Microwave Assisted Organomolybdenum Lewis Acid Catalyzed Mukaiyama Aldol

Microwave Assisted Organomolybdenum Lewis Acid Catalyzed Mukaiyama Aldol Reactions Student : Wanchen Lee Supervisor : Prof. Shuchun Joyce Yu 2005 / 07 / 28 Department of Chemistry & Biochemistry Chung Cheng University 1

Mukaiyama Reaction Silyl enol ether Ketone or Aldehyde Mukaiyama, T. et. al. Chem. Lett. 1973, 1011 -1014 2

Lewis Acid Catalyzed Mukaiyama Reactions I. Traditional Lewis Acids BF 3 O(Et)2、Al. Cl 3、In. Cl 3、Sn. Cl 4、Ti. Cl 4、Fe. Cl 3、ZSM-5 Loh, T. P. et. al. Chem. Commun. 1996, 1819 -1820 Mukaiyama, T. et. al. Chem. Lett. 1973, 1011 -1014 Saigo, K. et. al. Chem. Lett. 1974, 323 -326 Sasidharan, M. et. al. Chem. Lett. 2003, 32, 624 -625 Takeshi, O. et. al. Tetrahedron Lett. 2002, 43, 8959 -8962 II. Organometallic Lewis Acids a. Early Transition Metals: Sc (III)、V(IV)、W(0) 1 -methyl imidazole Chen, C. T. et. al. Synlett. 1999, 816 -818 Kobayashi, S. et. al. Tetrahedron Lett. 1997, 26, 4559 -4562 3

b. Late Transition Metals: Fe (II) 、Ru (II) 、Cu (II) Chem. Commun. 1992, 1634 Bosnich, B. et. al. Tetrahedron Lett. 1992, 39, 5729 -5732 Kobayashi, S. et. al. Tetrahedron 1999, 55, 8739 -8746 Kobayashi, S. et. al. Green Chem. 1999, 4, 175 -177 4

III. Lanthanide Metal Triflate (OTf) Complexes : Yb (III) 、La (III) 、Pr (III) 、Nd (III) 、 Sm (III) 、Eu (III) 、Gd (III) 、Dy (III) 、 Ho (III) 、Er (III) Kobayashi, S. et. al. J. Org. Chem. 1994, 13, 3590 -3596 Kobayashi, S. et. al. Tetrahedron Lett. 1997, 26, 4559 -4562 5

IV. Non-Metallic Catalysts a. PS-Formamide b. Ionic liquids c. Brønsted acid ex: citric acid and benzolic acid d. Lewis base ex: lithium acetate、potassium acetate and sodium acetate Ogawa, C. ; Sugiura, M. ; Kobayashi, S. Chem. Commun. 2003, 192 -193 Chen, S. L. ; Ji, S. J. ; Loh, T. P. Tetrahedron Lett. 2004, 375 -377 Li, G. L. ; Zhao, G. J. Org. Chem. 2005, 70, 4272 -4278 Mukaiyama, T. ; Kawano, Y. ; Fujisawa, H. Chem. Lett. 2005, 34, 88 -89 6

Solvent Systems for Mukaiyama Reactions I. Molecular Organic Solvents CH 2 Cl 2、CH 3 NO 2、CH 3 CN、DMF II. Mixed Solvent System Et. OH-H 2 O、THF-H 2 O III. Green Solvents R. T. Ionic Liquids (Bmim. PF 6) Water 7

Indium Trichloride Catalyzed Mukaiyama Aldol Reaction in Water Loh, T. P. et. al. Chem. Commun. 1996, 1819 -1820 8

Sc(OTf)3 -Catalyzed Aqueous Aldol Reaction in Micellar Systems Kobayashi, S. et. al. Tetrahedron Lett. 1997, 26, 4559 -4562 9

Oxovanadium(IV) Biphenolate Catalyzed Mukaiyama Aldol Reaction 10 mol% L = 1 -methyl imidazole Chen, C. T. et. al. Synlett. 1999, 816 -818 10
![[Ru(salen)(NO)H 2 O]Sb. F 6 Catalyzed Mukaiyama Aldol Reaction Bosnich, B. et. al. Tetrahedron [Ru(salen)(NO)H 2 O]Sb. F 6 Catalyzed Mukaiyama Aldol Reaction Bosnich, B. et. al. Tetrahedron](http://slidetodoc.com/presentation_image/25a8b45cbbe2ed1e547202de00ae9dfd/image-11.jpg)
[Ru(salen)(NO)H 2 O]Sb. F 6 Catalyzed Mukaiyama Aldol Reaction Bosnich, B. et. al. Tetrahedron Lett. 1992, 39, 5729 -5732 11

Lewis Acid-Surfactant Combined Catalyst Systems (10 mol %) 20 mol % Kobayashi, S. et. al. Green Chem. 1999, 4, 175 -177 12

