Microwave Spectroscopy or Rotational Spectroscopy PHYSICAL Chemistry Course





- Slides: 15

Microwave Spectroscopy or Rotational Spectroscopy PHYSICAL Chemistry Course: CH-361 Dr. P. R. PATIL

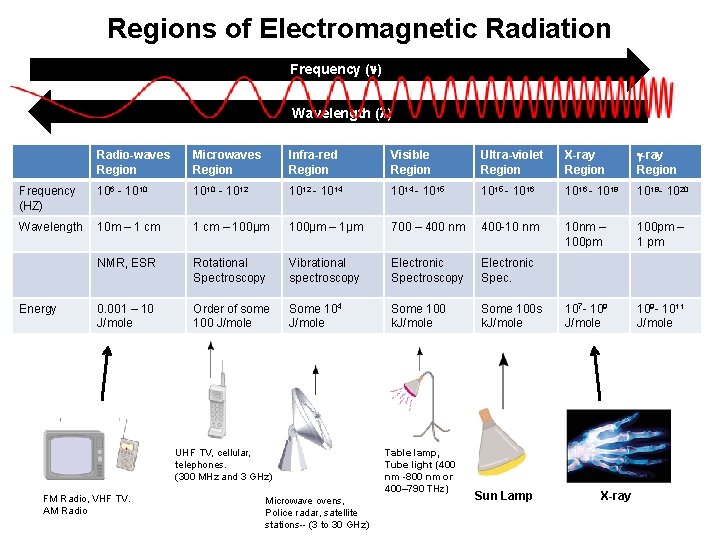
Regions of Electromagnetic Radiation Frequency ( ) Wavelength ( ) Radio-waves Region Microwaves Region Infra-red Region Visible Region Ultra-violet Region X-ray Region Frequency (HZ) 106 - 1010 - 1012 - 1014 - 1015 - 1016 - 1018 - 1020 Wavelength 10 m – 1 cm – 100µm – 1µm 700 – 400 nm 400 -10 nm 10 nm – 100 pm – 1 pm NMR, ESR Rotational Spectroscopy Vibrational spectroscopy Electronic Spec. 0. 001 – 10 J/mole Order of some 100 J/mole Some 104 J/mole Some 100 k. J/mole Some 100 s k. J/mole 107 - 109 J/mole 109 - 1011 J/mole Energy UHF TV, cellular, telephones. (300 MHz and 3 GHz) FM Radio, VHF TV. AM Radio Microwave ovens, Police radar, satellite stations-- (3 to 30 GHz) Table lamp, Tube light (400 nm -800 nm or 400– 790 THz) Sun Lamp X-ray

