Microwave Spectroscopy Measurements of the gas phase structure



![Measured and calculated rotational transitions for C 5 H 5184 W(CO)3 H [in MHz]. Measured and calculated rotational transitions for C 5 H 5184 W(CO)3 H [in MHz].](https://slidetodoc.com/presentation_image_h2/144ce502acd1b0bb3a1b9cab95ba6432/image-6.jpg)
![Observed frequency transition [ 651 550 ] for WH showing four different peaks deriving Observed frequency transition [ 651 550 ] for WH showing four different peaks deriving](https://slidetodoc.com/presentation_image_h2/144ce502acd1b0bb3a1b9cab95ba6432/image-8.jpg)


- Slides: 16

Microwave Spectroscopy Measurements of the gas phase structure of Cyclopentadienyltungstentricarbonyl hydride Chandana Karunatilaka, Chakree Tanjaroon, Kristen S. Keck, & Stephen G. Kukolich. Department of Chemistry The University of Arizona.

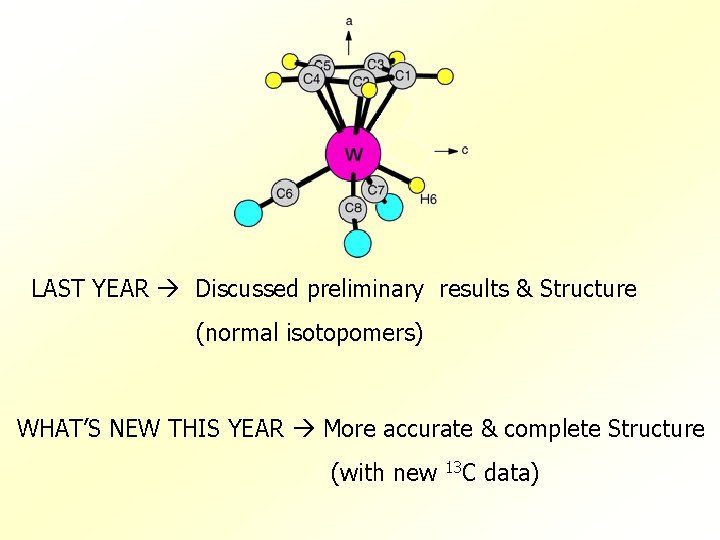
LAST YEAR Discussed preliminary results & Structure (normal isotopomers) WHAT’S NEW THIS YEAR More accurate & complete Structure (with new 13 C data)

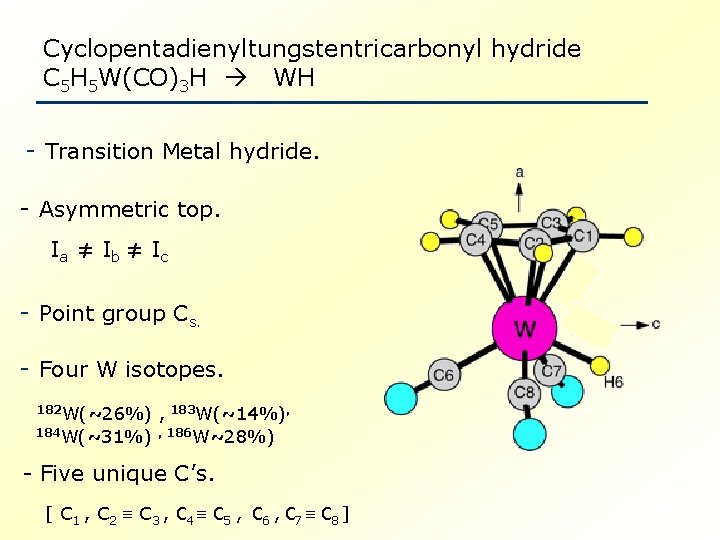
Cyclopentadienyltungstentricarbonyl hydride C 5 H 5 W(CO)3 H WH - Transition Metal hydride. - Asymmetric top. Ia ≠ Ib ≠ Ic - Point group Cs. - Four W isotopes. 182 W(~26%) , 183 W(~14%), 184 W(~31%) , 186 W~28%) - Five unique C’s. [ C 1 , C 2 C 3 , C 4 C 5 , C 6 , C 7 C 8 ]

EXPERIMENT - Cp. W(CO)3 H = WH resublimed prior to use. - Synthesized Cp. W(CO)3 D = WD using CH 3 CO 2 D as the deuterating agent. -Heated the sample 60 -65 0 C -Rotational transitions 4 – 12 GHz range using PBFTMW spectrometer.