Lanthanide Triflate as Water-Tolerant Lewis Acid Kobayashi, S. et. al. J. Org. Chem. 1994, 13, 3590 -3596 13

Polystyrene Supported Formamide Catalyzed Mukaiyama Aldol Reaction Ogawa, C. ; Sugiura, M. ; Kobayashi, S. Chem. Commun. 2003, 192 -193 14

Ionic Liquid Catalyzed Mukaiyama Aldol Reaction Loh, T. P. et. al. Tetrahedron Lett. 2004, 45, 375 -377 15

Motivation I. Traditional Lewis acids are difficult to handle II. Lanthanide metals are relatively expensive III. Low Oxidation State Transition Metals a. Relatively high moisture – and oxygen – stability b. Inexpensive c. Tunable electronic and steric environments around metal Lewis acid center IV. Green Chemistry a. Green solvents R. T. ionic liquids (Bmim. PF 6) and H 2 O b. Energy saving Catalysis under microwave irradiation 16

Preparation of Organomolybdenum Catalyst Î Thermal Conditions 17

Î Microwave Flash Heating Conditions 18

Crotonaldehyde-Lewis Acid Adduct Childs, R. F. et. al. Can. J. Chem. 1982, 60, 801 19
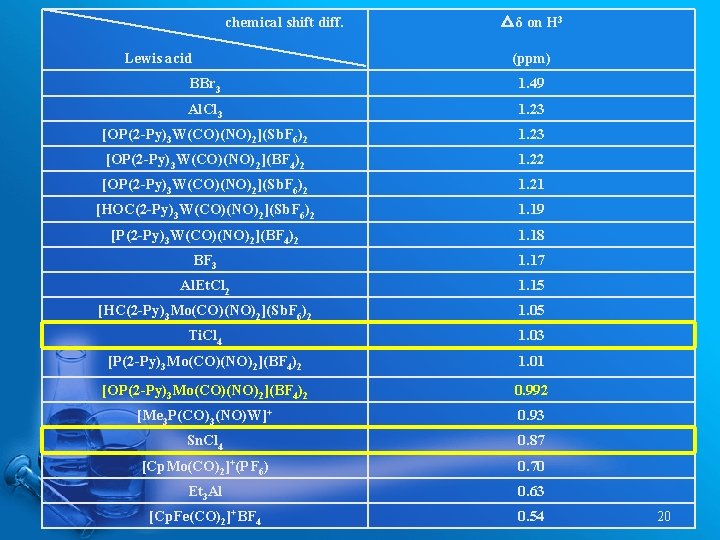
chemical shift diff. Lewis acid △δ on H 3 (ppm) BBr 3 1. 49 Al. Cl 3 1. 23 [OP(2 -Py)3 W(CO)(NO)2](Sb. F 6)2 1. 23 [OP(2 -Py)3 W(CO)(NO)2](BF 4)2 1. 22 [OP(2 -Py)3 W(CO)(NO)2](Sb. F 6)2 1. 21 [HOC(2 -Py)3 W(CO)(NO)2](Sb. F 6)2 1. 19 [P(2 -Py)3 W(CO)(NO)2](BF 4)2 1. 18 BF 3 1. 17 Al. Et. Cl 2 1. 15 [HC(2 -Py)3 Mo(CO)(NO)2](Sb. F 6)2 1. 05 Ti. Cl 4 1. 03 [P(2 -Py)3 Mo(CO)(NO)2](BF 4)2 1. 01 [OP(2 -Py)3 Mo(CO)(NO)2](BF 4)2 0. 992 [Me 3 P(CO)3(NO)W]+ 0. 93 Sn. Cl 4 0. 87 [Cp. Mo(CO)2]+(PF 6) 0. 70 Et 3 Al 0. 63 [Cp. Fe(CO)2]+BF 4 0. 54 20

