Microstructure Evolution Statistical Thermodynami ByeongJoo Lee cs POSTECH

Microstructure Evolution Statistical Thermodynami Byeong-Joo Lee cs POSTECH - MSE calphad@postech. ac. kr Byeong-Joo Lee www. postech. ac. kr/~calphad

Warming Up – Mathematical Skills 1. Stirling’s approximation 2. Evaluation of the Integral 3. Lagrangian Undetermined Multiplier Method Byeong-Joo Lee www. postech. ac. kr/~calphad

Basic Concept of Statistical Mechanics – Macro vs. Micro View Point Macroscopic vs. Microscopic State Macrostate vs. Microstate Byeong-Joo Lee www. postech. ac. kr/~calphad

Particle in a Box – Microstates of a Particle n = 1, 2, 3, … for 66 : 8, 1, 1 7, 4, 1, 5, 5, 4 Byeong-Joo Lee www. postech. ac. kr/~calphad
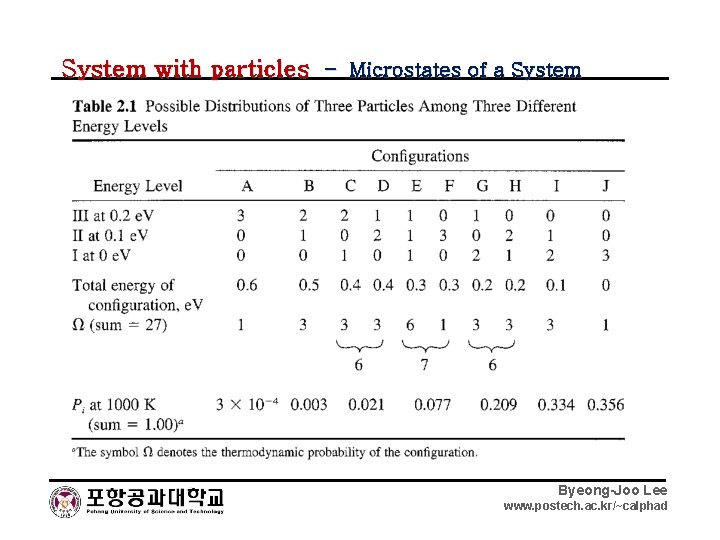
System with particles – Microstates of a System Byeong-Joo Lee www. postech. ac. kr/~calphad
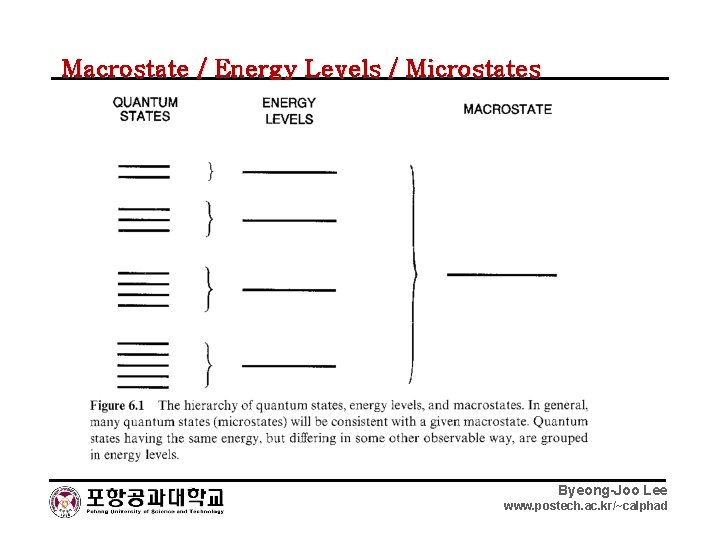
Macrostate / Energy Levels / Microstates Byeong-Joo Lee www. postech. ac. kr/~calphad

Scope and Fundamental Assumptions of Statistical Mechanics ▷ Microstate: each of the possible states for a macrostate. (n 1, n 2, …, nk)로 정의되는 하나의 macrostate를 만들기 위해, 있을 수 있는 수많은 경우의 수 하나하나를 microstate라 한다. ▷ Ensemble: mental collection of macrostates 어떠한 시스템에 가능한 (quantum mechanically accessible 한) macrostate (하나하나가 (n 1, n 2, …, nk)로 정의되는)의 mental collection을 ensemble이라 한다. ▷ Each microstate is equally probability. 같은 energy level에서 모든 microstate의 실현 확률은 동등하 다. ▷ Ensemble average = time average Byeong-Joo Lee www. postech. ac. kr/~calphad

Number of ways of distribution : in k cells with gi and Ei ▷ Distinguishable without Pauli exclusion principle ▷ Indistinguishable without Pauli exclusion principle for gi with ni ▷ Indistinguishable with Pauli exclusion principle for gi with ni Byeong-Joo Lee www. postech. ac. kr/~calphad

Evaluation of the Most Probable Macrostate – Boltzmann Byeong-Joo Lee www. postech. ac. kr/~calphad

Evaluation of the Most Probable Macrostate – B-E & F-D Bose-Einstein Distribution → Fermi-Dirac Distribution Byeong-Joo Lee www. postech. ac. kr/~calphad

