Microwave and ab initio Studies of the XeCH

Microwave and ab initio Studies of the Xe-CH 4 van der Waals Complex Qing Wen and Wolfgang Jäger Department of Chemistry, University of Alberta, Edmonton, Alberta, Canada 1

Motivation n Xenon has a large electron cloud which is easily polarized by interaction with its surroundings. n Applications of the 129 Xe (I = ½) nucleus in NMR. ¨ Probe the structures of proteins and microporous materials. ¨ Spin-polarized Xenon increases NMR signals by 104 (imaging applications). n Xe-CH 4 is a test case to investigate Xenon-molecule intermolecular interactions. 2

ab initio Calculations n n n Program: MOLPRO 2002. 6 package CCSD(T) level of theory Basis sets: ¨ Xe: aug-cc-p. VQZ-PP[1] ¨ C, H: aug-cc-p. VTZ n n n [1] Midbond functions (3 s, 3 p, 2 d, 2 f, 1 g) Supermolecule approach Counterpoise correction to reduce the Basis Set Superposition Error Effective Core Potential. K. Peterson et. al. , J. Chem. Phys. 119, 11113 (2003). 3
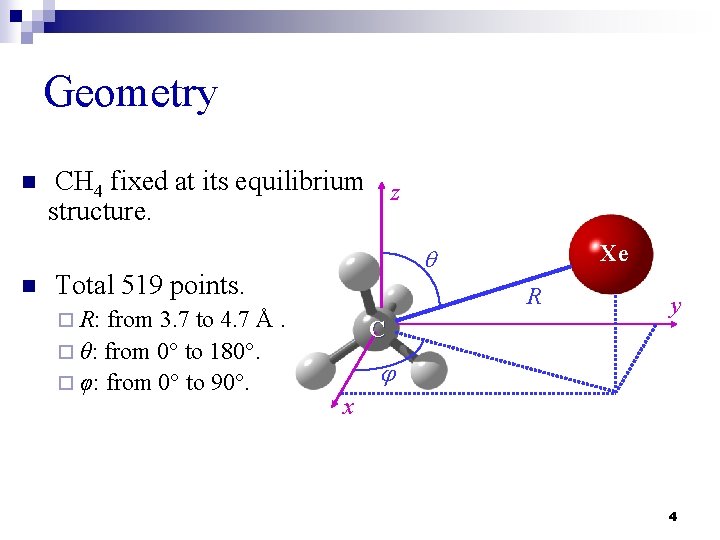
Geometry n n CH 4 fixed at its equilibrium z structure. Total 519 points. from 3. 7 to 4. 7 Å. ¨ θ: from 0 to 180. ¨ φ: from 0 to 90. Xe q R ¨ R: C y j x 4

