Microscopy Based Biosensors and Functional Assays Receptor Inputs
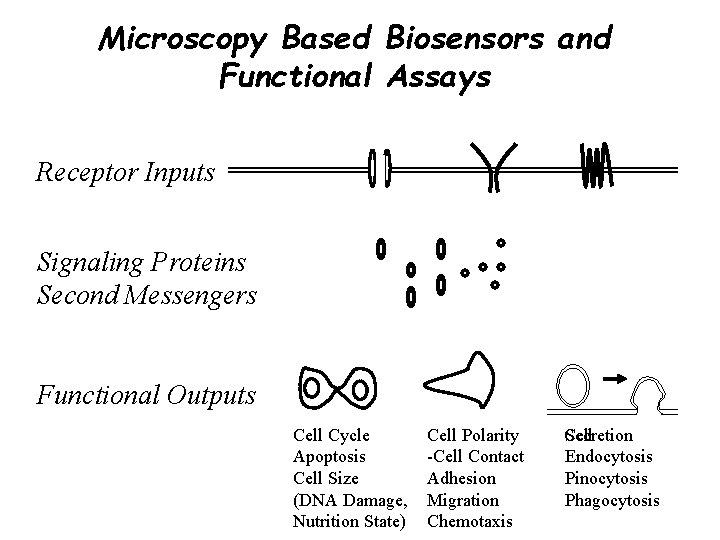
Microscopy Based Biosensors and Functional Assays Receptor Inputs Signaling Proteins Second Messengers Functional Outputs Cell Cycle Apoptosis Cell Size (DNA Damage, Nutrition State) Cell Polarity -Cell Contact Adhesion Migration Chemotaxis Cell Secretion Endocytosis Pinocytosis Phagocytosis
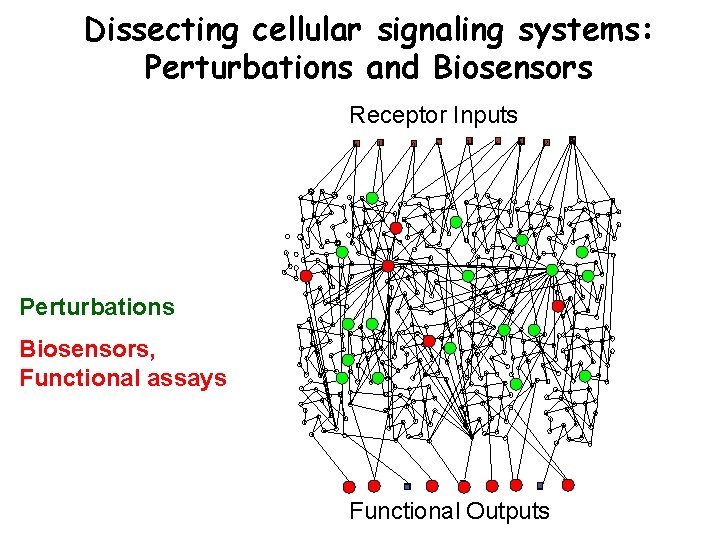
Dissecting cellular signaling systems: Perturbations and Biosensors Receptor Inputs Perturbations Biosensors, Functional assays Functional Outputs

1. Microscopy Strategies to Explore Signaling Systems 1. Epifluorescence Imaging 2. Confocal Imaging 3. Total Internal Reflection Microscopy
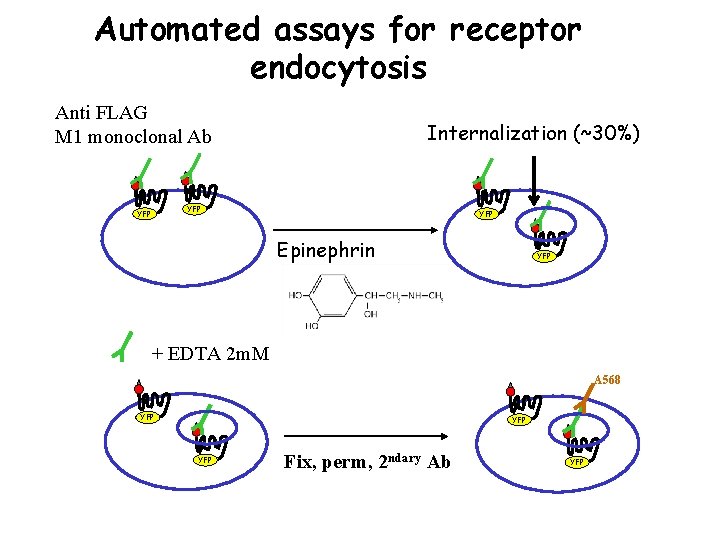
Automated assays for receptor endocytosis Anti FLAG M 1 monoclonal Ab Internalization (~30%) YFP YFP Epinephrin YFP + EDTA 2 m. M A 568 YFP YFP Fix, perm, 2 ndary Ab YFP
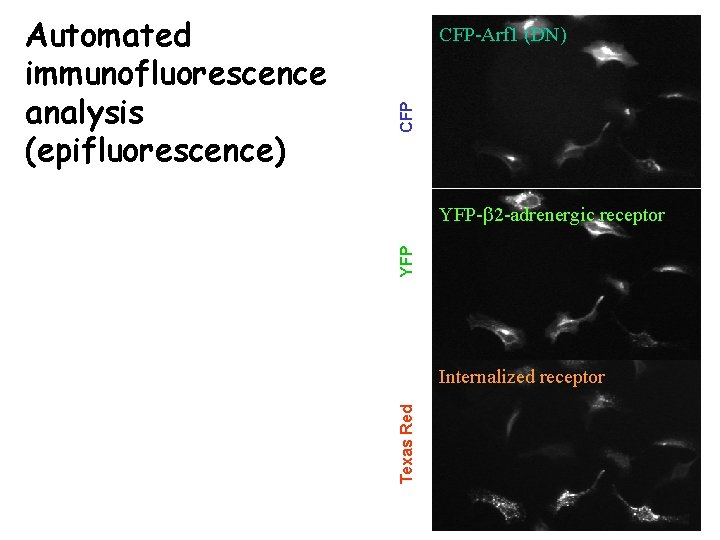
CFP CFP-Arf 1 (DN) YFP-b 2 -adrenergic receptor Internalized receptor Texas Red Automated immunofluorescence analysis (epifluorescence)
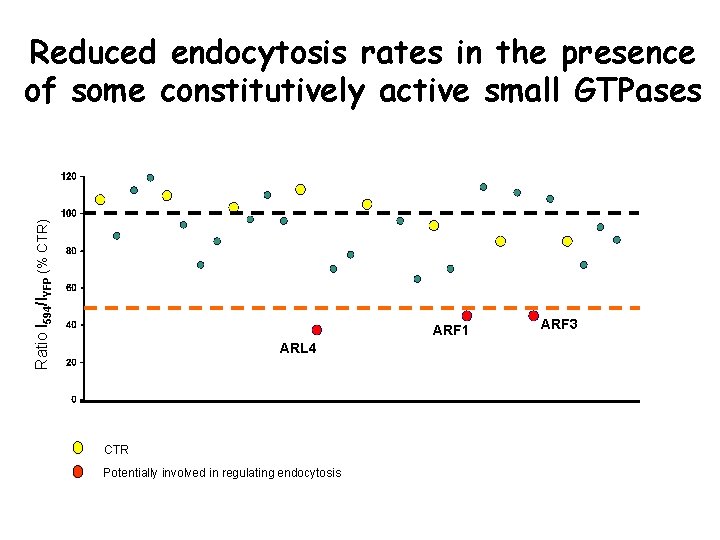
