MICROWAVE ASSISTED EXTRACTION KHADIJAH HANIM BT ABDUL RAHMAN

MICROWAVE- ASSISTED EXTRACTION KHADIJAH HANIM BT ABDUL RAHMAN Chemical Engineering Technology Department, FETech, Uni. MAP SEM II, 2018/2019

Microwave-assisted Extraction (MAE): INTRODUCTION
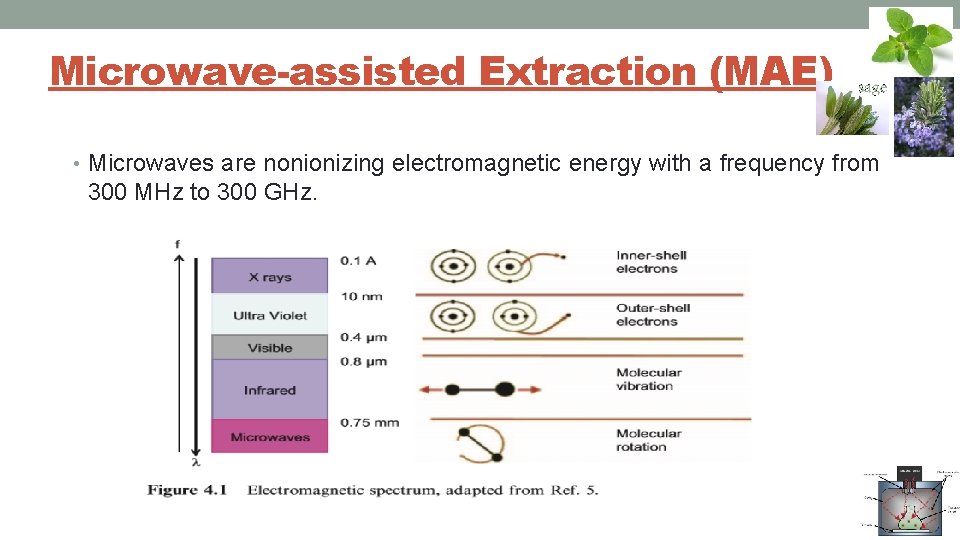
Microwave-assisted Extraction (MAE) • Microwaves are nonionizing electromagnetic energy with a frequency from 300 MHz to 300 GHz.

Microwave-assisted Extraction (MAE) • This energy is transmitted as waves, which can penetrate in biomaterials and interact with polar molecules inside the materials, such as water, to generate heat. • MAE is a process that uses the effect of microwaves to extract biological materials. • Materials that absorb the microwave energy are easily heated, such as polar liquids, are named dielectrics. • The physical principle of this technique is based on the ability of polar chemical compounds to absorb microwave energy according to its nature

Microwave-assisted Extraction (MAE) • The water/solvent contained in the solid matrix is responsible for the absorption of microwave energy. • Microwaves cause molecular motion by migration of ions and rotation of dipoles, and by solvent heating and improves its penetration. • The effect of microwaves in the material is strongly dependent on the dielectric susceptibility of both the solvent and the solid matrix. • Therefore, the material undergoes internal superheating. • As a result, the cell structure is disrupted, and the flow out of the chemical constituents from the solid matrix is facilitated. • The phenomenology of this process is quite different from the conventional solvent extraction where the solvent diffuses in the solid matrix and dissolves the compounds.
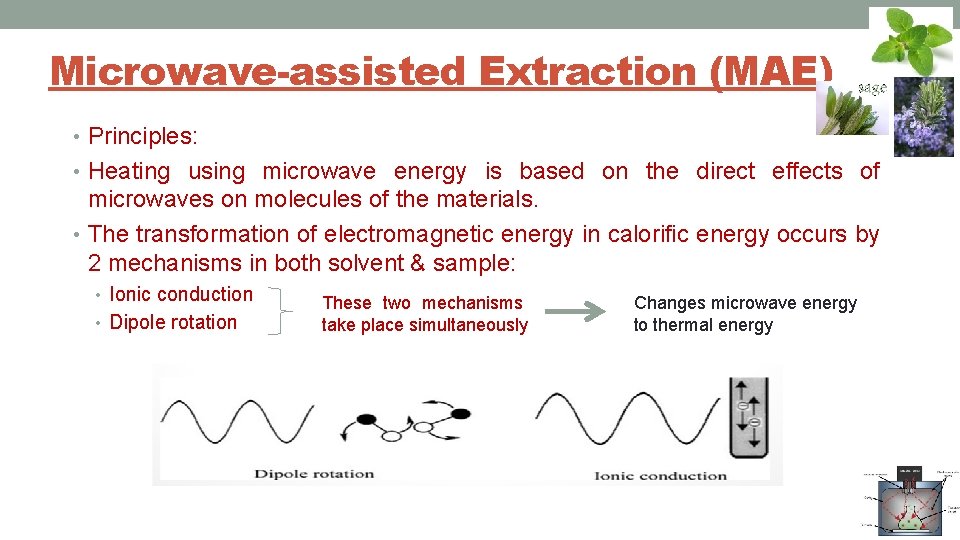
Microwave-assisted Extraction (MAE) • Principles: • Heating using microwave energy is based on the direct effects of microwaves on molecules of the materials. • The transformation of electromagnetic energy in calorific energy occurs by 2 mechanisms in both solvent & sample: • Ionic conduction • Dipole rotation These two mechanisms take place simultaneously Changes microwave energy to thermal energy