Special Configurations C 3 face C 3 vertex C 2 edge 5
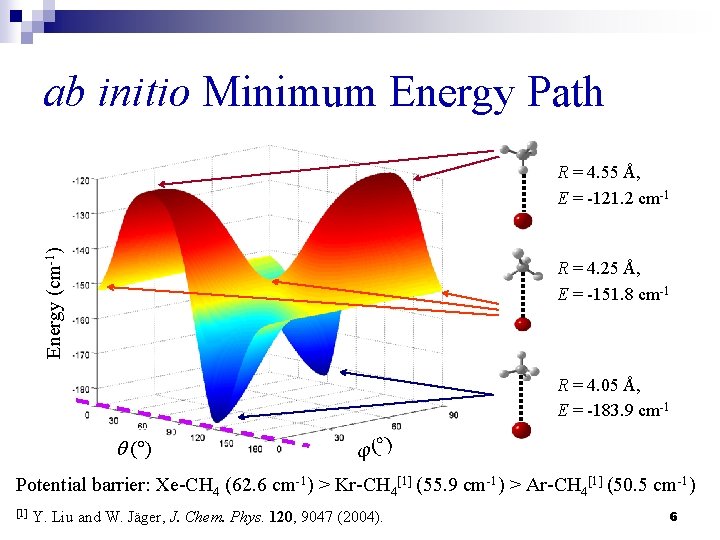
ab initio Minimum Energy Path Energy (cm-1) R = 4. 55 Å, E = -121. 2 cm-1 R = 4. 25 Å, E = -151. 8 cm-1 R = 4. 05 Å, E = -183. 9 cm-1 q ( ) j ( ) Potential barrier: Xe-CH 4 (62. 6 cm-1) > Kr-CH 4[1] (55. 9 cm-1) > Ar-CH 4[1] (50. 5 cm-1) [1] Y. Liu and W. Jäger, J. Chem. Phys. 120, 9047 (2004). 6
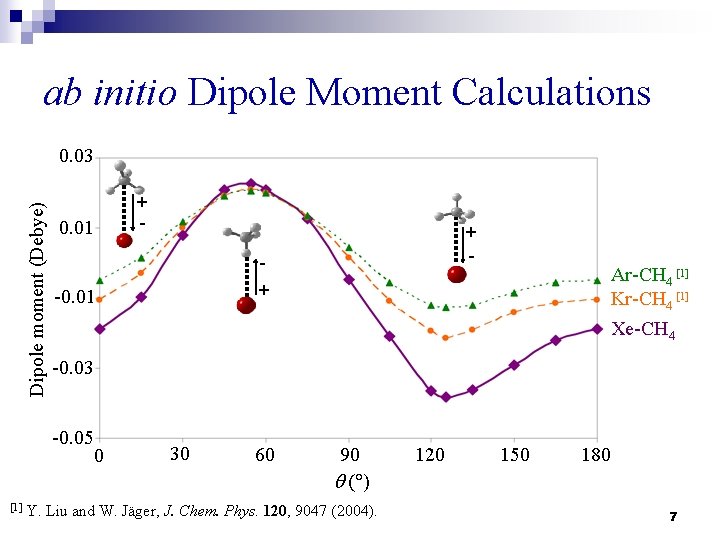
ab initio Dipole Moment Calculations Dipole moment (Debye) 0. 03 0. 01 + -0. 01 Ar-CH 4 [1] Kr-CH 4 [1] Xe-CH 4 -0. 03 -0. 05 0 [1] + - 30 60 90 q ( ) Y. Liu and W. Jäger, J. Chem. Phys. 120, 9047 (2004). 120 150 180 7

Rotational Levels n j Rotational level labelling scheme: ¨ j: for methane internal rotation. ¨ K: the projection of j onto molecular axis. ¨ J: for overall rotation of the complex. n At low rotational temperature (< 1 K), three internal rotor states are well populated. ¨j ¨j ¨j = 0, K = 0 state (A) = 1, K = 0 state (F) = 2, K = 1 state (E) 8

Experimental Setup n Balle-Flygare-type Fourier transform microwave spectrometer ¨ 3. 5 n to 18 GHz range Sample conditions ¨ 0. 7% Xe, 1. 0% CH 4 ¨ Ne backing gas at 6 atm 9