RESULTS - A total of over 350 a - dipole transitions - Over 60 a - dipole transitions WH & WD 182, 183, 184, 186 W Five unique 13 C isotopomers
![Measured and calculated rotational transitions for C 5 H 5184 WCO3 H in MHz Measured and calculated rotational transitions for C 5 H 5184 W(CO)3 H [in MHz].](https://slidetodoc.com/presentation_image_h2/144ce502acd1b0bb3a1b9cab95ba6432/image-6.jpg)
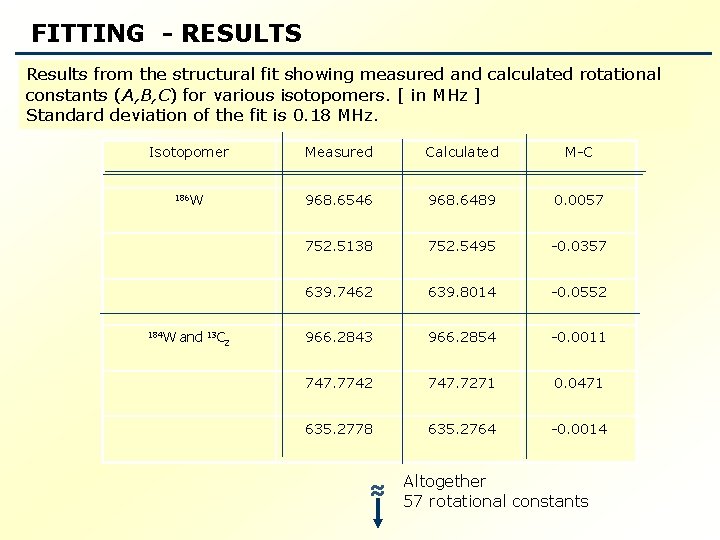
Measured and calculated rotational transitions for C 5 H 5184 W(CO)3 H [in MHz]. The standard deviation of the fit, fit = 3. 9 k. Hz. Isotopomer C 5 H 5184 W(CO)3 H J Ka Kc 2 0 2 2 J’ Ka’ Kc ’ Measured Calculated Residual 3 0 3 4057. 348 4057. 350 -0. 002 1 3 2 2 4176. 853 4176. 851 0. 002 2 0 3 2 1 4296. 352 -0. 001 2 1 1 3 1 2 4320. 501 4320. 499 0. 002 3 2 2 4 2 3 5542. 126 5542. 123 0. 003 3 3 1 4 3 2 5621. 500 0. 002 3 3 0 4 3 1 5659. 270 5659. 271 -0. 001 ≈ Over 350 -transitions