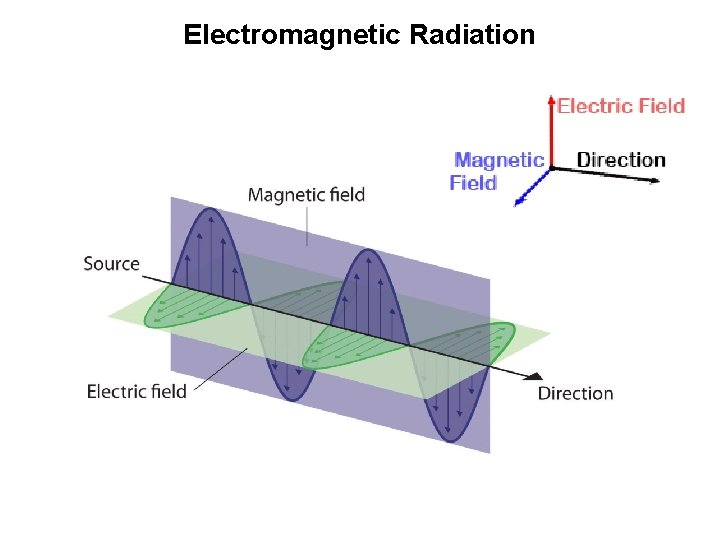
Electromagnetic Radiation

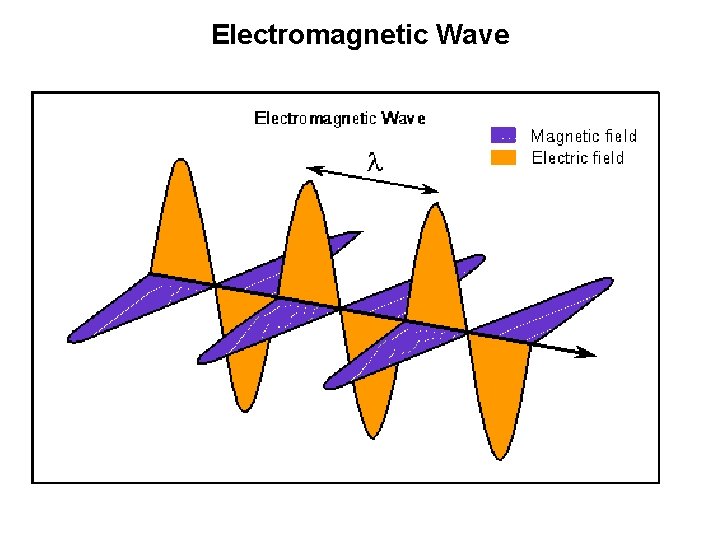
Electromagnetic Wave

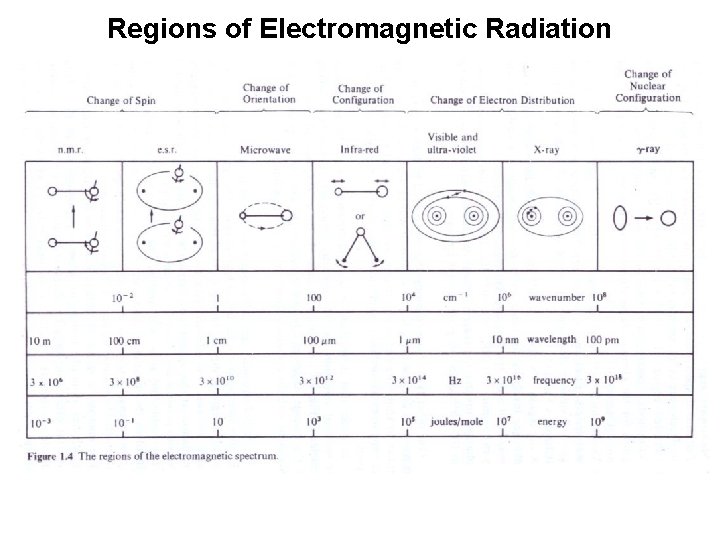
Regions of Electromagnetic Radiation

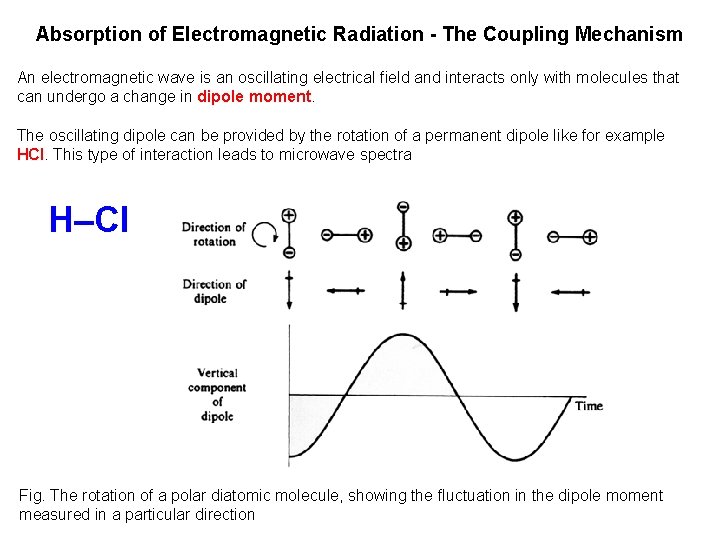
Absorption of Electromagnetic Radiation - The Coupling Mechanism An electromagnetic wave is an oscillating electrical field and interacts only with molecules that can undergo a change in dipole moment. The oscillating dipole can be provided by the rotation of a permanent dipole like for example HCl. This type of interaction leads to microwave spectra H Cl Fig. The rotation of a polar diatomic molecule, showing the fluctuation in the dipole moment measured in a particular direction