Microwave Spectra n The rotational spectra for 25 isotopomers were recorded (5 Xe isotopomers & 5 CH 4 isotopomers). n Three or four ΔJ = 1 transitions were measured within each internal rotor state. n Nuclear quadrupole hyperfine structures due to 131 Xe (I = 3/2) were observed and assigned. 10
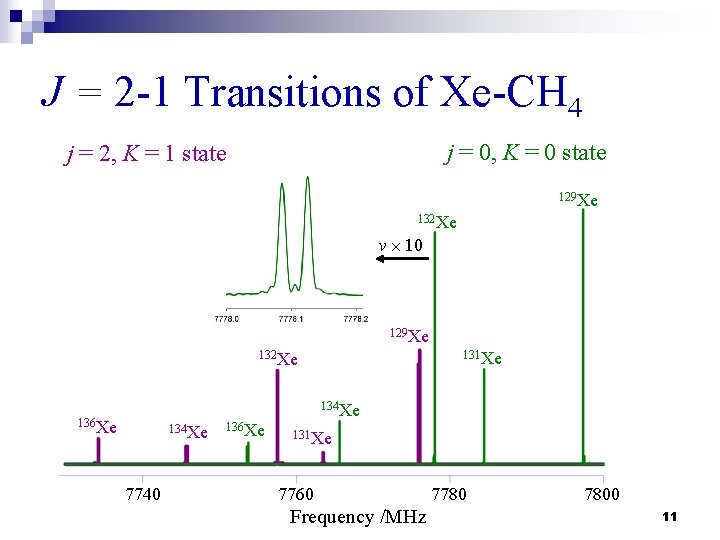
J = 2 -1 Transitions of Xe-CH 4 j = 0, K = 0 state j = 2, K = 1 state 129 Xe 132 Xe ν 10 129 Xe 131 Xe 132 Xe 134 Xe 136 Xe 134 Xe 7740 136 Xe 131 Xe 7760 Frequency /MHz 7780 7800 11
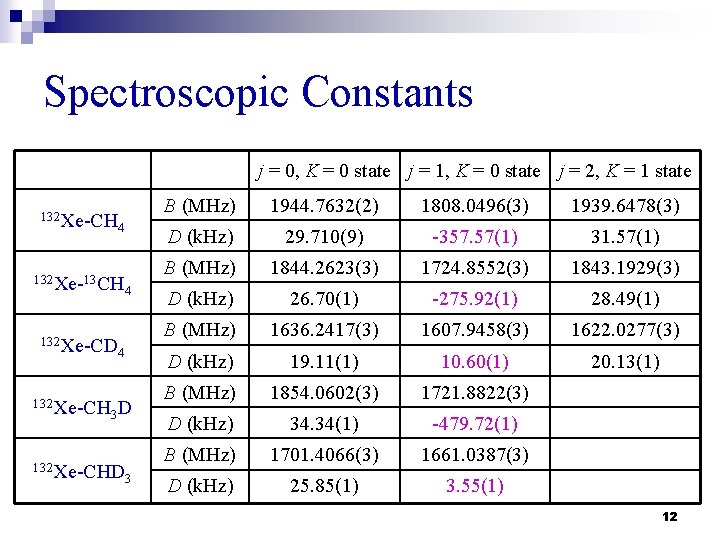
Spectroscopic Constants j = 0, K = 0 state j = 1, K = 0 state j = 2, K = 1 state 132 Xe-CH 4 132 Xe-13 CH 132 Xe-CD 132 Xe-CH 4 4 3 D 132 Xe-CHD 3 B (MHz) 1944. 7632(2) 1808. 0496(3) 1939. 6478(3) D (k. Hz) 29. 710(9) -357. 57(1) 31. 57(1) B (MHz) 1844. 2623(3) 1724. 8552(3) 1843. 1929(3) D (k. Hz) 26. 70(1) -275. 92(1) 28. 49(1) B (MHz) 1636. 2417(3) 1607. 9458(3) 1622. 0277(3) D (k. Hz) 19. 11(1) 10. 60(1) 20. 13(1) B (MHz) 1854. 0602(3) 1721. 8822(3) D (k. Hz) 34. 34(1) -479. 72(1) B (MHz) 1701. 4066(3) 1661. 0387(3) D (k. Hz) 25. 85(1) 3. 55(1) 12

