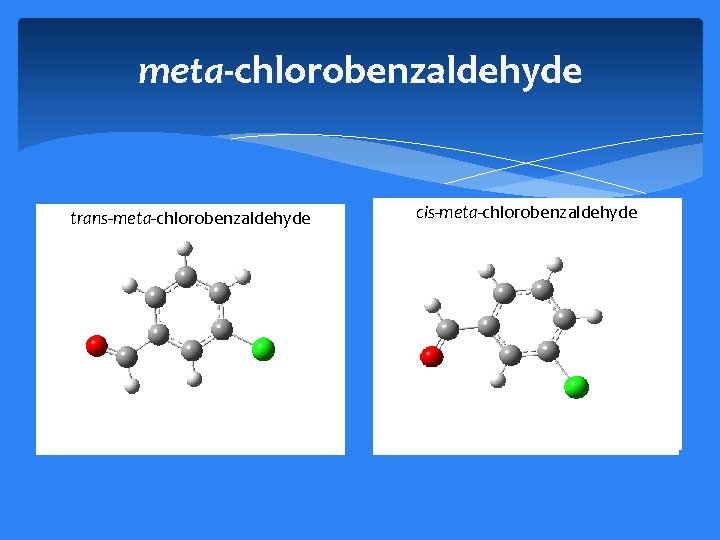
meta-chlorobenzaldehyde trans-meta-chlorobenzaldehyde cis-meta-chlorobenzaldehyde
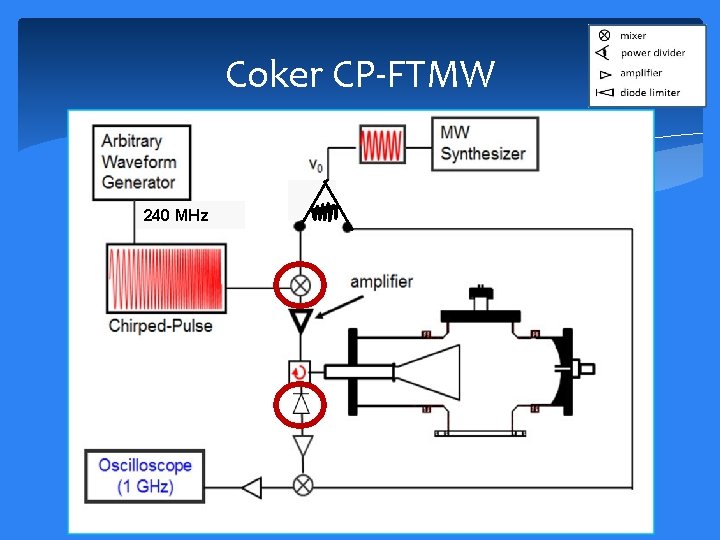
Coker CP-FTMW 240 MHz
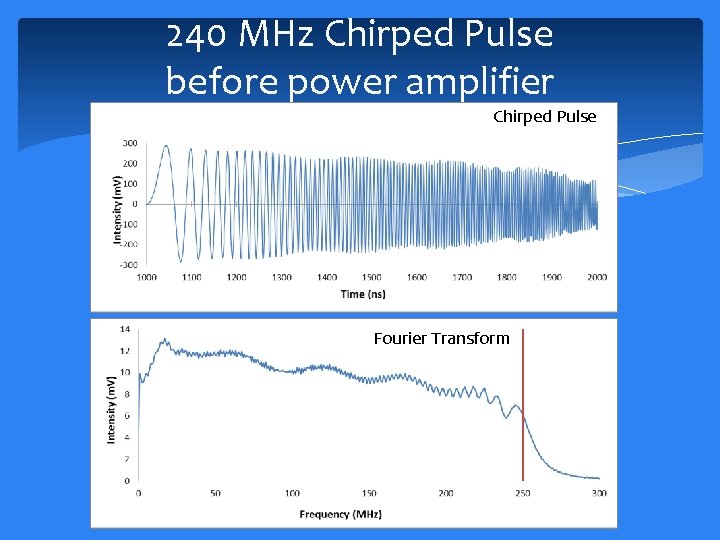
240 MHz Chirped Pulse before power amplifier Chirped Pulse Fourier Transform
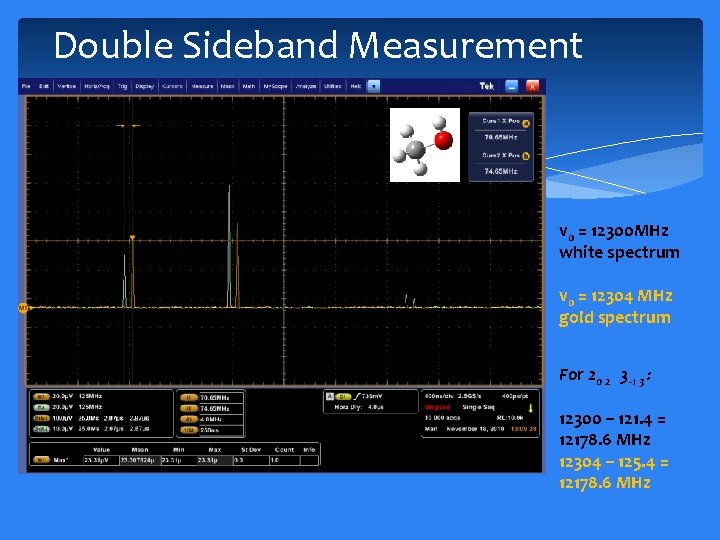
Double Sideband Measurement 20 2 3 -1 3 165 12 174 13 v 0 = 12300 MHz white spectrum 