MAE Principles: Ionic conduction • Due to the electrophoretic migration of ions when electromagnetic field is applied • The resistance of the solution to this flow of ions & the collisions between molecules because the direction of ions changes as many times as the field changes sign will result in friction, thus heat the solution. • The migration of dissolved ions increases solvent penetration into the matrix & thus facilitates the solvation of target compounds.

MAE Principles: Dipole rotation • Related to alternative movement of polar molecules that have dipole moments which try to line up with the electric field. • Figure 4. 2 - schematic behavior of dipolar molecules without an electric field & under continuous or high frequency electric fields. • The larger the dipole moment of the molecule, the more vigorous is the oscillation in the microwave field. • This dipole rotation leads to the disruption of weak hydrogen bonds.
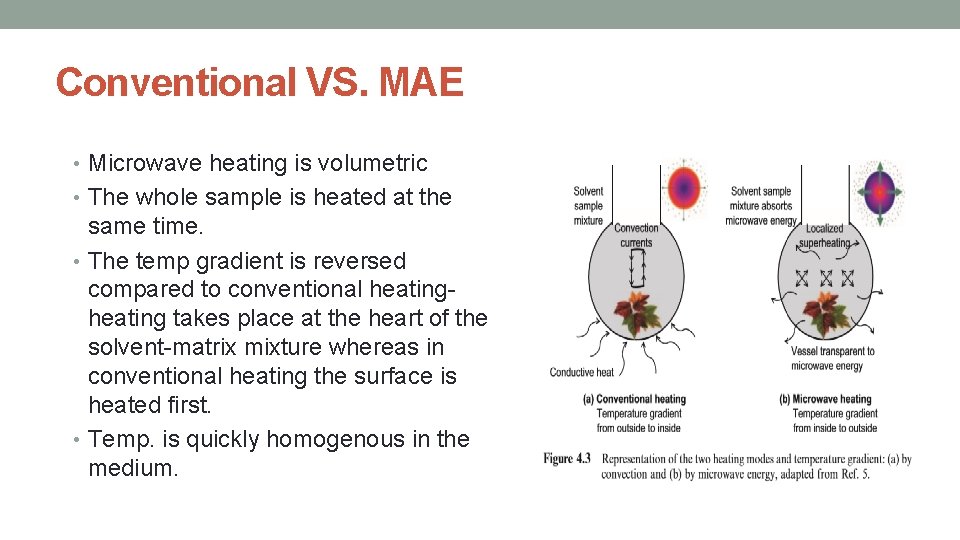
Conventional VS. MAE • Microwave heating is volumetric • The whole sample is heated at the same time. • The temp gradient is reversed compared to conventional heating- heating takes place at the heart of the solvent-matrix mixture whereas in conventional heating the surface is heated first. • Temp. is quickly homogenous in the medium.

Microwave-assisted Extraction (MAE) Principle of microwave-assisted extraction • Dried plants contain a minute microscopic trace of moisture which serves as target for microwave heating. • High temperature and pressure is generated inside the oven. • High temperature causes dehydration of cellulose which accounts for its reduction of mechanical strength. • The MAE process is as follows: • • – Microwave radiation – Moisture get heated up – Moisture evaporates – Generation of tremendous pressure on cell wall – Swelling of plant cell – Rupture of the cell – Leaching out of phyto-constituents • This phenomenon can be intensified if the plant matrix is impregnated with solvents with higher heating efficiency under microwave.