Organomolybdenum Lewis Acid Catalyzed Mukaiyama Aldol Reactions under Thermal Conditions 21
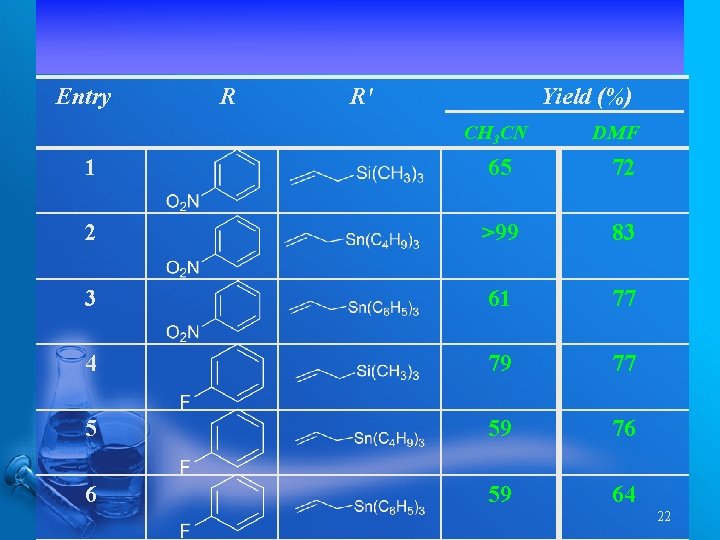
Entry R R' Yield (%) CH 3 CN DMF 1 65 72 2 >99 83 3 61 77 4 79 77 5 59 76 6 59 64 22
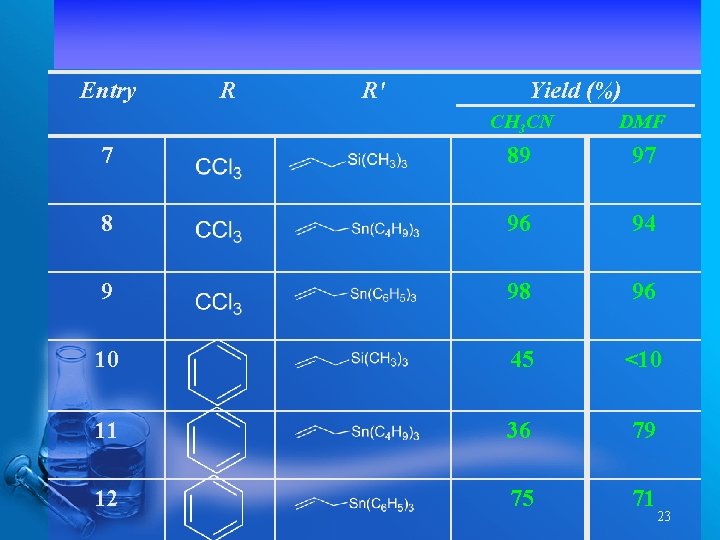
Entry R R' Yield (%) CH 3 CN DMF 7 89 97 8 96 94 9 98 96 10 45 <10 11 36 79 12 75 71 23
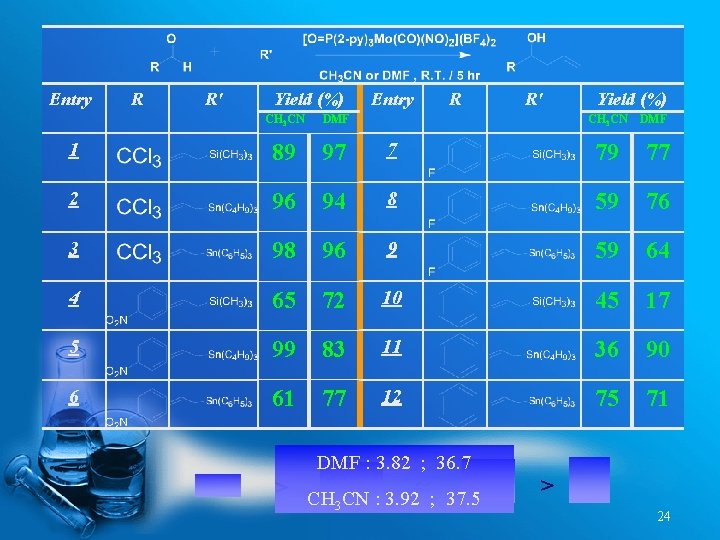
Entry R R' Yield (%) CH 3 CN DMF CH 3 CN 1 89 97 7 79 77 2 96 94 8 59 76 3 98 96 9 59 64 4 65 72 10 45 17 5 99 83 11 36 90 6 61 77 12 75 71 DMF : 3. 82 ; 36. 7 > CH CN : 3. 92≈ ; 37. 5 3 DMF > 24