Ratio I 594/IYFP (% CTR) Reduced endocytosis rates in the presence of some constitutively active small GTPases ARF 1 ARL 4 CTR Potentially involved in regulating endocytosis ARF 3
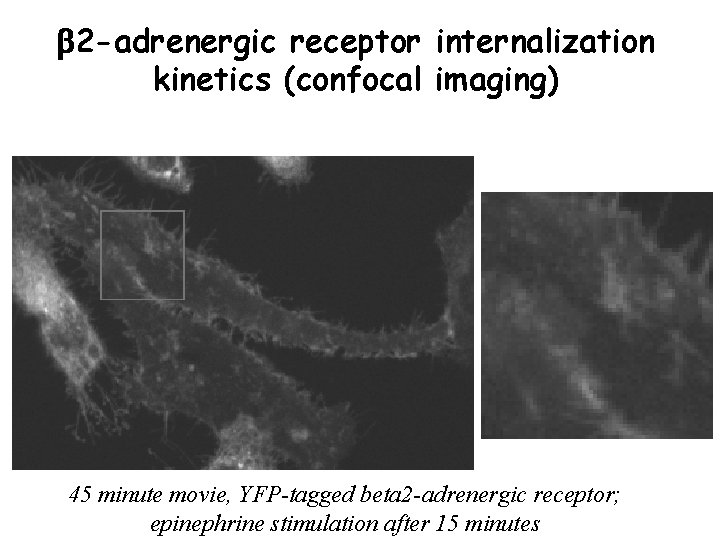
b 2 -adrenergic receptor internalization kinetics (confocal imaging) 45 minute movie, YFP-tagged beta 2 -adrenergic receptor; epinephrine stimulation after 15 minutes
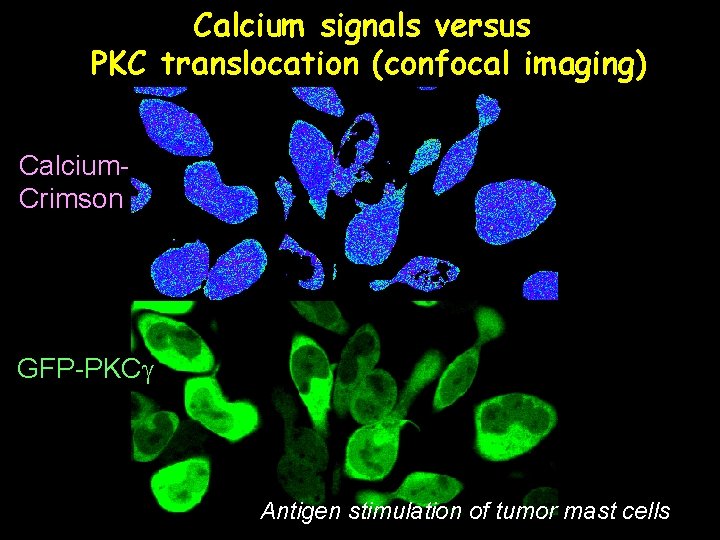
Calcium signals versus PKC translocation (confocal imaging) Calcium. Crimson GFP-PKCg Antigen stimulation of tumor mast cells
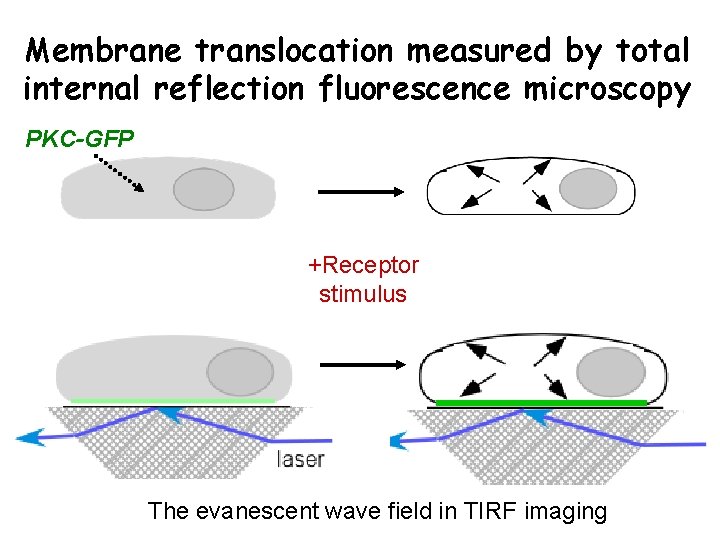
Membrane translocation measured by total internal reflection fluorescence microscopy PKC-GFP +Receptor stimulus The evanescent wave field in TIRF imaging
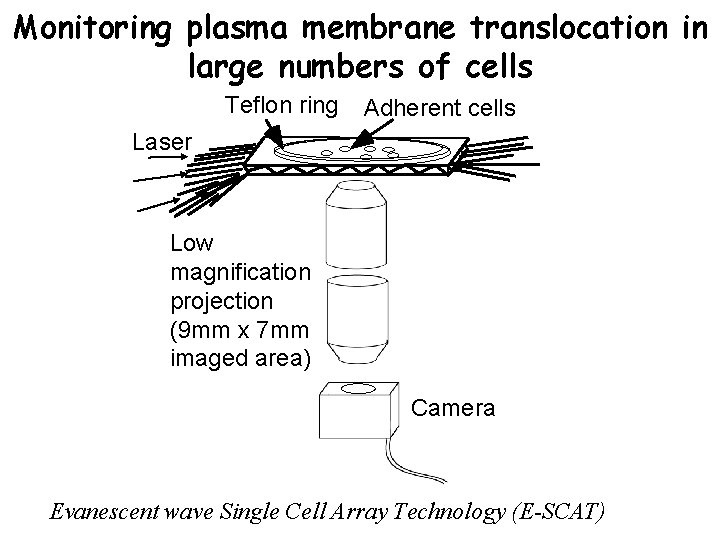
Monitoring plasma membrane translocation in large numbers of cells Teflon ring Adherent cells Laser Low magnification projection (9 mm x 7 mm imaged area) Camera Evanescent wave Single Cell Array Technology (E-SCAT)
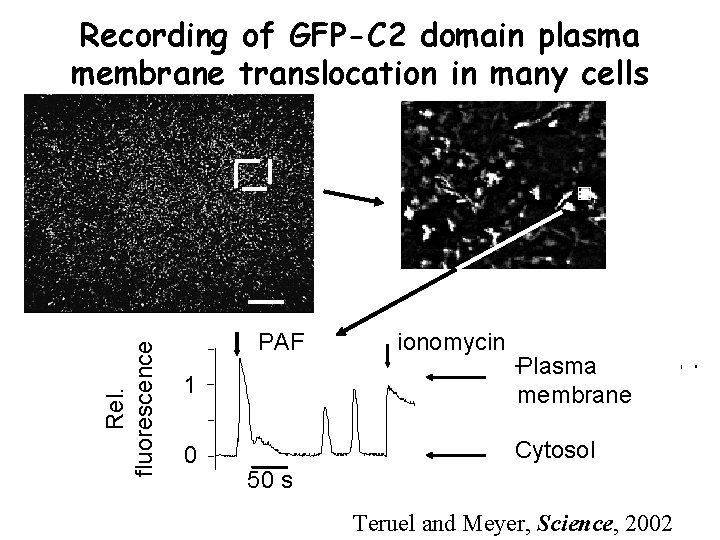
Rel. fluorescence Recording of GFP-C 2 domain plasma membrane translocation in many cells PAF ionomycin 1 Plasma membrane 0 Cytosol 50 s Teruel and Meyer, Science, 2002

2. Perturbation strategies suitable for microscopy 1. RNAi 2. Expression constructs (wt, DN, CA) 3. Small molecule perturbations (for example induced translocation)