Microwave-assisted Extraction (MAE) Mechanism of microwave-assisted extraction • Microwave is an electromagnetic radiation with a wavelength from 0. 001 m to 1 m (i. e. with a frequency from 3 x • • • 1011 Hz to 3 x 108 Hz), which can be transmitted as the wave. When microwave passes through the medium, its energy may be absorbed and converted into thermal energy. In general, the heating using microwave energy is based on two principles, ionic conduction and dipole rotation. Ionic conduction refers to the electrophoretic migration of the charge carriers (e. g. , ions and electrons) under the influence of the electric field produced by microwave. The resulting “friction” between the flowed ions and the medium is capable of causing heating. Dipole rotation happens when the dipolar molecules attempts to follow the electric field in the same alignment. The induced oscillation leads to collisions between dipoles and surrounding molecules, and thus creates heat. At a frequency of 2. 45 x 109 Hz, which is the most common frequency assigned to the domestic and industrial microwave systems, the phenomenon of alignment, randomization and realignment of dipoles takes place 4. 9 x 109 times per second. Thus the molecular movement is intensified and the heating is triggered. Usually, ionic conduction and dipole rotation occur simultaneously, which effectively change microwave energy to thermal one.

Microwave-assisted Extraction (MAE) Mechanism of microwave-assisted extraction (continued) • During microwave assisted extraction of secondary metabolites from plants, microwave • • rapidly delivers energy to extractant and plant matrix. The energy is efficiently absorbed by some substances inside vegetable materials, especially the polar molecules like water. Consequently, the internal temperature of the plant cells increases drastically. The interior superheating may cause the liquid vaporization within the cells, which may rupture the cell walls and/or plasma membranes. Since a lot of plant secondary metabolites (e. g. , proanthocyanidins) naturally occur in the cell walls or cytoplasm, the cell disruptions can facilitate the mass transfer of solvent into plant materials and secondary metabolites into solvent, thus allowing the effective extraction.

Microwave Heating applied to Plant Matrices • The effect of microwave energy is strongly dependent on the nature of both the solvent and matrix. • Most of the time- solvent selected has a high dielectric constant- so that it strongly absorbs the microwave energy. • However, for some thermolabile compounds- only sample matrix may be heated- so that the solutes are released in a cold solvent. • The forced heating of water in the core of the material may cause liquid vaporization within the cells- lead to rupture of the cell walls/plasma membranes.

Microwave Heating applied to Plant Matrices • This cell disruption can shorten the diffusion path & facilitate the mass transfer of the solvent into plant material & of the secondary metabolites into the solvent- allowing effective extraction. • Different from Soxhlet extraction which depends on a series of permeation & solubilization processes to wash out the intracellular constituents out of the plant matrix.

INSTRUMENTATIONS

Instrumentations • Both monomode & multimode systems comprised of 4 components. • Constitutes of magnetron tube, wave guide, applicator & circulators. • Magnetron tube- generates microwaves at a fixed frequency (ie 2450 MHz). • Wave guide – propagate microwave source to microwave cavity. • Applicator – heat the sample by exposing it to microwave field. • Circulator – allow microwave to move in forward direction.

Open vessel system: Monomode • The wave guide transmits the microwave from the source of cavity. • The sample introduced in a dedicated extraction vessel (reactor) is then placed in the cavity and the circulator is used for reflection and homogenization of radiation. • The monomode cavity can generate a frequency that excites only one mode of resonance. • The sample can be placed in the wave guide where microwaves are focused, on the max of the electrical field, as the distribution of the field is known.


Open vessel in monomode focused microwave oven • Open systems • Open quartz vessels- extraction is made at atmospheric pressure (Focused microwave-assisted extraction (FMAE)). • The maximum possible temperature is determined by the boiling point of the solvent at that pressure. • Losses of vapors are prevented by the presence of a cooling system on the top of extraction vessel that causes condensation of solvent vapors. • The power can be modulated and the field is homogenous & reproducible. - heating of sample is homogenous & very efficient. • Some system can extract up to 100 kg of fresh material.