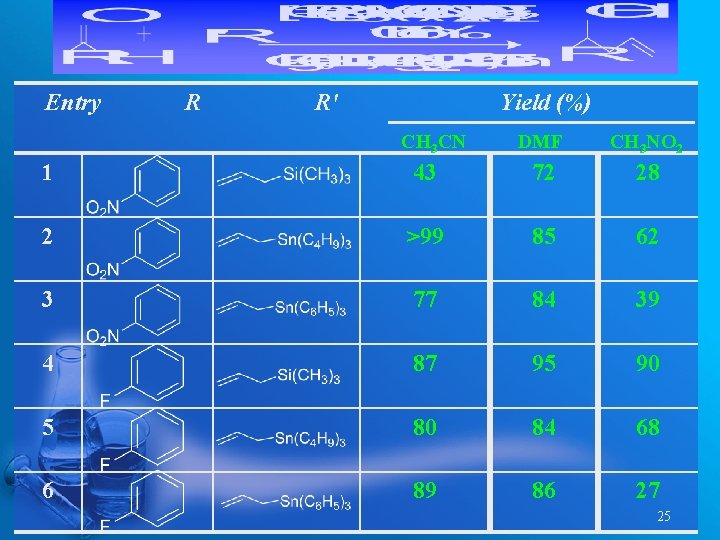
Entry R R' Yield (%) CH 3 CN DMF CH 3 NO 2 1 43 72 28 2 >99 85 62 3 77 84 39 4 87 95 90 5 80 84 68 6 89 86 27 25
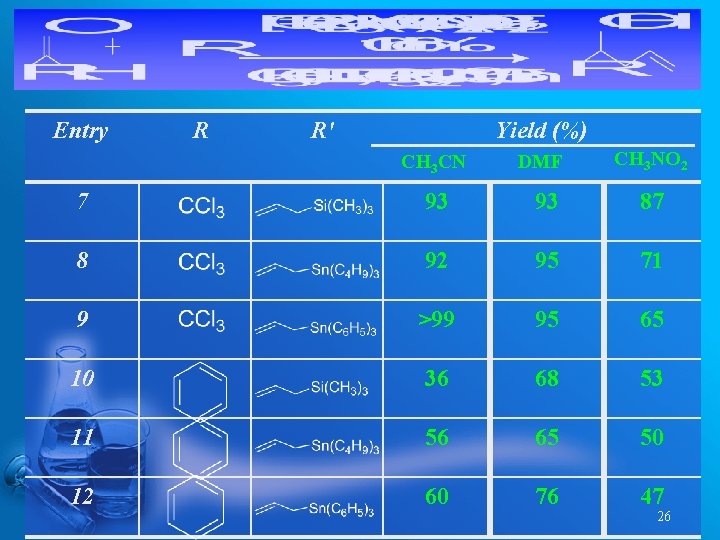
Entry R R' Yield (%) CH 3 CN DMF CH 3 NO 2 7 93 93 87 8 92 95 71 9 >99 95 65 10 36 68 53 11 56 65 50 12 60 76 47 26
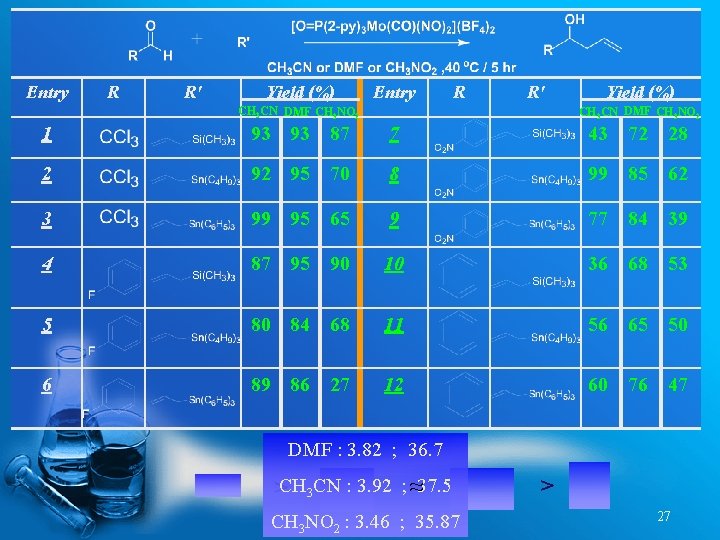
Entry R R' Yield (%) Entry R R' CH 3 CN DMF CH 3 NO 2 Yield (%) CH 3 CN DMF CH 3 NO 2 1 93 93 87 7 43 72 28 2 92 95 70 8 99 85 62 3 99 95 65 9 