Coriolis Interaction n n The j = 1, K = 0 state is perturbed by a Coriolis interaction. j = 1, K = 1 1+ 10 j = 1, K = 0 Lower angular anisotropy of interaction potential → larger the Coriolis effect → more negative the D constant. Ar-CH 4 [1] D (k. Hz) 84 Kr-CH [1] 4 132 Xe-CH -1380 -357. 57 4 ←——– The degree of Coriolis effect increases ab initio barrier (cm-1) 50. 5 55. 9 62. 6 Angular anisotropy of potential increases ——–→ [1] Y. Liu and W. Jäger, J. Chem. Phys. 120, 9047 (2004). 13
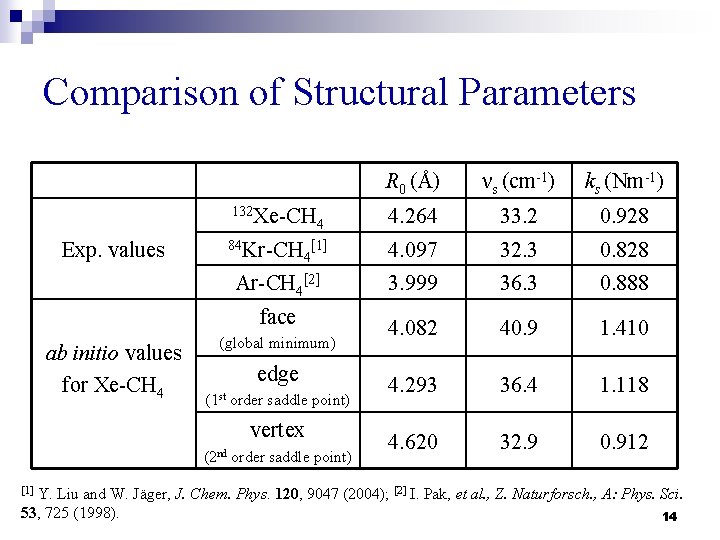
Comparison of Structural Parameters Exp. values R 0 (Å) νs (cm-1) ks (Nm-1) 132 Xe-CH 4. 264 33. 2 0. 928 84 Kr-CH 4. 097 32. 3 0. 828 3. 999 36. 3 0. 888 4. 082 40. 9 1. 410 4. 293 36. 4 1. 118 4. 620 32. 9 0. 912 4 [1] 4 Ar-CH 4[2] face ab initio values for Xe-CH 4 (global minimum) edge (1 st order saddle point) vertex (2 nd order saddle point) Y. Liu and W. Jäger, J. Chem. Phys. 120, 9047 (2004); [2] I. Pak, et al. , Z. Naturforsch. , A: Phys. Sci. 53, 725 (1998). 14 [1]
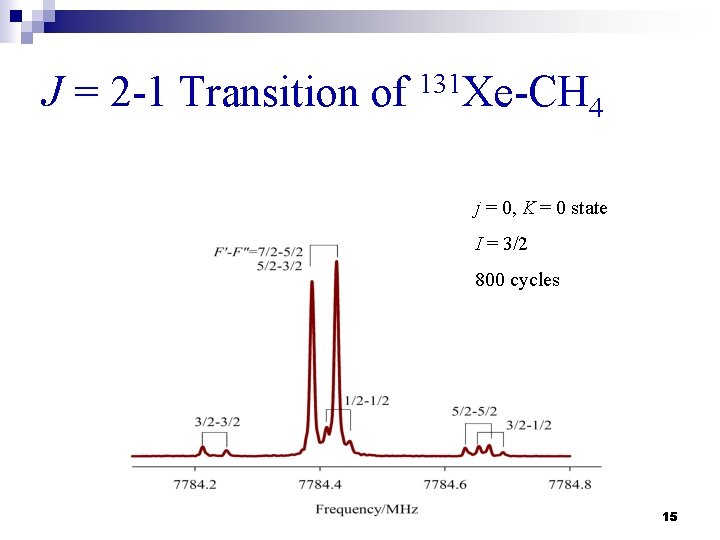
J = 2 -1 Transition of 131 Xe-CH 4 j = 0, K = 0 state I = 3/2 800 cycles 15
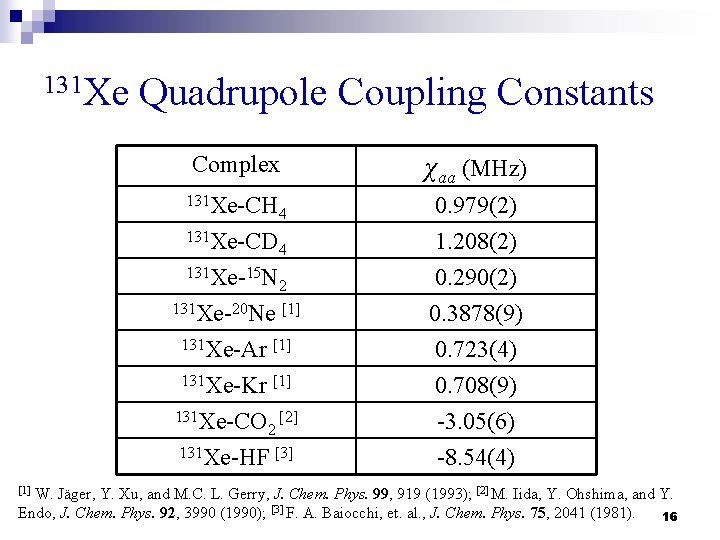
131 Xe Quadrupole Coupling Constants Complex χaa (MHz) 131 Xe-CH 4 131 Xe-CD 4 0. 979(2) 1. 208(2) 0. 290(2) 131 Xe-15 N 2 131 Xe-20 Ne [1] 131 Xe-Ar [1] 131 Xe-Kr [1] 131 Xe-CO [2] 2 131 Xe-HF [3] 0. 3878(9) 0. 723(4) 0. 708(9) -3. 05(6) -8. 54(4) W. Jäger, Y. Xu, and M. C. L. Gerry, J. Chem. Phys. 99, 919 (1993); [2] M. Iida, Y. Ohshima, and Y. Endo, J. Chem. Phys. 92, 3990 (1990); [3] F. A. Baiocchi, et. al. , J. Chem. Phys. 75, 2041 (1981). 16 [1]

Conclusions n Methane undergoes large amplitude internal rotation motion within the Xe-CH 4 complex. n The j = 1, K = 0 state is perturbed by a Coriolis interaction. n The charge distribution around Xenon nucleus is very sensitive to its surroundings. 17

Acknowledgement n Dr. Jäger and Xu group for helpful discussions and suggestions. n L. Tang for designing and implementing the MWFT program. 18
- Slides: 18