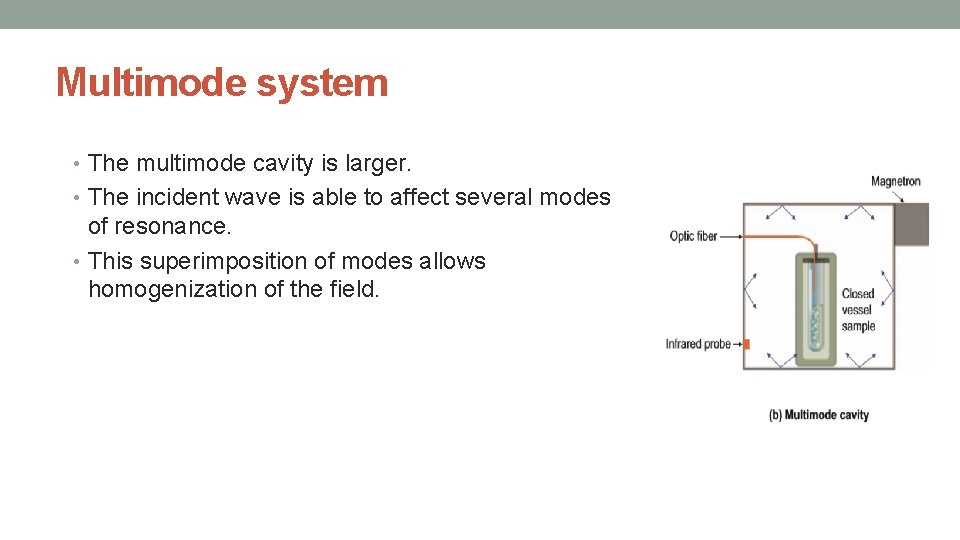
Multimode system • The multimode cavity is larger. • The incident wave is able to affect several modes of resonance. • This superimposition of modes allows homogenization of the field.

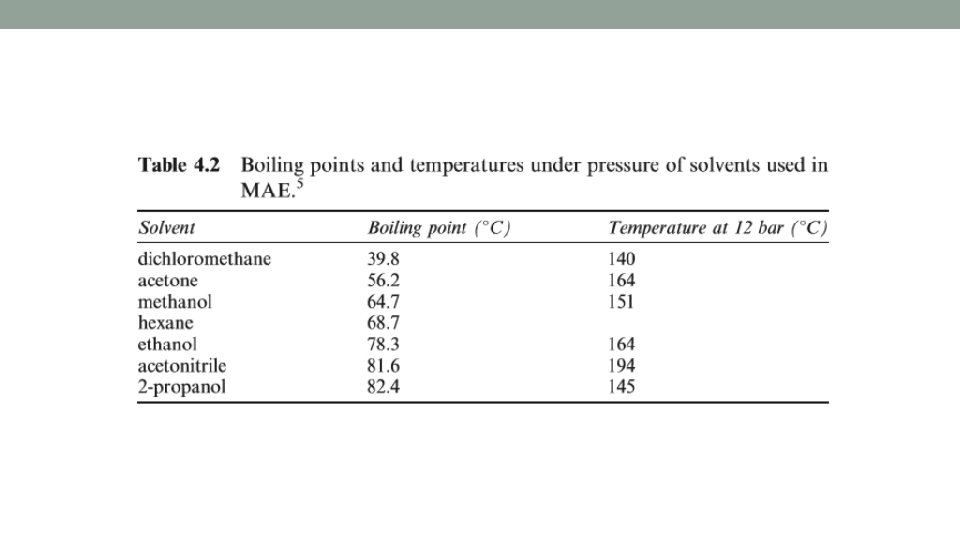
Multimode System: Closed vessel • Closed system • Closed vessel- pressurized microwave-assisted extraction (PMAE) performed under pressure. • Typical pressures reached are below 14 bar, new technology can reach up to 100 bar. • The pressure allows temperatures above boiling points of the solvents to be reached, enhancing both extraction speed & efficiency (Table 4. 2). • Commonly, power, temp, pressure are controlled to avoid overpressure. • Pressure measured by water manometer & temp probe is placed inside reactor or infra-red cell placed in the cavity.


Multimode System • Main drawback: • If temp inside the vessel rises rapidly, partitioning of the more volatile solutes into the headspace may occur- leading to losses of these compounds. • Once extraction is finished- vessels must be cooled to room temp before opening to avoid losses of volatile solutes- increases overall extraction time. • Due to this safety aspect, the size of closed vessels is limited to hundreds of m. L and carousels are used to increase raw material amount extracted.





HOMEWORK: DISCUSS ON 5 th MARCH 2019 • Evaluation of a simple and promising method for extraction of antioxidants from sea buckthorn (Hippophaë rhamnoides L. ) berries: Pressurised solvent-free microwave assisted extraction • Thomas Michel, Emilie Destandau, and Claire Elfakir • Food Chemistry, 126 (2011)

ASSIGNMENT 2 & PRESENTATION • Form a group of 4 to 5 members • Each group is assigned to: • Identify ONE (1) type of bioactive compound that can be extracted from specific raw materials below. • Based on the extraction technology available (conventional methods, microwave assisted extraction and ultrasound assisted extraction), DESIGN ONE (1) appropriate method to extract and quantify this compound. Ø Dragon fruit peel Ø Habbatus sauda seeds Ø Seaweeds Ø Serai wangi Ø Lemuju (Coleus aromaticus) Ø Misai kucing