77 84 39 4 87 95 90 10 36 68 53 5 80 84 68 11 56 65 50 6 89 86 27 12 60 76 47 DMF : 3. 82 ; 36. 7 >CH 3 CN : 3. 92 ; ≈37. 5 CH 3 NO 2 : 3. 46 ; 35. 87 > 27
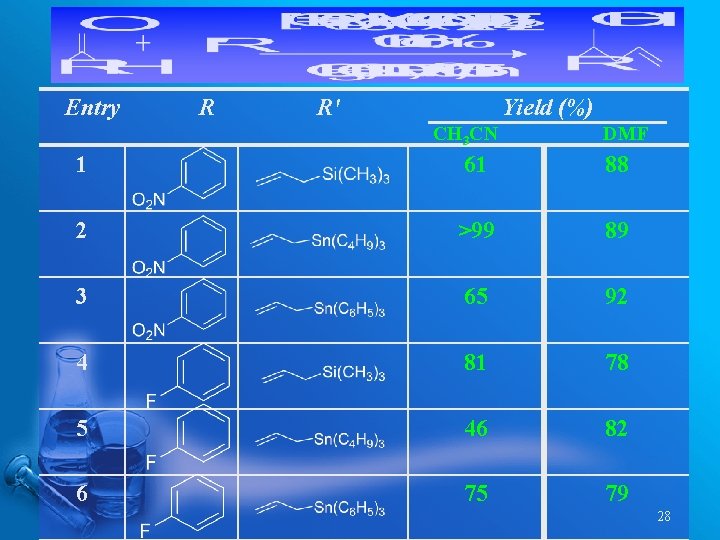
Entry R R' Yield (%) CH 3 CN DMF 1 61 88 2 >99 89 3 65 92 4 81 78 5 46 82 6 75 79 28
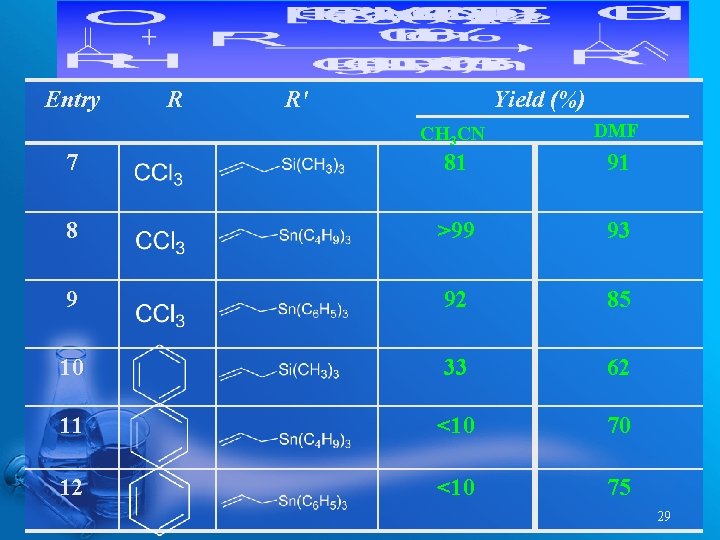
Entry R R' Yield (%) CH 3 CN DMF 7 81 91 8 >99 93 9 92 85 10 33 62 11 <10 70 12 <10 75 29
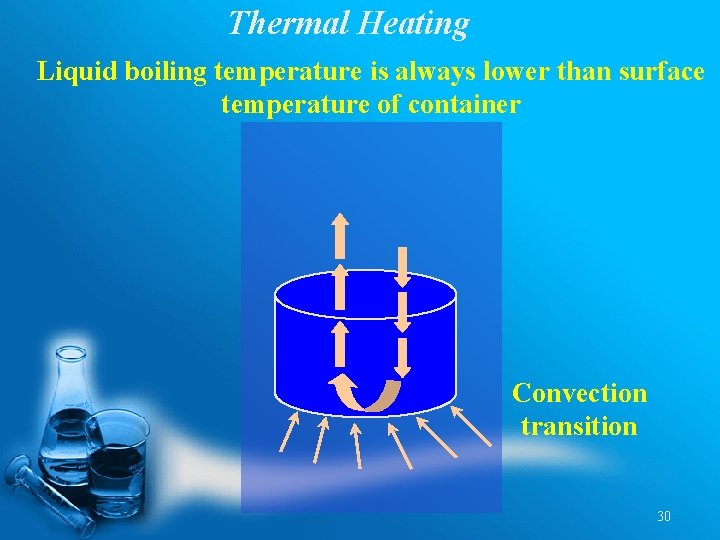
Thermal Heating Liquid boiling temperature is always lower than surface temperature of container Convection transition 30