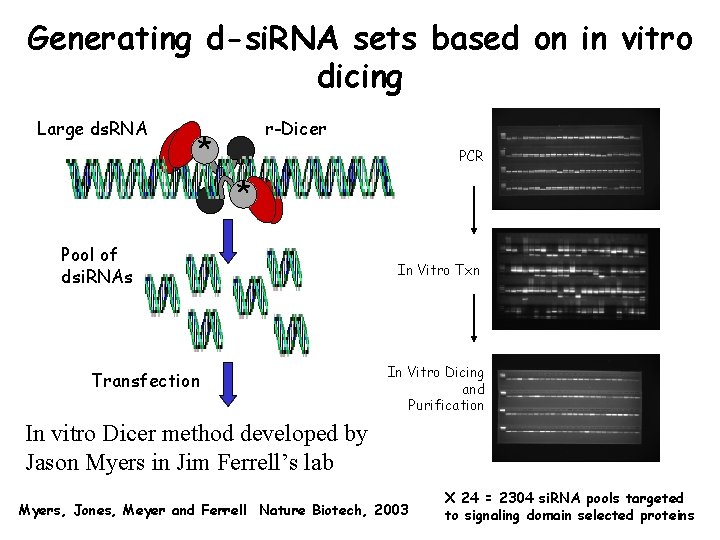
Generating d-si. RNA sets based on in vitro dicing Large ds. RNA r-Dicer * PCR * Pool of dsi. RNAs Transfection In Vitro Txn In Vitro Dicing and Purification In vitro Dicer method developed by Jason Myers in Jim Ferrell’s lab Myers, Jones, Meyer and Ferrell Nature Biotech, 2003 X 24 = 2304 si. RNA pools targeted to signaling domain selected proteins
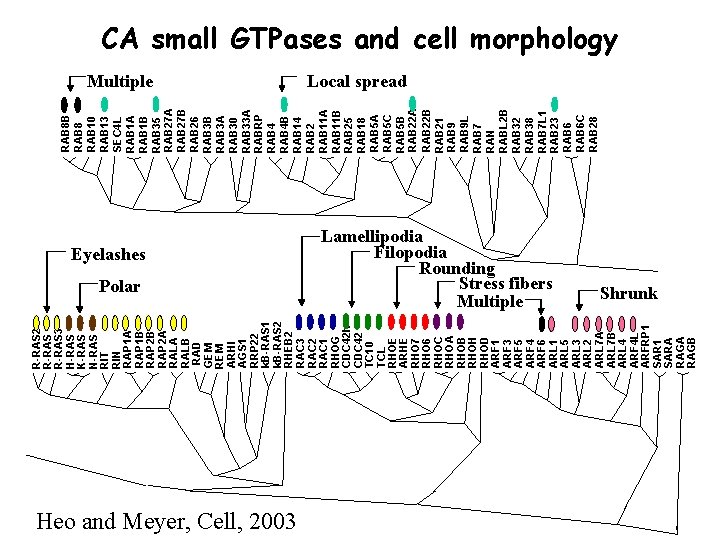
R-RAS 2 R-RAS 3 H-RAS K-RAS N-RAS RIT RIN RAP 1 A RAP 1 B RAP 2 A RALB RAD GEM REM ARHI AGS 1 RRP 22 k. B-RAS 1 k. B-RAS 2 RHEB 2 RAC 3 RAC 2 RAC 1 RHOG CDC 42 h CDC 42 TC 10 TCL RHOE ARHE RHO 7 RHO 6 RHOC RHOA RHOB RHOH RHOD ARF 1 ARF 3 ARF 5 ARF 4 ARF 6 ARL 1 ARL 5 ARL 3 ARL 2 ARL 7 A ARL 7 B ARL 4 ARF 4 L ARFRP 1 SARA RAGB RAB 8 RAB 10 RAB 13 SEC 4 L RAB 1 A RAB 1 B RAB 35 RAB 27 A RAB 27 B RAB 26 RAB 3 B RAB 3 A RAB 30 RAB 33 A RABRP RAB 4 B RAB 14 RAB 2 RAB 11 A RAB 11 B RAB 25 RAB 18 RAB 5 A RAB 5 C RAB 5 B RAB 22 A RAB 22 B RAB 21 RAB 9 L RAB 7 RAN RABL 2 B RAB 32 RAB 38 RAB 7 L 1 RAB 23 RAB 6 C RAB 28 CA small GTPases and cell morphology Multiple Eyelashes Polar Heo and Meyer, Cell, 2003 Local spread Lamellipodia Filopodia Rounding Stress fibers Multiple Shrunk