In the report & presentation must include: ü Type of bioactive compound to be extracted & its demand in the industry. ü Suggestion on extraction technology & its procedure ü Suggestion on the parameters need to be control in order to get a high quality of bioactive compound. ü Suggestion on the appropriate quantification technique for the bioactive compound identified. ü Justification for your suggestions. ü Conclusion The source of your report (including illustration) must be cited. Originality of idea is taken seriously during the assessment. • The report submitted must be not more than 5 pages. • For presentation: • All group members have to participate and individual mark will be given during the presentation session. • Duration for each presentation is 15 minutes and Q&A session will be conducted at the end of presentation. Presentation date: 21 st March 2019 Report submission: 19 th March 2019
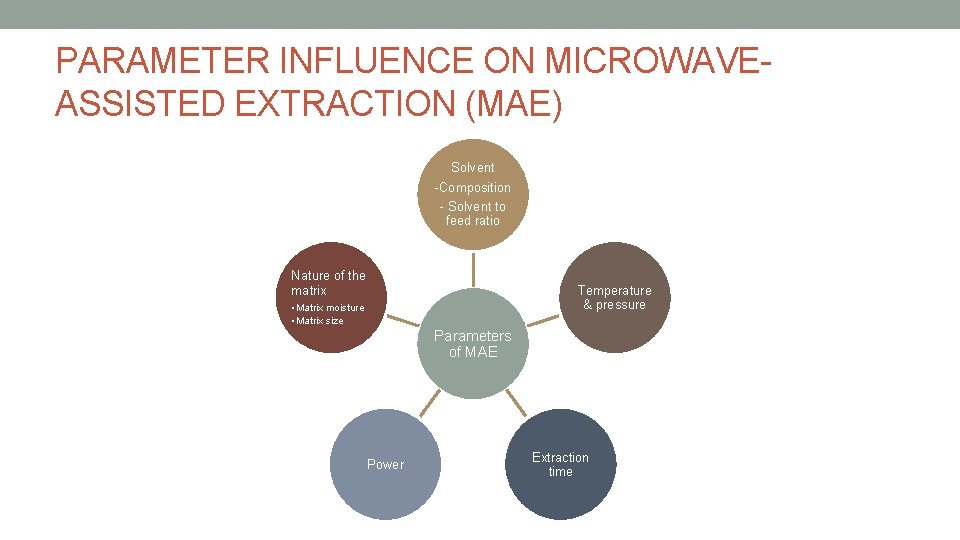
PARAMETER INFLUENCE ON MICROWAVEASSISTED EXTRACTION (MAE) Solvent -Composition - Solvent to feed ratio Nature of the matrix Temperature & pressure • Matrix moisture • Matrix size Parameters of MAE Power Extraction time

• To have an optimize extraction yield & extraction time: • Solvent composition • Solvent to feed ratio

MAE PARAMETER- Solvent composition • According to current developments, three alternative methods for extraction with solvent under microwaves exist. They are: 1. The sample could be immersed in a single solvent or mixtures of solvents that strongly absorb microwave energy 2. The sample could be extracted in a combined solvent containing solvents mixture with both high & low dielectric losses mixed in various. 3. A sample that has high dielectric loss & is able to absorb microwave could be immersed in solvents with low dielectric losses & transparent to microwave energy.

MAE PARAMETER- Solvent composition 1. The sample could be immersed in a single solvent or mixtures of solvents that strongly absorb microwave energy. - These polar solvents could be heated up to their boiling point in an open vessel or above their boiling point in a closed vessel and compounds could be extracted with hot solvent. - Eg: flavonoids from Hippophae rhamnoides were extracted in a focused microwave and ethanol was chosen because alcohols are widely used for antioxidant extraction.

MAE PARAMETER- Solvent composition 2. The sample could be extracted in a combined solvent containing solvents mixture with both high & low dielectric losses mixed in various. - Heating of the solvent is then related to the polar solvent proportion, thus compounds could be extracted in controlled temp conditions. - Eg: MAE of carotenoids from paprika. - 30 different water: organic solvents mixtures in several proportions were evaluated. - Organic solvents tested: acetone, dioxane, ethanol, methanol & tetrahydrofuran in vol ratio 15, 30, 45, 60, 75 and 90%.

MAE PARAMETER- Solvent composition - Both extraction efficiency & selectivity were significantly dependent on the dielectric constant of the extracting solvent mixture. - HPLC profiles showed different absolute values of peak areas demonstrating variation of efficiency and different peak ratio demonstrating the different selectivity according to solvent mixture. - Optimized condition for MAE of pigment from paprika: 120 s extraction time, 50 W energy & temp of 60 o. C and consequently limits molecule rearrangements that could lead to a decrease of total carotene content.