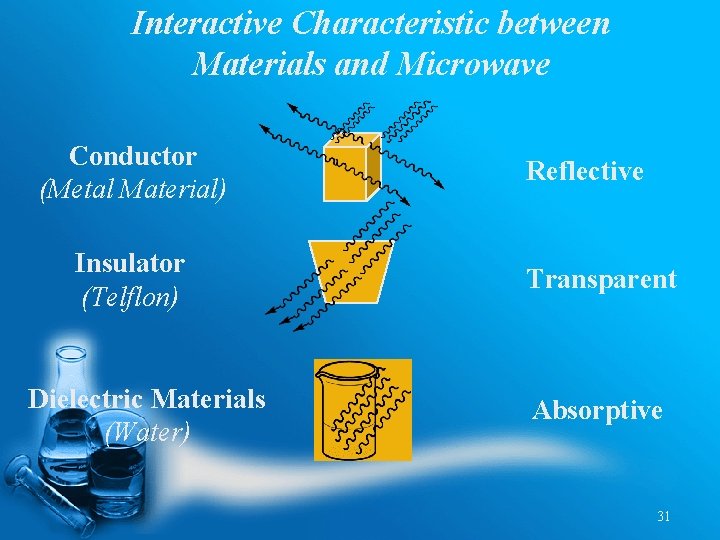
Interactive Characteristic between Materials and Microwave Conductor (Metal Material) Insulator (Telflon) Dielectric Materials (Water) Reflective Transparent Absorptive 31
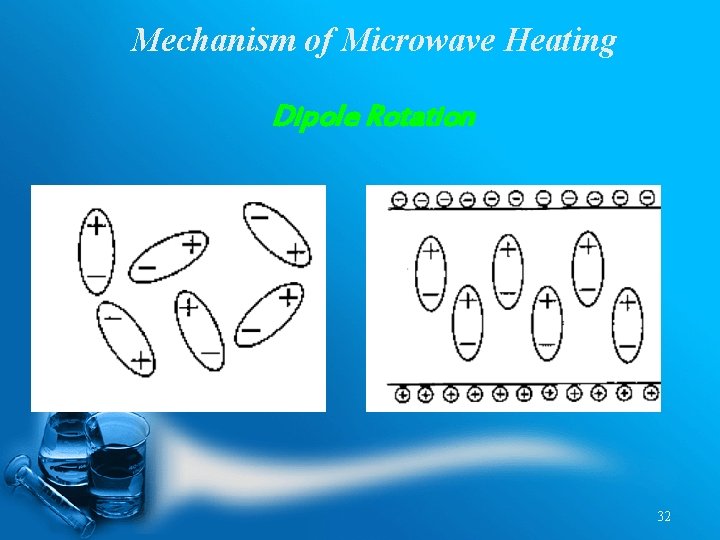
Mechanism of Microwave Heating Dipole Rotation 32
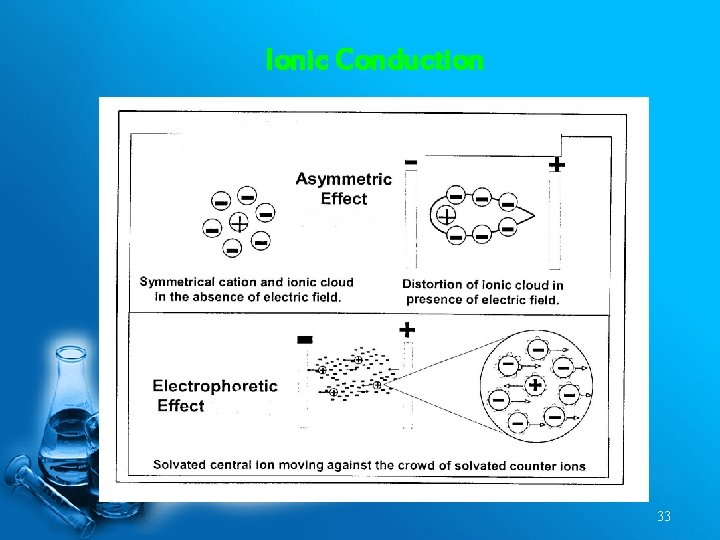
Ionic Conduction 33
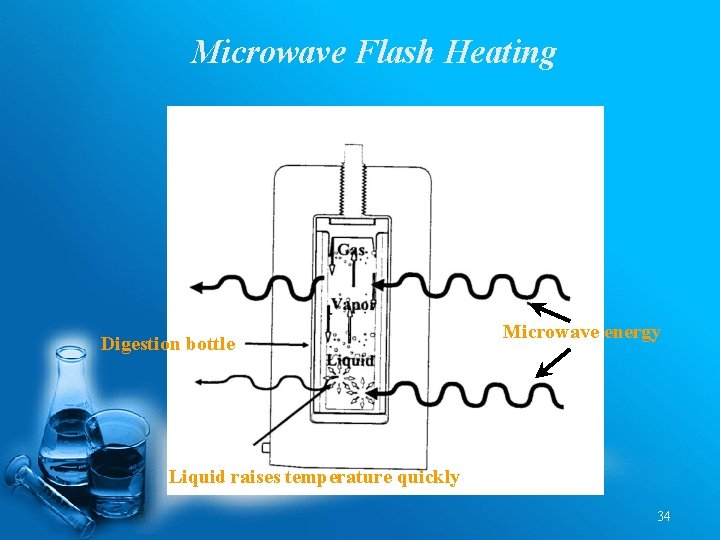
Microwave Flash Heating Digestion bottle Microwave energy Liquid raises temperature quickly 34