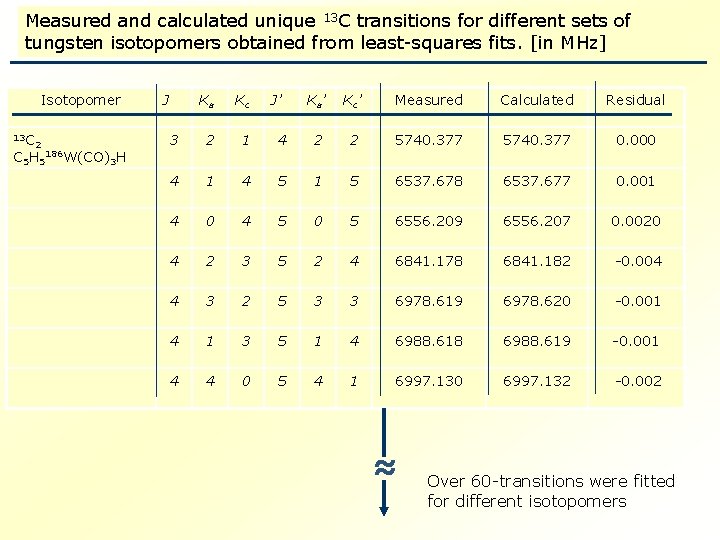
Measured and calculated unique 13 C transitions for different sets of tungsten isotopomers obtained from least-squares fits. [in MHz] Isotopomer 13 C 2 C 5 H 5 186 W(CO) 3 H J Ka Kc 3 2 1 4 J’ Ka’ Kc ’ Measured Calculated Residual 4 2 2 5740. 377 0. 000 4 5 1 5 6537. 678 6537. 677 0. 001 0 4 5 0 5 6556. 209 6556. 207 0. 0020 4 2 3 5 2 4 6841. 178 6841. 182 -0. 004 4 3 2 5 3 3 6978. 619 6978. 620 -0. 001 4 1 3 5 1 4 6988. 618 6988. 619 -0. 001 4 4 0 5 4 1 6997. 130 6997. 132 -0. 002 ≈ Over 60 -transitions were fitted for different isotopomers
![Observed frequency transition 651 550 for WH showing four different peaks deriving Observed frequency transition [ 651 550 ] for WH showing four different peaks deriving](https://slidetodoc.com/presentation_image_h2/144ce502acd1b0bb3a1b9cab95ba6432/image-8.jpg)
Observed frequency transition [ 651 550 ] for WH showing four different peaks deriving from four different tungsten isotopes. 182 W J’ = 651 J = 550 183 W 184 W 186 W

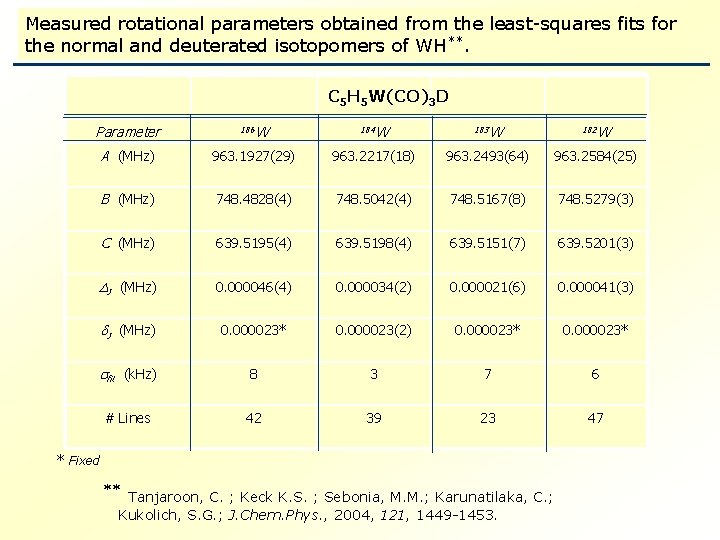
Measured rotational parameters obtained from the least-squares fits for the normal and deuterated isotopomers of WH**. C 5 H 5 W(CO)3 H C 5 H 5 W(CO)3 D Parameter AA (MHz) 186 W W 184 W W 183 W W 182 W W 968. 6540(13) 963. 1927(29) 968. 6999(12) 963. 2217(18) 968. 7242(20) 963. 2493(64) 968. 7454(6) 963. 2584(25) BB (MHz) 752. 5160(3) 748. 4828(4) 752. 5415(4) 748. 5042(4) 752. 5542(8) 748. 5167(8) 752. 5683(2) 748. 5279(3) CC (MHz) 639. 7440(3) 639. 5195(4) 639. 7433(4) 639. 5198(4) 639. 7439(10) 639. 5151(7) 639. 7433(2) 639. 5201(3) JJ (MHz) 0. 000042(3) 0. 000046(4) 0. 000034(2) 0. 000028(4) 0. 000021(6) 0. 000036(1) 0. 000041(3) JJ (MHz) 0. 000015* 0. 000023* 0. 000015(2) 0. 000023(2) 0. 000020(6) 0. 000023* 0. 000015* 0. 000023* fitfit (k. Hz) 88 43 57 46 # # Lines 56 42 54 39 43 23 47 47 * Fixed Tanjaroon, C. ; Keck K. S. ; Sebonia, M. M. ; Karunatilaka, C. ; Kukolich, S. G. ; J. Chem. Phys. , 2004, 121, 1449 -1453.