MAE PARAMETER- Solvent composition 3. A sample that has high dielectric loss & is able to absorb microwave could be immersed in solvents with low dielectric losses & transparent to microwave energy. - The hot sample releases compounds in cold solvent avoiding any degradation of thermolabile components. - If the sample not able to absorb microwave energy, water could be added to the sample matrix increasing its dielectric losses. - The increased rate of extraction and selectivity were observed when a transparent solvent, petroleum ether was used to extract piperine from pepper compared to dichloromethane and ethanol in MAE.

MAE PARAMETER- Solvent composition • Recently, some alternatives solvents have been used for MAE of natural compounds. • Ionic liquids- composed of bulky organic cations and inorganic or organic anions, liquid around room temp. • They have attracted much research interest because: • Negligible vapor pressure • Good thermal stability • Wide liquid range • Tunable viscosity • Miscibility with water & organic solvents • Good solubility & extractibility for various organic compounds

MAE PARAMETER- Solvent composition • Considered as ‘green’ alternatives to conventional volatile organic solvents. • IL can efficiently absorb and transfer microwave energy • IL MAE have been used in the extraction of essential oil Rosmarinus officinalis, polyphenolic compounds from Psidium guajava leaves and Smilax china and lichens from Pertusaria pseudocorallina. • The proposed method gave better results compared to conventional methods as well as faster, simple operation that is free of organic solvents.

MAE PARAMETER- Solvent composition • Another specific development of MAE- solvent-free microwave extraction (SFME). • SFME- microwave heating + hydro-distillation performed at atmospheric pressure. • In SFME- fresh or moistened samples with high content of water (at least 70%) is extracted without addition of solvent. • Water contained in the matrix is used as the extraction solvent.

MAE PARAMETER- Solvent composition • A correct choice of solvent is fundamental for obtaining an optimal extraction process. Consideration should be given to the microwave absorbing properties of the solvent, the interaction of the solvent with the matrix and the target compound’s solubility in the solvent. • The solvent should have a high selectivity towards compounds of interest and also able to absorb microwave energy.

MAE PARAMETER- Solvent to feed ratio • A higher solvent to feed ratio in extraction techniques can increase the recovery. • The solvent volume must be sufficient to ensure that the entire sample is immersed, so that the material can swell during extraction. • Since the mechanism underlying the impact of solvent to feed ratio on MAE remains unclear, this ratio has to be adapted and optimized for each raw material.

MAE PARAMETER- Temperature & Pressure • Temperature • Important parameter since it contributes to an increase in yield. • With temp increase- the solvent has higher capacity to solubilize the target compounds, while surface tension & solvent viscosity decrease, improves sample wetting & matrix penetration. • Increased temperature leads to efficient desorption of compounds from the active site in the matrix leading to high extraction recoveries. • In MAE, temp depends on the solvent’s ability to absorb microwaves and on the microwave energy applied (power). • However, temperature should not be too high- it may cause degradation in thermolabile substances.

MAE PARAMETER- Temperature & Pressure • In closed vessel, the temp could exceed the boiling temp of solvent- pressure becomes an important variable. • Pressure is directly dependent on temperature & allows heating above the boiling temp.

MAE PARAMETER- Extraction time • Main advantages of MAE- very short time taken compared to conventional methods (several minutes or seconds). • Duration of microwave radiation of 5 min to 30 min was studied for extraction of flavonoids from Radix astragali. At the beginning the yield increased with the increase of time and reached its maximum at 25 min, then fell down slightly. • With thermolabile compounds, a long extraction time may result in degradation.
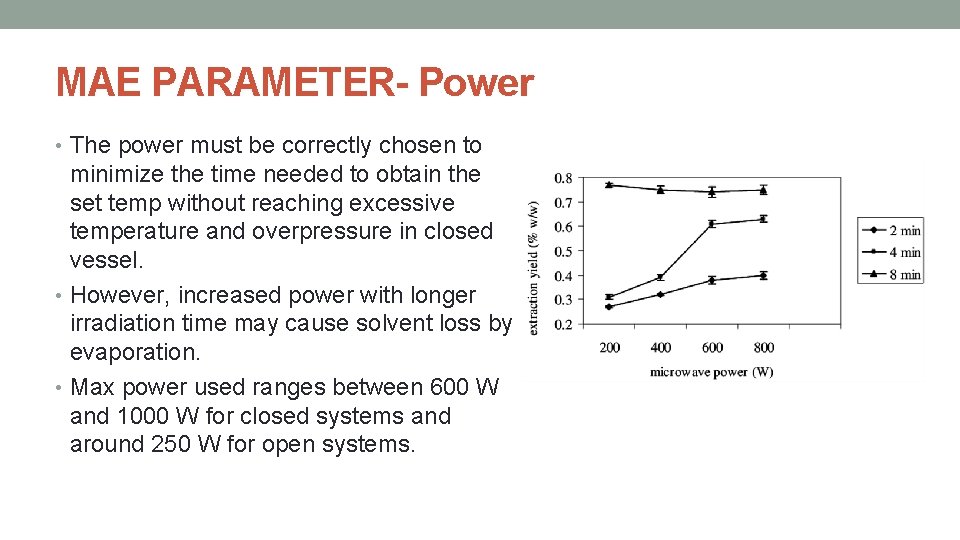
MAE PARAMETER- Power • The power must be correctly chosen to minimize the time needed to obtain the set temp without reaching excessive temperature and overpressure in closed vessel. • However, increased power with longer irradiation time may cause solvent loss by evaporation. • Max power used ranges between 600 W and 1000 W for closed systems and around 250 W for open systems.