Organomolybdenum Lewis Acid Catalyzed Mukaiyama Aldol Reactions under Microwave Irradiation Conditions 35

Ionic Liquids Seddon, K. R. et. al. Pure Appl. Chem. 2000, 72, 2275– 2287 36
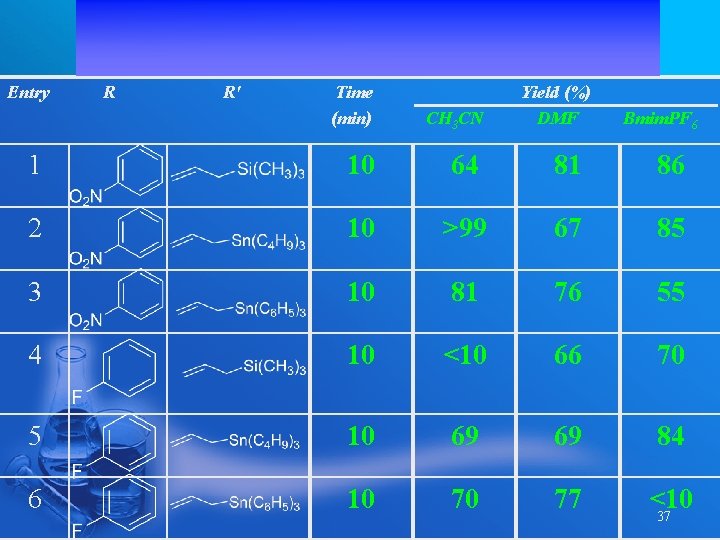
Entry R R' Time (min) Yield (%) DMF CH 3 CN 1 10 64 81 86 2 10 >99 67 85 3 10 81 76 55 4 10 <10 66 70 5 10 69 69 84 6 10 70 77 <10 37 Bmim. PF 6
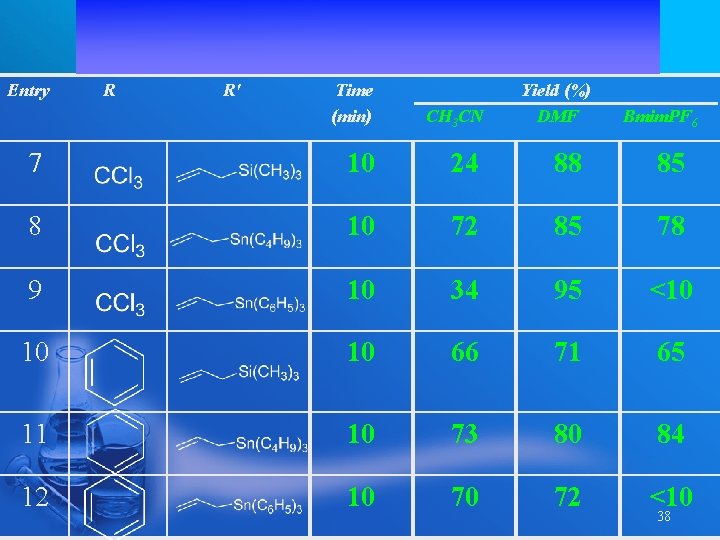
Entry R R' Time (min) Yield (%) DMF CH 3 CN 7 10 24 88 85 8 10 72 85 78 9 10 34 95 <10 10 10 66 71 65 11 10 73 80 84 12 10 70 72 <10 Bmim. PF 6 38
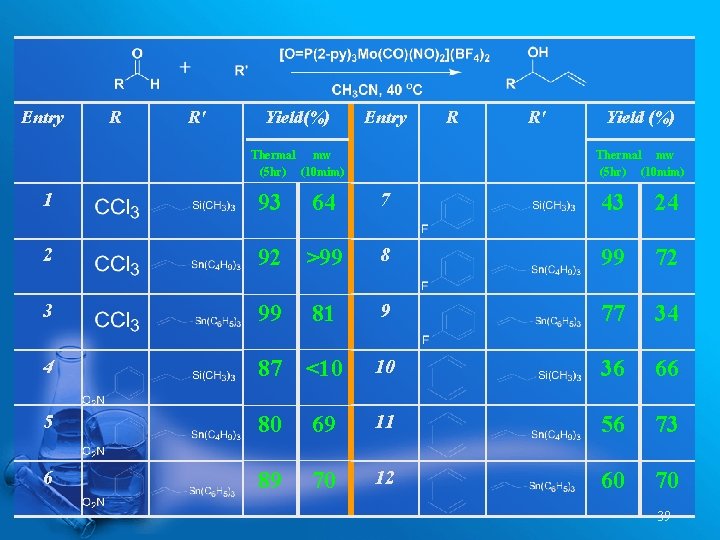
Entry R R' Yield(%) Entry Thermal mw (5 hr) (10 mim) 1 93 2 R R' Yield (%) Thermal mw (5 hr) (10 mim) 7 43 24 92 >99 8 99 72 3 99 9 77 34 4 87 <10 10 36 66 5 80 69 11 56 73 6 89 70 12 60 70 64 81 39
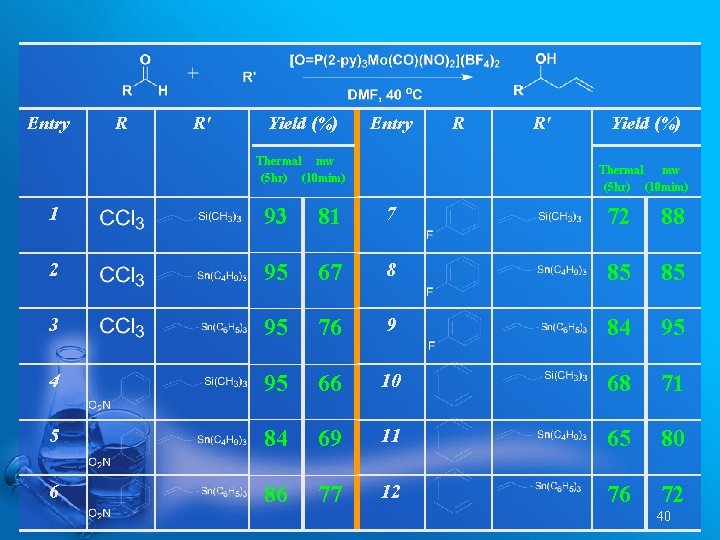
Entry R R' Yield (%) Entry Thermal mw (5 hr) (10 mim) R R' Yield (%) Thermal mw (5 hr) (10 mim) 1 93 81 7 72 88 2 95 67 8 85 85 3 95 76 9 84 95 66 10 68 71 5 84 69 11 65 80 6 86 77 12 76 72 40
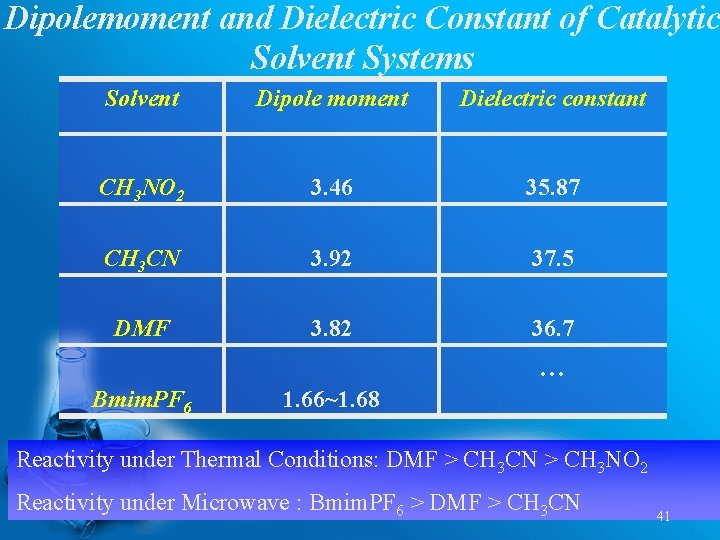
Dipolemoment and Dielectric Constant of Catalytic Solvent Systems Solvent Dipole moment Dielectric constant CH 3 NO 2 3. 46 35. 87 CH 3 CN 3. 92 37. 5 DMF 3. 82 36. 7 … Bmim. PF 6 1. 66~1. 68 Reactivity under Thermal Conditions: DMF > CH 3 CN > CH 3 NO 2 Reactivity under Microwave : Bmim. PF 6 > DMF > CH 3 CN 41