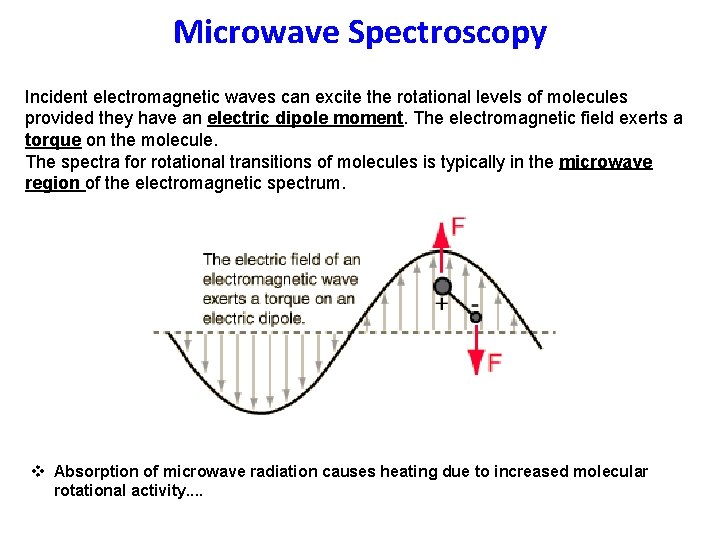
Microwave Spectroscopy Incident electromagnetic waves can excite the rotational levels of molecules provided they have an electric dipole moment. The electromagnetic field exerts a torque on the molecule. The spectra for rotational transitions of molecules is typically in the microwave region of the electromagnetic spectrum. v Absorption of microwave radiation causes heating due to increased molecular rotational activity. .

Microwave Spectroscopy Incident electromagnetic waves can excite the rotational levels of molecules provided they have an electric dipole moment. The electromagnetic field exerts a torque on the molecule. v Homonuclear diatomic molecules (such as H 2, O 2, N 2 , Cl 2) – have zero dipole (non polar) -- have zero change of dipole during the rotation – hence NO interaction with radiation -- hence homonuclear diatomic molecules are microwave inactive v Heteronuclear diatomic molecules (such as HCl, HF, CO) – have permanent dipolemoment (polar compound) -- change of dipole occurs during the rotation – hence interaction with radiation takes place – Therefore, heteronuclear diatomic molecules are microwave active.

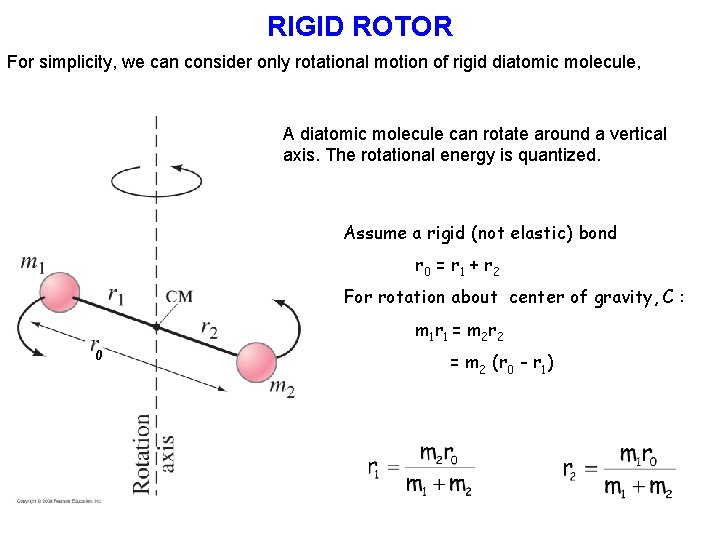
RIGID ROTOR For simplicity, we can consider only rotational motion of rigid diatomic molecule, A diatomic molecule can rotate around a vertical axis. The rotational energy is quantized. Assume a rigid (not elastic) bond r 0 = r 1 + r 2 For rotation about center of gravity, C : 0 m 1 r 1 = m 2 r 2 = m 2 (r 0 - r 1)

RIGID ROTOR Moment of inertia about C: IC = m 1 r 12 + m 2 r 22 = m 2 r 2 r 1 + m 1 r 1 r 2 = r 1 r 2 (m 1 + m 2) = reduced mass, A diatomic molecule can rotate around a vertical axis. The rotational energy is quantized. By the using Schrödinger equation, the rotational energy levels allowed to the rigid diatomic molecule are given by, J = Rotational quantum number (J = 0, 1, 2, …) I = Moment of inertia = mr 2 = reduced mass = m 1 m 2 / (m 1 + m 2) r = internuclear distance

Energy is quantized Planck suggests that radiation (light, energy) can only come in quantized packets that are of size hν. Planck, 1900 Planck’s constant h = 6. 626 × 10 -34 J·s Energy (J) Frequency (s-1) Note that we can specify the energy by specifying any one of the following: 1. The frequency, n (units: Hz or s-1): 2. The wavelength, λ, (units: m or cm or mm): Recall: 3. The wavenumber, Recall: (units: cm-1 or m-1)

Rotational Spectra of Rigid Diatomic molecule Rotational Energy Levels for rigid rotor: Where