Kraitchman Analysis & W-H bond length Atomic coordinates in the center of mass (abc) coordinated system obtained from Kraitchman analysis*. Atom a b c H (186 W) 0. 6801(15) 0. 4319(9) (-) 1. 7720(38) H (184 W) 0. 6777(13) 0. 4295(8) (-) 1. 7739(34) H (183 W) 0. 6808(23) 0. 4269(15) (-) 1. 7728(60) H (182 W) 0. 6768(11) 0. 4286(7) (-) 1. 7750(28) H average (+) 0. 6788(16) 0. 0 a (-) 1. 7734(11) W average (+) 0. 0215(45) 0. 0 a (-) 0. 109(23) r W-H (Å) a 1. 79(4) W and H atomic b coordinates are fixed at zero in the fit. * Tanjaroon, C. ; Keck K. S. ; Sebonia, M. M. ; Karunatilaka, C. ; Kukolich, S. G. ; J. Chem. Phys. , 2004, 121, 1449 -1453.

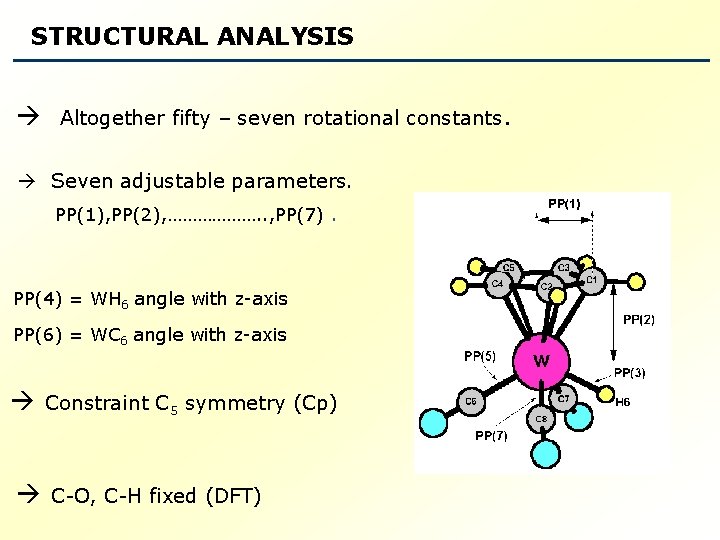
STRUCTURAL ANALYSIS Altogether fifty – seven rotational constants. Seven adjustable parameters. PP(1), PP(2), ………………. . , PP(7). PP(4) = WH 6 angle with z-axis PP(6) = WC 6 angle with z-axis Constraint C 5 symmetry (Cp) C-O, C-H fixed (DFT)

FITTING - RESULTS Results from the structural fit showing measured and calculated rotational constants (A, B, C) for various isotopomers. [ in MHz ] Standard deviation of the fit is 0. 18 MHz. Isotopomer Measured Calculated M-C 186 W 968. 6546 968. 6489 0. 0057 752. 5138 752. 5495 -0. 0357 639. 7462 639. 8014 -0. 0552 966. 2843 966. 2854 -0. 0011 747. 7742 747. 7271 0. 0471 635. 2778 635. 2764 -0. 0014 184 W and 13 C 2 ≈ Altogether 57 rotational constants