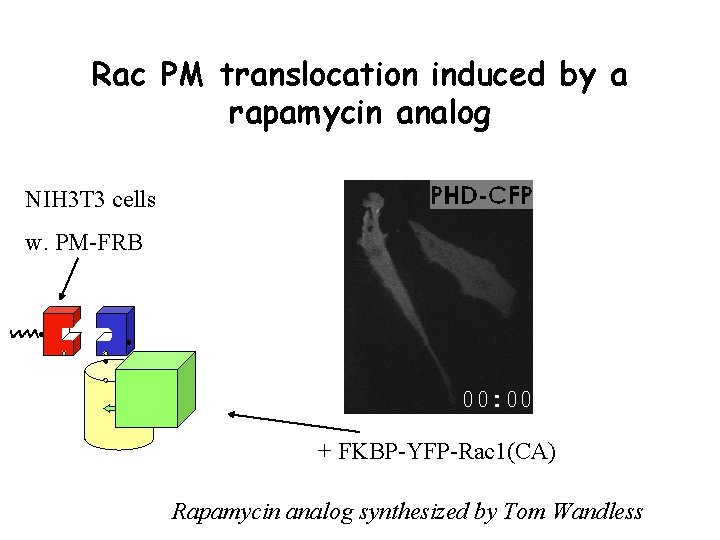
Rac PM translocation induced by a rapamycin analog NIH 3 T 3 cells w. PM-FRB + FKBP-YFP-Rac 1(CA) Rapamycin analog synthesized by Tom Wandless

3. Automated Microscopy Based Biosensors and Functional Assays 1. FRET Biosensors 2. Phospecific Antibody and Related Fixed Cell Assays 3. Translocation Biosensors 4. Live and Fixed Cell Functional Assays (Outputs) 5. Many critical assays are lacking
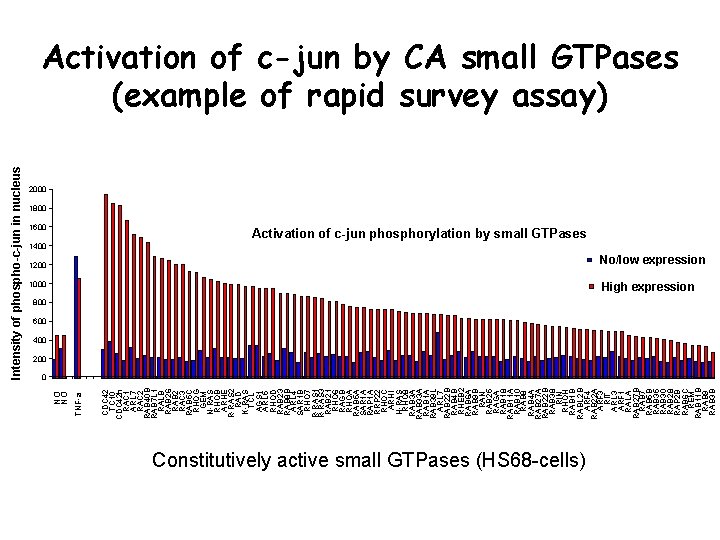
1600 CDC 42 TC 10 CDC 42 h RAC 1 ARL 7 RAC 2 RAB 40 B RAB 7 L 1 RALB RAB 26 RAB 2 RAC 3 RAB 5 C RHOG GEM N-RAS RHOB ARHE R-RAS 2 RAD K-RAS TCL AGS 1 ARL 5 RHOD RAB 23 RAB 8 B ARL 4 SAR 1 B RHO 7 R-RAS 1 R-RAS 3 RAB 21 RHO 6 RAGB RHOA RAB 5 A SAR 1 A RAP 1 A RRP 22 RHOC ARHI H-RAS RHO 8 RAB 3 A RAB 33 A RAB 1 A RAB 39 L ARL 7 RAB 22 B RAB 4 B RHEB 2 RAB 6 A RAB 9 B RAN RAB 25 RAGA RAB 18 RAB 11 A RAB 10 RAB 8 RAB 4 A RAB 27 A RAB 22 B RAB 38 RIN RHOH RAB 1 B RABL 2 B ARF 4 RAB 22 A ARF 3 RIT ARL 3 ARF 1 RALA RAB 27 B RAB 7 RAB 5 B RAB 35 RAB 30 RAB 28 RAP 2 B RAB 6 C REM RAB 11 B RAB 9 RAB 3 B TNF-a NO NO Intensity of phospho-c-jun in nucleus Activation of c-jun by CA small GTPases (example of rapid survey assay) 2000 1800 1400 Activation of c-jun phosphorylation by small GTPases 1200 No/low expression 1000 High expression 800 600 400 200 0 Constitutively active small GTPases (HS 68 -cells)