51 5 41 4 v 0 = 12304 MHz gold spectrum For 20 2 3 -1 3 : 12300 – 121. 4 = 12178. 6 MHz 12304 – 125. 4 = 12178. 6 MHz

Coker CP-FTMW
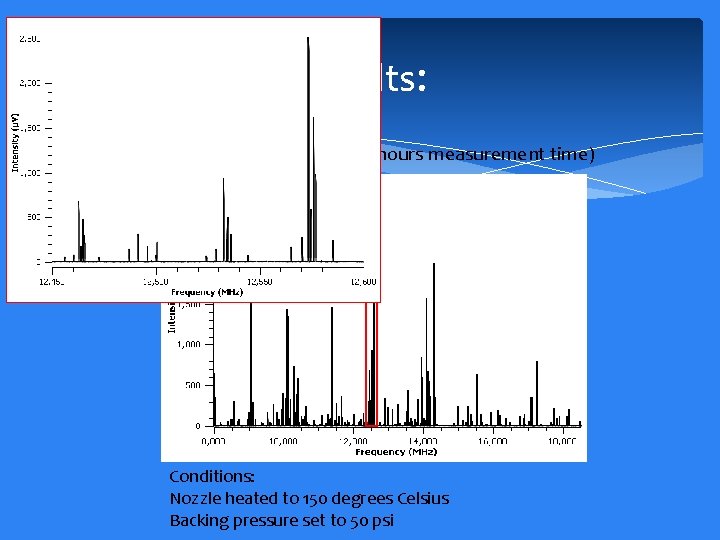
Results: 10, 000 signal averages (~40 hours measurement time) Conditions: Nozzle heated to 150 degrees Celsius Backing pressure set to 50 psi
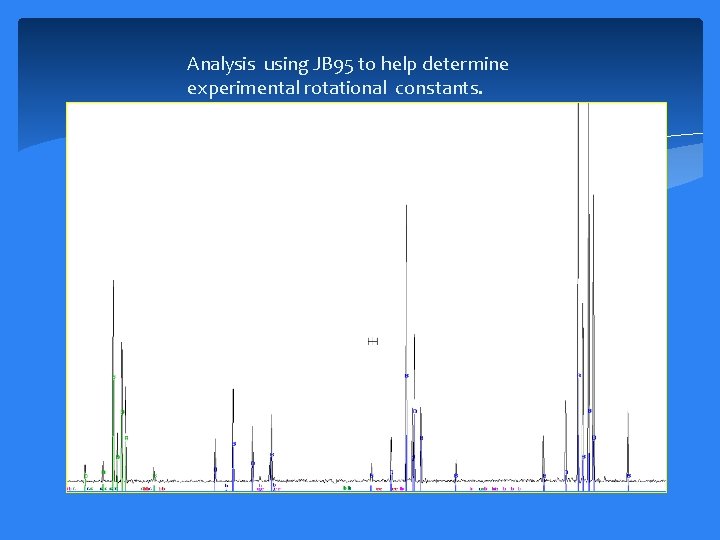
Analysis using JB 95 to help determine experimental rotational constants.