Definition of Entropy and Significance of β ▷ Consider an Isolated System composed of two part in Thermal contact. @ equilibrium in Classical Thermodynamics: maximum entropy (S) in Statistical mechanics: maximum probability (Ω) ▷ There exists a monotonic relation between S and Ω → Byeong-Joo Lee www. postech. ac. kr/~calphad

Calculation of Macroscopic Properties from Partition Function Byeong-Joo Lee www. postech. ac. kr/~calphad
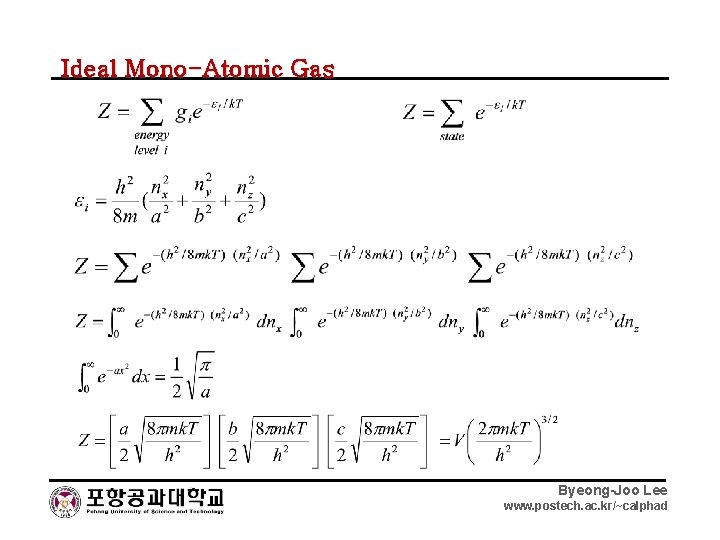
Ideal Mono-Atomic Gas Byeong-Joo Lee www. postech. ac. kr/~calphad
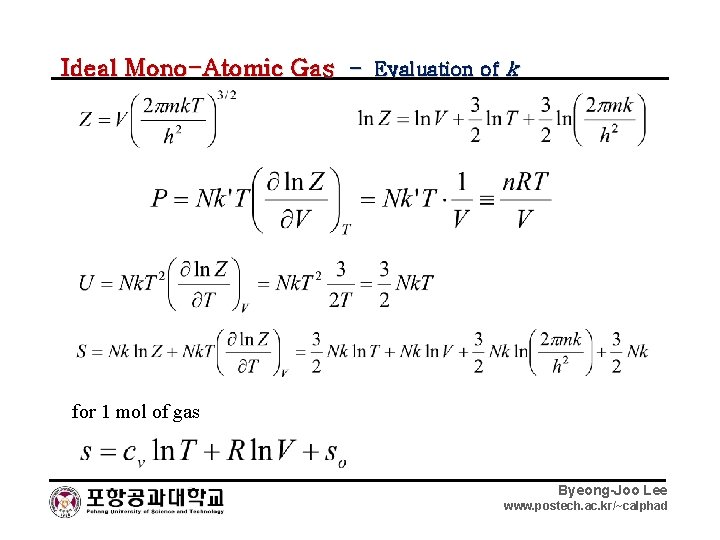
Ideal Mono-Atomic Gas – Evaluation of k for 1 mol of gas Byeong-Joo Lee www. postech. ac. kr/~calphad

Entropy – S = k ln W Byeong-Joo Lee www. postech. ac. kr/~calphad

Equipartition Theorem The average energy of a particle per independent component of motion is translational kinetic energy : rotational kinetic energy : vibrational energy : kinetic energy for each independent component of motion has a form of Byeong-Joo Lee www. postech. ac. kr/~calphad

Equipartition Theorem The average energy of a particle per independent component of motion is ※ for a monoatomic ideal gas : for diatomic gases : for polyatomic molecules which are soft and vibrate easily with many frequencies, say, q: ※ for liquids and solids, the equipartition principle does not work Byeong-Joo Lee www. postech. ac. kr/~calphad

Einstein & Debye Model for Heat Capacity – Background & Concept 3 N independent (weakly interacting) but distinguishable simple harmonic oscillators. for N simple harmonic vibrators average energy per vibrator Byeong-Joo Lee www. postech. ac. kr/~calphad

Einstein & Debye Model for Heat Capacity – number density Let d. Nv be the number of oscillators whose frequency lies between v and v + dv where g(v), the number of vibrators per unit frequency band, satisfy the condition The energy of N particles of the crystal Byeong-Joo Lee www. postech. ac. kr/~calphad

Einstein & Debye Model for Heat Capacity – Einstein All the 3 N equivalent harmonic oscillators have the same frequency v. E Defining Einstein characteristic temperature Byeong-Joo Lee www. postech. ac. kr/~calphad