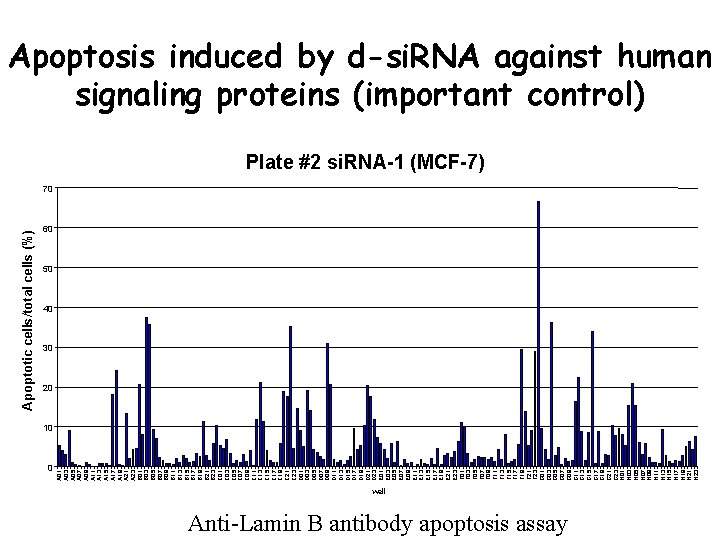
0 A 01 A 03 A 05 A 07 A 09 A 11 A 13 A 15 A 17 A 19 A 21 A 23 B 01 B 03 B 05 B 07 B 09 B 11 B 13 B 15 B 17 B 19 B 21 B 23 C 01 C 03 C 05 C 07 C 09 C 11 C 13 C 15 C 17 C 19 C 21 C 23 D 01 D 03 D 05 D 07 D 09 D 11 D 13 D 15 D 17 D 19 D 21 D 23 E 01 E 03 E 05 E 07 E 09 E 11 E 13 E 15 E 17 E 19 E 21 E 23 F 01 F 03 F 05 F 07 F 09 F 11 F 13 F 15 F 17 F 19 F 21 F 23 G 01 G 03 G 05 G 07 G 09 G 11 G 13 G 15 G 17 G 19 G 21 G 23 H 01 H 03 H 05 H 07 H 09 H 11 H 13 H 15 H 17 H 19 H 21 H 23 Apoptotic cells/total cells (%) Apoptosis induced by d-si. RNA against human signaling proteins (important control) Plate #2 si. RNA-1 (MCF-7) 70 60 50 40 30 20 10 well Anti-Lamin B antibody apoptosis assay

Fluorescent Translocation Biosensors: Non -perturbing versus endpoint indicators SH 2 -domains to monitor local tyrosine phosphorylation (Stauffer et al. , JCB 1997) C 1 -domains to monitor localized diacylglycerol signals (Oancea et al. , JCB 1998) C 2 -domains to monitor local Ca 2+/PS-signals (Oancea et al. , Cell 1998) PH-domains to monitor local changes of phosphoinositides (Stauffer et al. , Current Biology 1998, PLC-delta; Kontos et al. , Mol. Pharm. 1998, Akt) Potentially many other useful domains (FYVE, PTB, …)

