Microstructural evolution and brazing mechanism of Ti 2

Microstructural evolution and brazing mechanism of Ti 2 Sn. C-Ti 6 Al 4 V joint by using Cu pure foil Wenbo Yu, Shibo Li , Yann Aman, Zhipeng Guo, Shoumei Xiong School of materials science and engineering Tsinghua University Beijing China School of Materials Science and Engineering
![MAX phases Mn+1 AXn [1] M: Early transition metal A: A Group element X: MAX phases Mn+1 AXn [1] M: Early transition metal A: A Group element X:](http://slidetodoc.com/presentation_image/65047dbab8729591bbc45d560482afd8/image-2.jpg)
MAX phases Mn+1 AXn [1] M: Early transition metal A: A Group element X: C and/or N Edge-sharing [M 6 X] octahedra interleaved with A layers Ceramic + Metal properties [1] M. W. Barsoum et al. , , Progress in Solid State Chemistry, 28 (1 -4), 201 -281, 2000 Good oxidation resistance Good damage tolerance School of Materials Science and Engineering

Applications Ti. C 0. 67 Al 2 O 3 Heating elements: Good oxidation resistance Pantograph: Good electrical conductivity Low friction coefficient Motor: Low density Good resistance at high temperature School of Materials Science and Engineering
![Background Ti 2 Sn. C/Al 2 O 3 composite [3] Ti 2 Sn. C[2] Background Ti 2 Sn. C/Al 2 O 3 composite [3] Ti 2 Sn. C[2]](http://slidetodoc.com/presentation_image/65047dbab8729591bbc45d560482afd8/image-4.jpg)
Background Ti 2 Sn. C/Al 2 O 3 composite [3] Ti 2 Sn. C[2] Crack healing 800 ℃ /1 h [2] Li, S. , et al. , . Journal of the European Ceramic Society, 2016. 36(1): p. 25 -32. [3] Bei et al. J. Am. Ceram. Soc. 98 (2015)1604 -1610 School of Materials Science and Engineering