Einstein & Debye Model for Heat Capacity – Debye A crystal is a continuous medium supporting standing longitudinal and transverse waves set Byeong-Joo Lee www. postech. ac. kr/~calphad
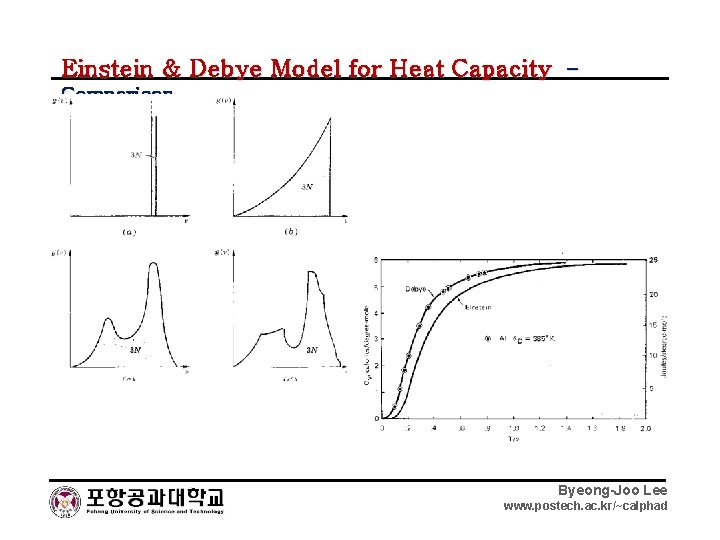
Einstein & Debye Model for Heat Capacity – Comparison Byeong-Joo Lee www. postech. ac. kr/~calphad
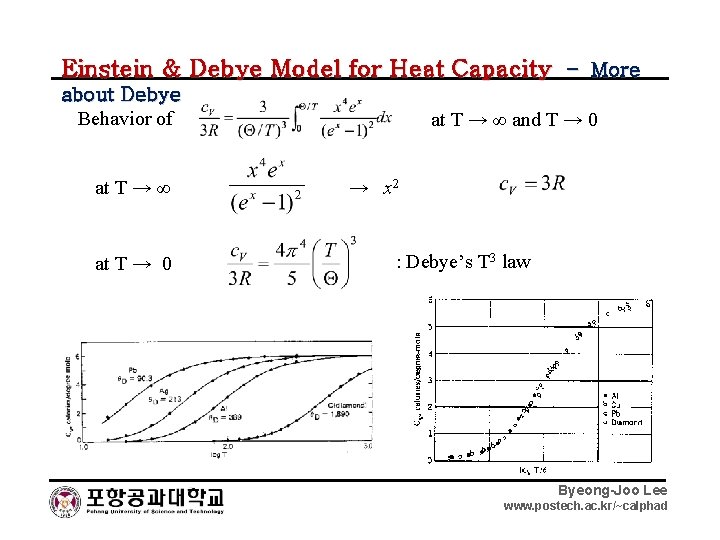
Einstein & Debye Model for Heat Capacity – More about Debye Behavior of at T → ∞ at T → 0 at T → ∞ and T → 0 → x 2 : Debye’s T 3 law Byeong-Joo Lee www. postech. ac. kr/~calphad
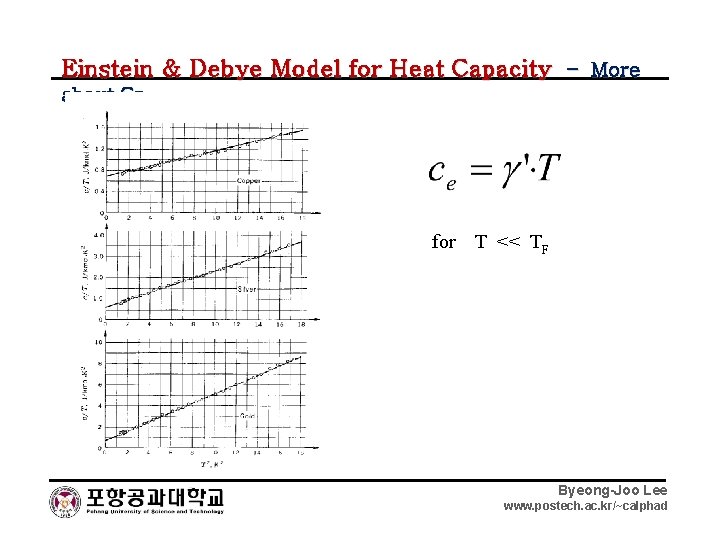
Einstein & Debye Model for Heat Capacity – More about Cp for T << TF Byeong-Joo Lee www. postech. ac. kr/~calphad

Statistical Interpretation of Entropy – Numerical Example A rigid container is divided into two compartments of equal volume by a partition. One compartment contains 1 mole of ideal gas A at 1 atm, and the other compartment contains 1 mole of ideal gas B at 1 atm. (a) Calculate the entropy increase in the container if the partition between the two compartments is removed. (b) If the first compartment had contained 2 moles of ideal gas A, what would have been the entropy increase due to gas mixing when the partition was removed? (c) Calculate the corresponding entropy changes in each of the above two situations if both compartments had contained ideal gas A. Byeong-Joo Lee www. postech. ac. kr/~calphad
- Slides: 25