Biosensors & Perturbations PH-domain selectivity PH Domain Binding selectivity CTH 3(PH 1 A) PI(3, 4, 5)P 3 Akt 3(PH 1 A) PLCd 1(PH 1 A) PI(3, 4, 5)P 3 PI(3, 4, )P 2 PI(4, 5)P 2 PI(3, 4, 5)P 3 Ral. GPS 2(PH 1 A) Hapip 1(PH 1 A) PI(3, 4)P 2 PI(4, 5)P 2 127 PH Domain constructs tested: PI(3, 4, 5)P 3 CTH 3(PH 1 A), Myo 10(PH 1 A), ITK(PH 1 A), H 056(PH 2 A), Et. OHD 4(PH 1 A), APS(PH 1 A), Afap(PH 1 A), TEC(PH 1 A) PI(3, 4)P 2 and PI(3, 4, 5)P 3 Gab 1(PH 1 A), Gab 2(PH 1 A), Bam 32(PH 1 A), CTH 2(PH 1 A), IRS-1(PH 1 A), Osbp 13(PH 1 A), Plek(PH 1 A), TNFidp(PH 1 A), Akt 2(PH 1 A), Akt 3(PH 1 A), Akt 1(PH 1 A), LL 5(PH 1 A), Arl 61(PH 1 A), BCRa(PH 1 a) PI(4, 5)P 2 and PI(3, 4, 5)P 3 PLCd 1(PH 1 A), Spnb 2(PH 1 A), Ral. GPS 2*(PH 1 A), Centb 5(PH 1 A), Cnk 2(PH 1 A) PI(3, 4)P 2 Plek 2(PH 2 A), Hapip 1(PH 1 A) Wei Sun Park, James Whalen, Takako Mukai & Nancy O’Rourke

PIP 3 production by constitutively active small GTPases Ras subfamily ECFP H-RAS K-RAS RALA RAP 2 B RHOH CDC 42 TC 10 RAB 23 RAB 30 ARF 1 Rho subfamily RAP 2 A RAC 1 RHOG Rab & Arf subfamily RAB 1 A RAB 2 Morphology changes make automated analysis more difficult