Background Ti-6 Al-4 V Widely biomedical application Such bone replacement 10µm Drawbacks: biotoxicity of vanadium sonication 800℃/1 h [5] Possible: using Ti 2 Sn. C to replace Ti 3 Si. C 2 The temperature can be strongly decreased 1200 ℃ /1 h [4] Gao et al. , . Journal of material research, 2002. 17(1): p. 25 -32. [5] Zhao et al. Transaction nonferrous metal society. 2009 20(2)5 p, 414 -417 School of Materials Science and Engineering
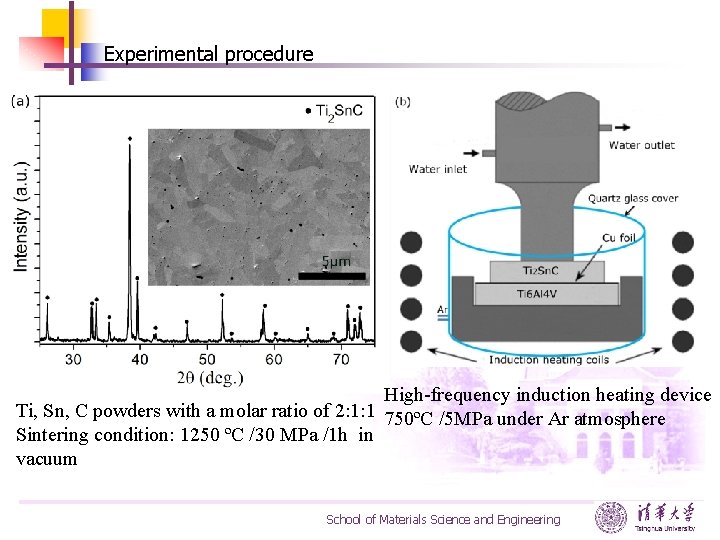
Experimental procedure High-frequency induction heating device Ti, Sn, C powders with a molar ratio of 2: 1: 1 750ºC /5 MPa under Ar atmosphere Sintering condition: 1250 ºC /30 MPa /1 h in vacuum School of Materials Science and Engineering
![750ºC /5 MPa/1 h Zhao et al. [5] layer I: migration of Cu atom 750ºC /5 MPa/1 h Zhao et al. [5] layer I: migration of Cu atom](http://slidetodoc.com/presentation_image/65047dbab8729591bbc45d560482afd8/image-7.jpg)
750ºC /5 MPa/1 h Zhao et al. [5] layer I: migration of Cu atom into β-Ti layer II: Cu 4 Ti 3 Layer III : rich Cu and Ti Layer IV: Cu. Ti 0. 5 Sn 0. 5 and Sn layer V: β-Cu(Sn) (bright area) and α-Cu(Sn) (grey area) School of Materials Science and Engineering
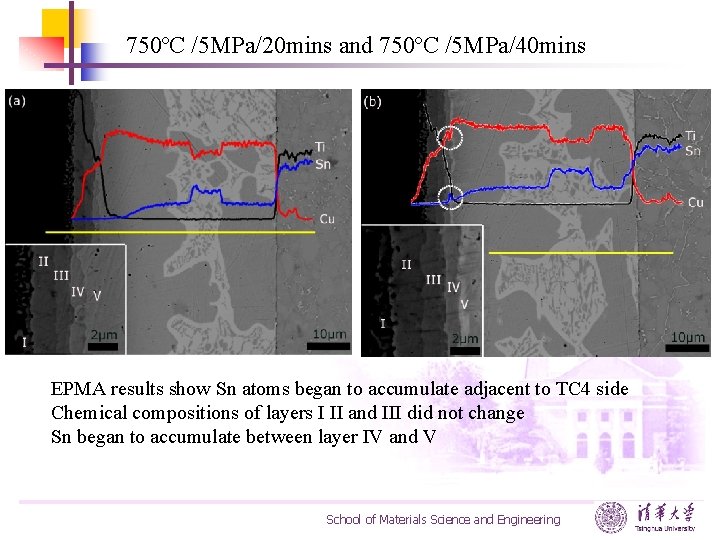
750ºC /5 MPa/20 mins and 750ºC /5 MPa/40 mins EPMA results show Sn atoms began to accumulate adjacent to TC 4 side Chemical compositions of layers I II and III did not change Sn began to accumulate between layer IV and V School of Materials Science and Engineering

The side of Cu and Ti 2 Sn. C FIB procedure 1 In Fig(d), BSE images indicates two contrasts, Grain 1 was surrounded by white dots. School of Materials Science and Engineering

TEM results Ti: Sn=1: 0. 73 Sn atoms diffuse out from Ti 2 Sn. C grains Cu atoms could diffuse into Ti 2 Sn. C grain boundaries and form Cu(Sn) solid solution. School of Materials Science and Engineering

Shear test shear strength : 85. 7± 10 MPa. crack propagated in Ti 2 Sn. C Intergranular fracture mode. School of Materials Science and Engineering

Conclusions The side adjacent to Ti 2 Sn. C, Ti 2 Sn. C → x. Sn +Ti 2 Sn 1 -x. C Sn +Cu → β-Cu (Sn) + α-Cu (Sn) (1) (2) The side adjacent to TC 4, the interdiffusion between Ti and Cu resulted into the following reaction. Cu + βTi → βTi (Cu) (layer I) (3) 3 Ti +5 Cu → Ti 3 Cu 4 layer (II) + Cu (Sn, Ti) (layer III) (4) With the increasing processing time, Sn atoms began to accumulate into Cu(Ti) layer and form Cu. Ti 0. 5 Sn 0. 5 intermetallic. 1. 5 Sn + Cu (Ti) → Sn +Cu. Ti 0. 5 Sn 0. 5 (layer IV) (5) School of Materials Science and Engineering

Thank you School of Materials Science and Engineering
- Slides: 13