MAE PARAMETER- Nature of matrix • Matrix moisture • Water added or naturally present in the sample is important- as water molecules have a high dipole moment, thus strongly absorb microwave energy. • Thus, water always has effect on the microwave absorbing ability & hence facilitates the heating process, increasing the polarity of the extracting solvent and allowing sample to be heated. • It also have a swelling effect on the matrix & influence the solute-matrix interactions- making solutes more available to the extracting solvent. • A dry raw material- re-hydrated before extraction to facilitate the heating process by changing its dielectric properties. • The sample can be soaked with water, varies from 10 min to 24 h.

MAE PARAMETER- Matrix moisture • Eg: essential oil extraction from dry seed of cardamom. • Dry seeds were moistened prior to extraction by soaking in water then draining the excess water. Humidity level of the sample was then studied from 30% to 70%, the optimum was found at 67%. • The effect of this parameter also depends on the extraction solvent. If the plant material contains enough water to absorb microwave energy, the surrounding solvent can have low dielectric constant thus remain cold during extraction- useful for thermolabile components.

MAE PARAMETER- Matrix size • The particle sizes of the extracted materials are usually in the range of 100 um to 2 mm. • Fine powders- enhance the extraction because the limiting step of the extraction is often the diffusion of chemicals out of the plant matrix. • Thus, smaller size materials have less diffusion depth for molecule diffusion out of the plant matrix to the surrounding solvent. • Moreover, the larger surface area of particles provides contact between the plant matrix & the solvent and smaller particles have less penetration depth that leads to uniform microwave exposure.

TRENDS IN MAE & APPLICATIONS • EXTRACTION OF SENSITIVE COMPOUNDS • MAE can be carried out under an inert atmosphere or under vacuum, so limiting the presence of oxygen in the reactor. • There are two types: • Nitrogen-protected Microwave-assisted Extraction (NPMAE) • Vacuum Microwave-assisted Extraction (VMAE)

Nitrogen protected Microwave-assisted extraction (NPMAE) • Oxidation of the active compounds during extraction process can be prevented using pressurized inert gas, such as nitrogen in a closed system. • The plant sample is introduced into the vessel with a certain vol of solvent. • The air in the flask is pumped out by a vacuum pump until certain degree of vacuum is obtained. • Then, the vessel is filled with N gas. • This technique has been employed in the extraction of ascorbic acid from guava, yellow pepper & cayenne pepper.