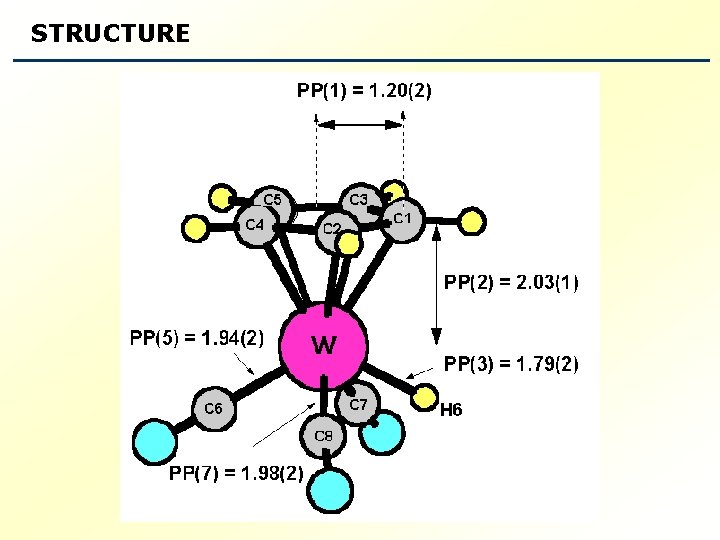
STRUCTURE

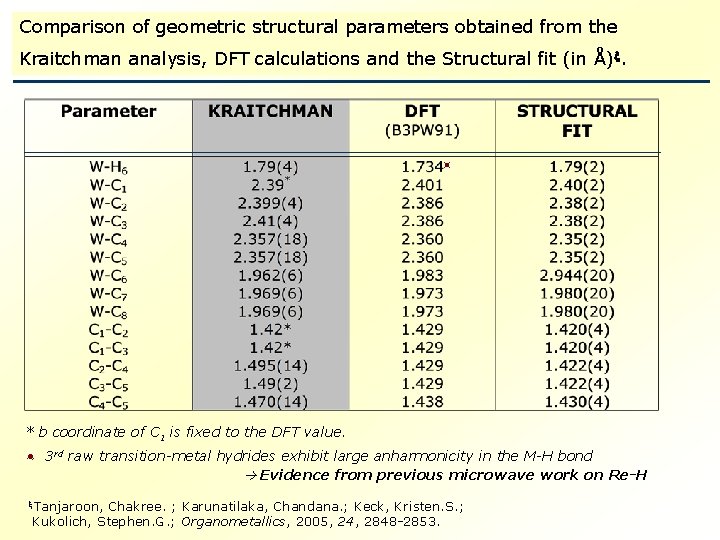
Comparison of geometric structural parameters obtained from the Kraitchman analysis, DFT calculations and the Structural fit (in Å) . * b coordinate of C 1 is fixed to the DFT value. 3 rd raw transition-metal hydrides exhibit large anharmonicity in the M-H bond Evidence from previous microwave work on Re-H Tanjaroon, Chakree. ; Karunatilaka, Chandana. ; Keck, Kristen. S. ; Kukolich, Stephen. G. ; Organometallics, 2005, 24, 2848 -2853.

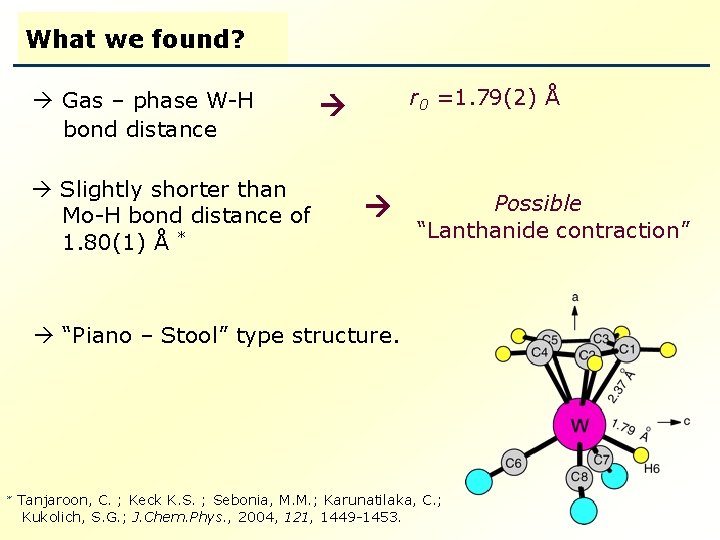
What we found? Gas – phase W-H bond distance Slightly shorter than Mo-H bond distance of 1. 80(1) Å * r 0 =1. 79(2) Å Possible “Lanthanide contraction” “Piano – Stool” type structure. Tanjaroon, C. ; Keck K. S. ; Sebonia, M. M. ; Karunatilaka, C. ; Kukolich, S. G. ; J. Chem. Phys. , 2004, 121, 1449 -1453.

Acknowledgements