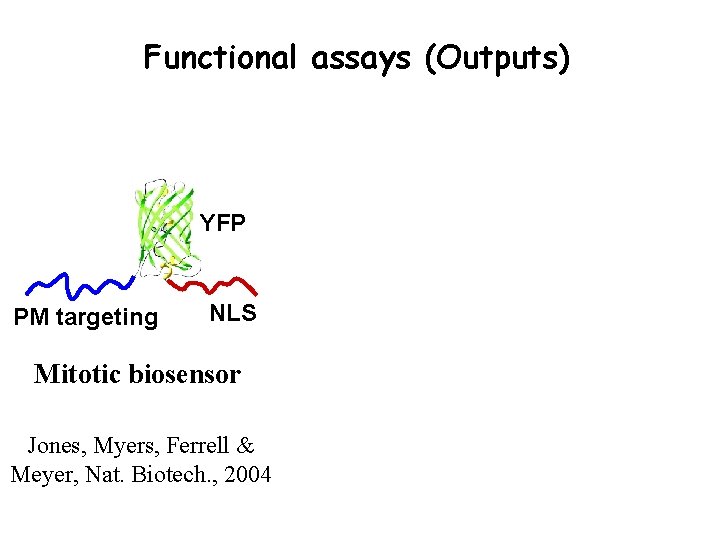
Functional assays (Outputs) YFP PM targeting NLS Mitotic biosensor Jones, Myers, Ferrell & Meyer, Nat. Biotech. , 2004

Watching 3 hours in the life of cycling cells Mitosis biosensor
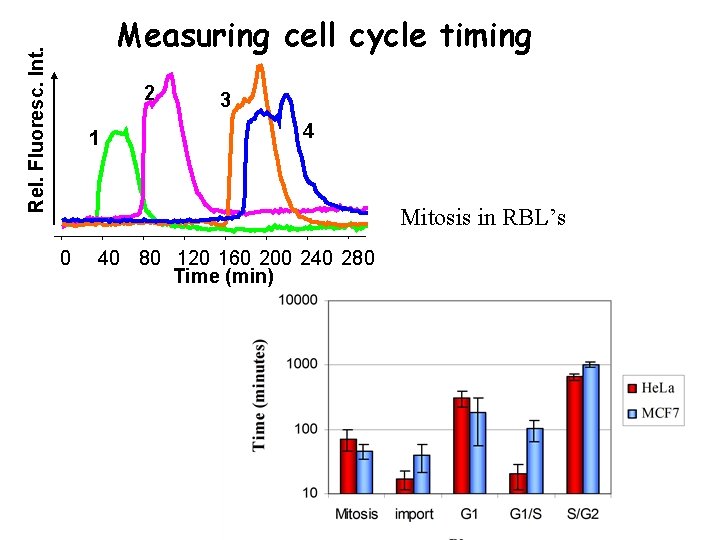
Rel. Fluoresc. Int. Measuring cell cycle timing 2 1 3 4 Mitosis in RBL’s 0 40 80 120 160 200 240 280 Time (min)
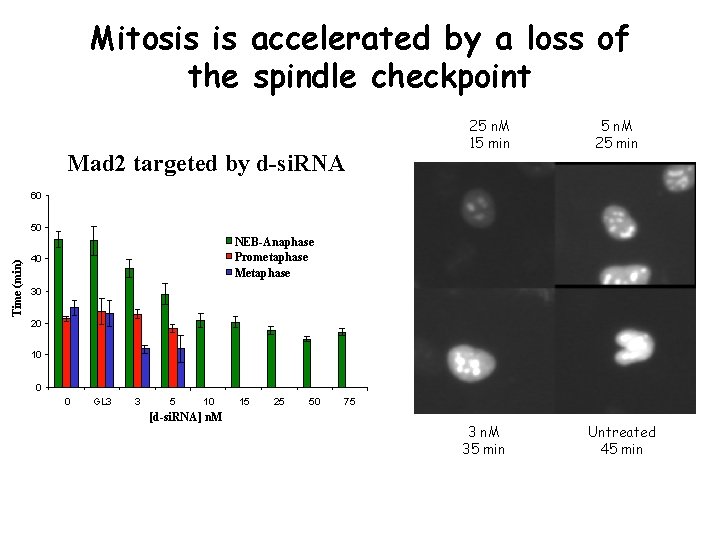
Mitosis is accelerated by a loss of the spindle checkpoint Mad 2 targeted by d-si. RNA 25 n. M 15 min 5 n. M 25 min 60 Time (min) 50 NEB-Anaphase Prometaphase Metaphase 40 30 20 10 0 0 GL 3 3 5 10 [d-si. RNA] n. M 15 25 50 75 3 n. M 35 min Untreated 45 min
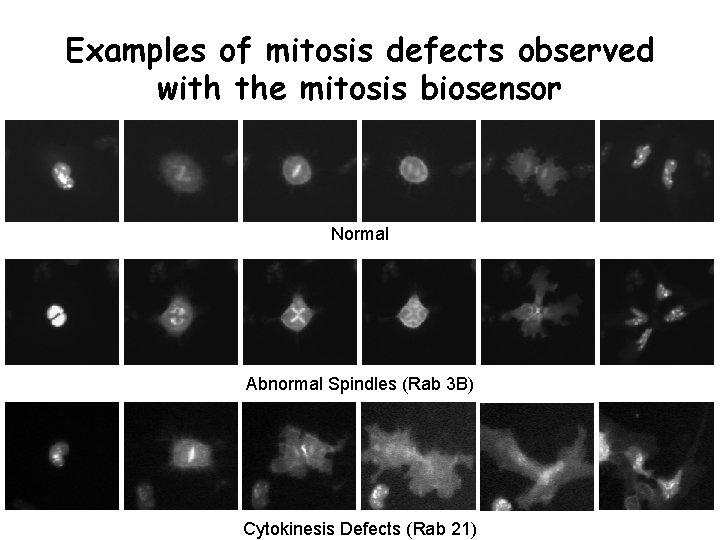
Examples of mitosis defects observed with the mitosis biosensor Normal Abnormal Spindles (Rab 3 B) Cytokinesis Defects (Rab 21)
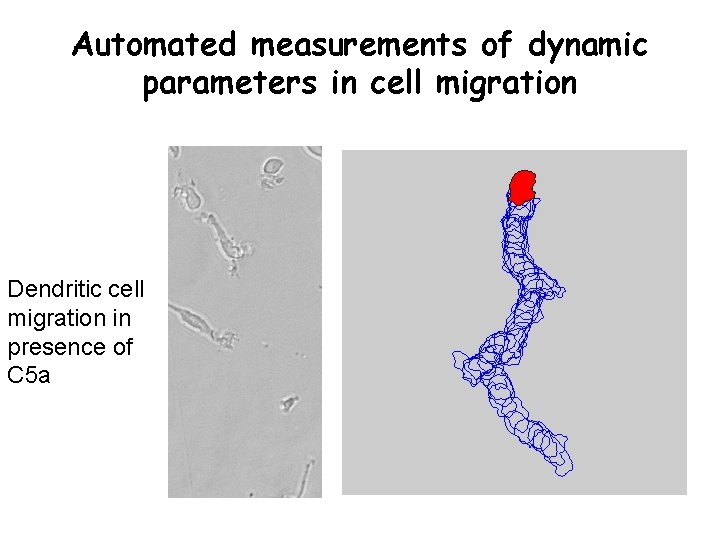
Automated measurements of dynamic parameters in cell migration Dendritic cell migration in presence of C 5 a
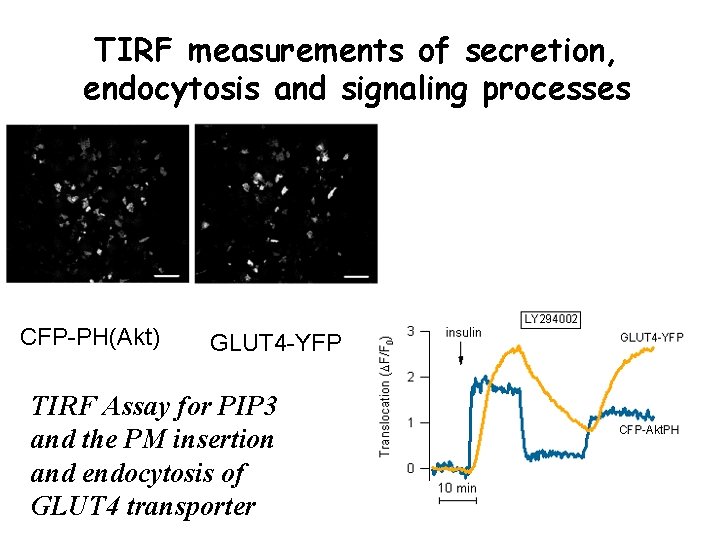
TIRF measurements of secretion, endocytosis and signaling processes CFP-PH(Akt) GLUT 4 -YFP TIRF Assay for PIP 3 and the PM insertion and endocytosis of GLUT 4 transporter