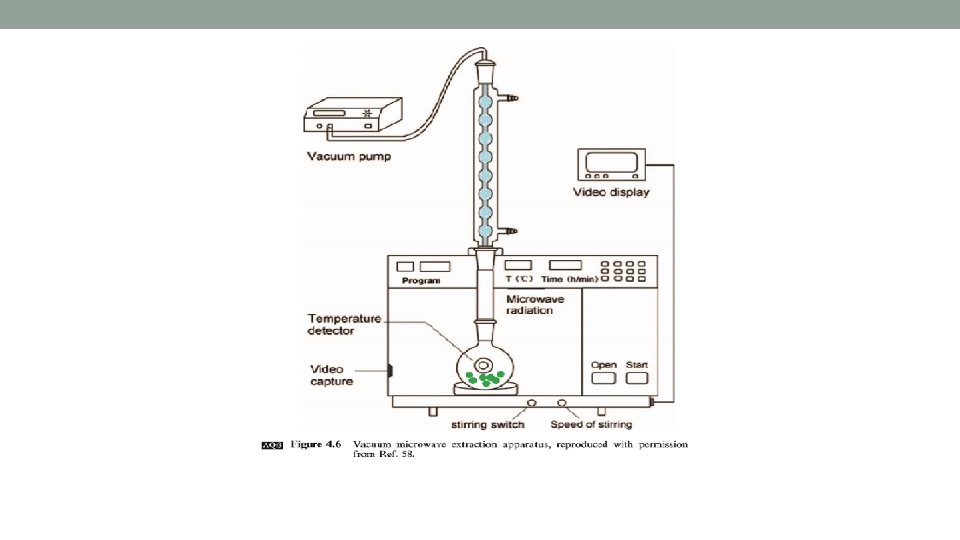
Vacuum Microwave-assisted Extraction (VMAE) • The boiling point (BP) of the extraction solvent under vacuum is lower than at ambient pressure. • Thus, the solvent can be kept boiling & refluxing at lower temp- good for extracting thermosensitive compounds while preventing their degradation. • Furthermore, the air in the extraction system is pumped out, so oxidation of oxygen-sensitive compounds is avoided or reduced. • It enhances mass transfer mechanism by promoting diffusion of active compounds to the solvent via the suction pressure.

Extraction methods improved by microwave heating • Rapid microwave heating of the solvent-sample mixture allows reducing extraction time & energy consumption- microwave energy speed up the heating & improved the efficiency of conventional extraction methods. • Focused Microwave-assisted Soxhlet extraction (FMASE) • Based on the same principles of conventional Soxhlet extraction but using microwave as auxiliary energy to accelerate the process. • The sample cartridge compartment is located in the irradiation zone of a microwave oven.

Focused Microwave-assisted Soxhlet extraction (FMASE) • FMASE preserves the advantages of conventional Soxhlet extraction while overcoming restrictions such as the long extraction time.

Microwave hydrodistillation (MWHD) • The HD apparatus is placed inside a microwave oven with a side orifice through which an external cooler joins the vessel containing the plant material and water inside the oven. • The oven is operated at full power, which causes water to boil and reflux.
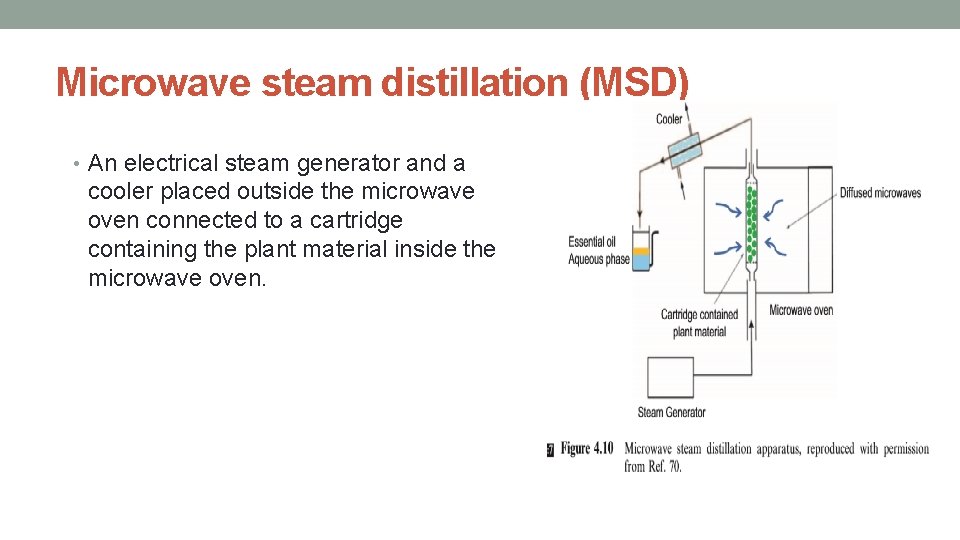
Microwave steam distillation (MSD) • An electrical steam generator and a cooler placed outside the microwave oven connected to a cartridge containing the plant material inside the microwave oven.

Microwave-assisted Extraction (MAE) • Advantages of MAE: • lower extraction time • lower solvent usage • higher selectivity • volumetric heating and controllable heating process • cost-effective when compared to accelerated solvent extraction • Disadvantages of MAE: • reduction of extraction yield: as it may modify the chemical structures of the target compounds • extraction efficiency of microwave technique may be very poor when either the target compounds or solvent are nonpolar, or when the viscosity of solvent is extremely high • Need to be extra cautious for the extraction of thermally labile compounds • apparatuses and equipment of microwave assisted extraction are more expensive and their operation is more difficult in many cases when compared with ultrasonic-assisted extraction.

- Slides: 61