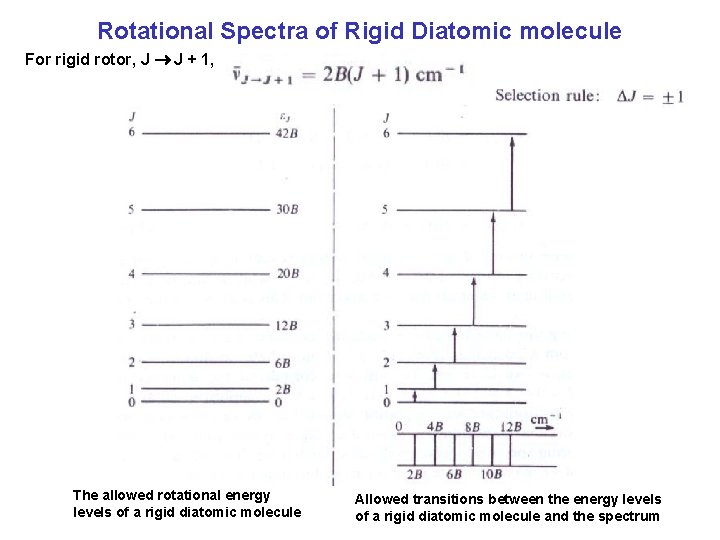
Rotational Spectra of Rigid Diatomic molecule For rigid rotor, J J + 1, The allowed rotational energy levels of a rigid diatomic molecule Allowed transitions between the energy levels of a rigid diatomic molecule and the spectrum

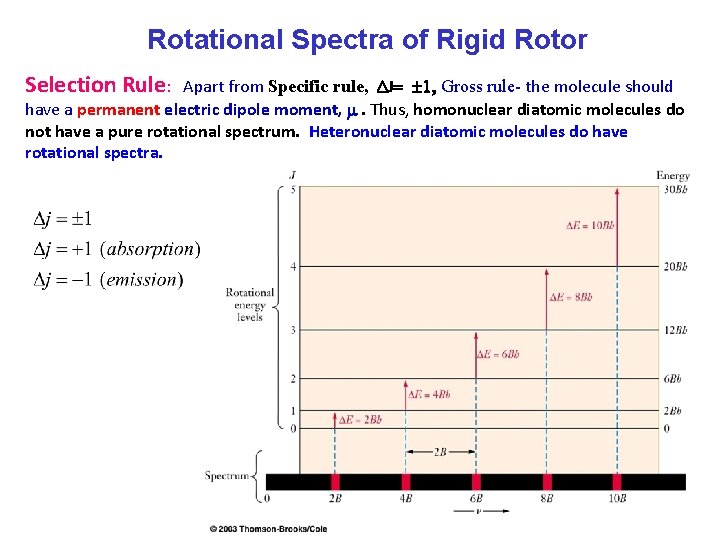
Rotational Spectra of Rigid Rotor Selection Rule: Apart from Specific rule, DJ= 1, Gross rule- the molecule should have a permanent electric dipole moment, m. Thus, homonuclear diatomic molecules do not have a pure rotational spectrum. Heteronuclear diatomic molecules do have rotational spectra.

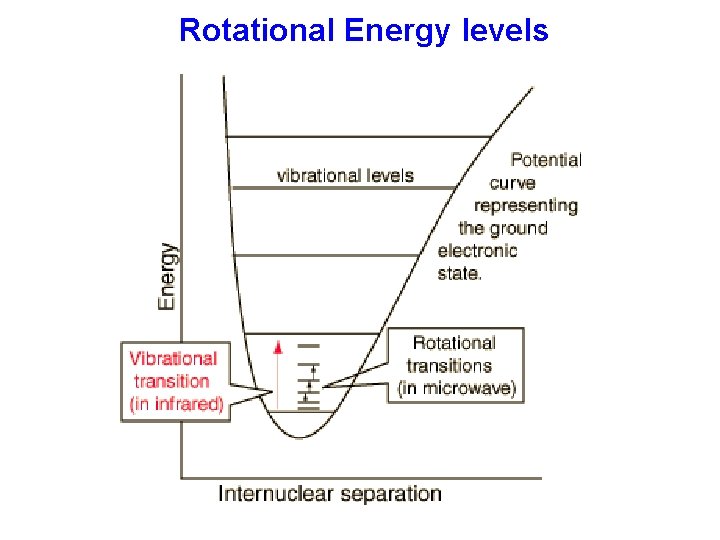
Rotational Energy levels