Acknowledgments Josh Jones Angie Hahn Calif. Ave. Af. CS Microscopy Onn Brandman lab: Annette Salmeen Grischa Chandy Nancy Cecile Arrieumerlou O’Rourke Wei Sun Takanari Inoue Park Jim Whalen Marc Fivaz Takako Mukai Madeleine Craske Mary Verghese Thierry Galvez Liz Gehrig Michael Bradshaw Sarah Lim Chuck Fink Mary Teruel James Ferrell, Jason Myers, Man Lyiang Kim Michal Ronen Won Do Heo Tom Wandless Jen Liou

Automated microscopy based signaling and functional assays 1. Phospecific antibodies and other fixed cell assays (analysis procedures can readily be developed; more suitable phospecific antibodies needed) 2. FRET biosensors (implementation of automated assays of existing biosensors is first needed) 3. Translocation biosensors (PM, nucleus, Golgi and vesicular structures could be automatically analyzed; development of new assays and implementation of automated assays needed) 4. Functional output assays (apoptosis, cell cycle, secretion endocytosis, phagocytosis, pinocytosis, cell migration, cell adhesion; Automated assays still need development) 5. Still fairly low biosensors coverage. More assays needed (how many? ). Microscopy can provide suitable assays for many of them.
- Slides: 30