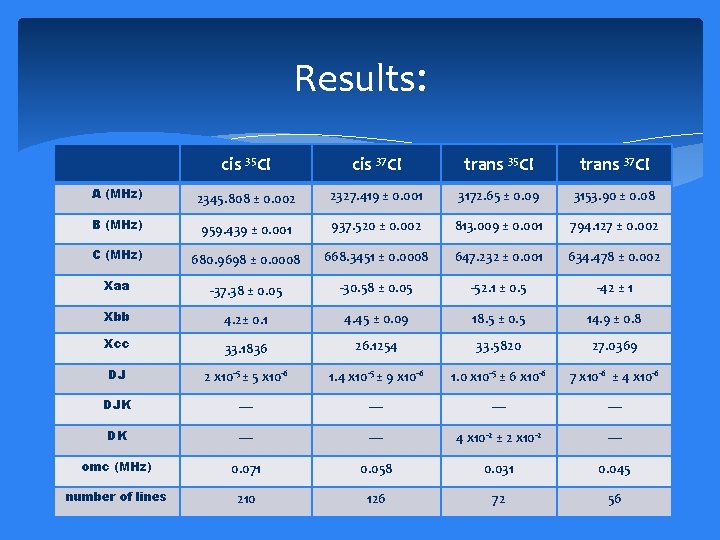
Results: cis 35 Cl cis 37 Cl trans 35 Cl trans 37 Cl A (MHz) 2345. 808 ± 0. 002 2327. 419 ± 0. 001 3172. 65 ± 0. 09 3153. 90 ± 0. 08 B (MHz) 959. 439 ± 0. 001 937. 520 ± 0. 002 813. 009 ± 0. 001 794. 127 ± 0. 002 C (MHz) 680. 9698 ± 0. 0008 668. 3451 ± 0. 0008 647. 232 ± 0. 001 634. 478 ± 0. 002 Xaa -37. 38 ± 0. 05 -30. 58 ± 0. 05 -52. 1 ± 0. 5 -42 ± 1 Xbb 4. 2± 0. 1 4. 45 ± 0. 09 18. 5 ± 0. 5 14. 9 ± 0. 8 Xcc 33. 1836 26. 1254 33. 5820 27. 0369 DJ 2 x 10 -5 ± 5 x 10 -6 1. 4 x 10 -5 ± 9 x 10 -6 1. 0 x 10 -5 ± 6 x 10 -6 7 x 10 -6 ± 4 x 10 -6 DJK — — DK — — 4 x 10 -2 ± 2 x 10 -2 — omc (MHz) 0. 071 0. 058 0. 031 0. 045 number of lines 210 126 72 56
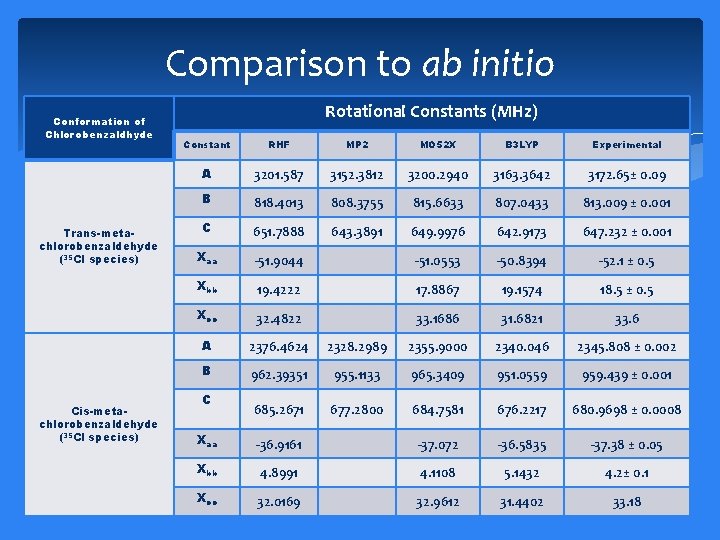
Comparison to ab initio Conformation of Chlorobenzaldhyde Trans-metachlorobenzaldehyde (35 Cl species) Cis-metachlorobenzaldehyde (35 Cl species) Rotational Constants (MHz) Constant RHF MP 2 MO 52 X B 3 LYP Experimental A 3201. 587 3152. 3812 3200. 2940 3163. 3642 3172. 65± 0. 09 B 818. 4013 808. 3755 815. 6633 807. 0433 813. 009 ± 0. 001 C 651. 7888 643. 3891 649. 9976 642. 9173 647. 232 ± 0. 001 Xaa -51. 9044 -51. 0553 -50. 8394 -52. 1 ± 0. 5 Xbb 19. 4222 17. 8867 19. 1574 18. 5 ± 0. 5 Xcc 32. 4822 33. 1686 31. 6821 33. 6 A 2376. 4624 2328. 2989 2355. 9000 2340. 046 2345. 808 ± 0. 002 B 962. 39351 955. 1133 965. 3409 951. 0559 959. 439 ± 0. 001 685. 2671 677. 2800 684. 7581 676. 2217 680. 9698 ± 0. 0008 C Xaa -36. 9161 -37. 072 -36. 5835 -37. 38 ± 0. 05 Xbb 4. 8991 4. 1108 5. 1432 4. 2± 0. 1 Xcc 32. 0169 32. 9612 31. 4402 33. 18
Conclusions: trans-meta-chlorobenzaldehyde cis-meta-chlorobenzaldehyde Measured 8 -18. 5 GHz spectrum of meta-chlorobenzaldehyde Determined experimental rotational, distortion, and quadrupole coupling constants. Compared constants to ab-initio calculations

Acknowledgements American Chemical Society-Petroleum Research Fund (PRF #50746 -UN 16) Coker College National Science Foundation (CHE-1213560)
- Slides: 13