Proposed Mechanism σ-donation 42

43
![Catalytic Reactivity of [A(2 -py)3 M(CO)(NO)2]2+ on Mukaiyama Aldol Reaction Catalysts Yield (%) A(2 Catalytic Reactivity of [A(2 -py)3 M(CO)(NO)2]2+ on Mukaiyama Aldol Reaction Catalysts Yield (%) A(2](http://slidetodoc.com/presentation_image/25a8b45cbbe2ed1e547202de00ae9dfd/image-44.jpg)
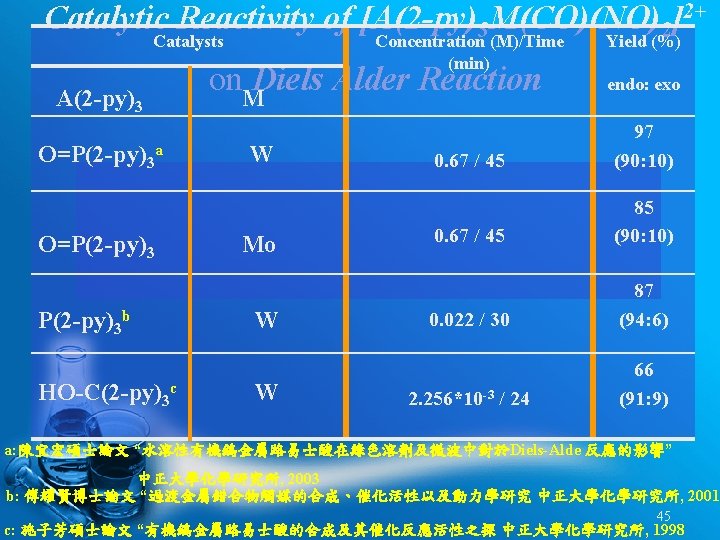
Catalytic Reactivity of [A(2 -py)3 M(CO)(NO)2]2+ on Mukaiyama Aldol Reaction Catalysts Yield (%) A(2 -py)3 M O=P(2 -py)3 Mo 93 P(2 -py)3 O=P(2 -py)3 Mo W 85 56 P(2 -py)3 W 45 44

- Slides: 46