MICROBIOLOGICAL TREATMENT PROGRAMS FOR COOLING WATER PRESENTER H


MICROBIOLOGICAL TREATMENT PROGRAMS FOR COOLING WATER PRESENTER H. M. ASIF MANAGEMENT ASSOCIATE OPERATIONS UTILITIES

OBJECTIVE To compare various microbiological treatment programs and select the best for our cooling water system.
CONTENTS Cooling water system Cooling water problems Microbiological growth Treatment for Microbiological growth
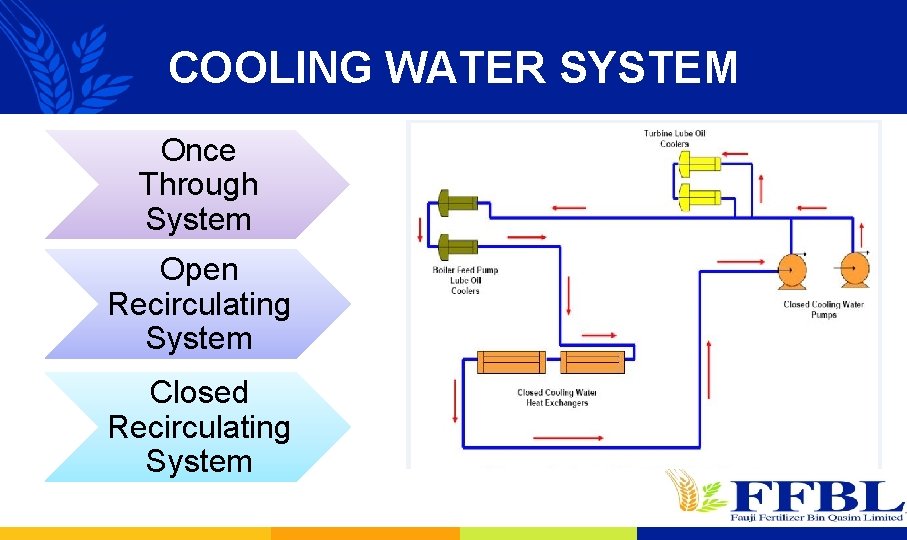
COOLING WATER SYSTEM Once Through System Open Recirculating System Closed Recirculating System

COOLING WATER PROBLEMS Scaling Fouling Corrosion Microbial Growth
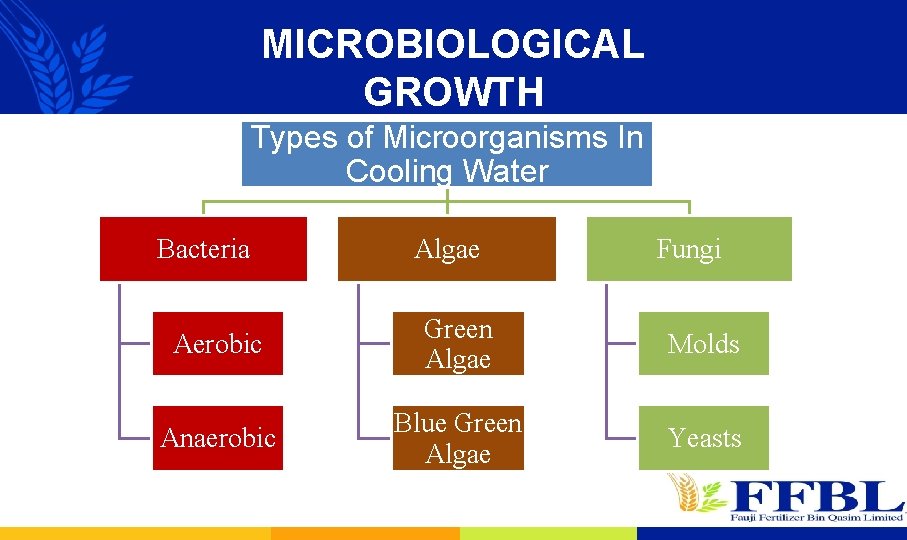
MICROBIOLOGICAL GROWTH Types of Microorganisms In Cooling Water Bacteria Algae Fungi Aerobic Green Algae Molds Anaerobic Blue Green Algae Yeasts
MICROBIOLOGICAL GROWTH MECHANISMS Binary Fission Budding Tip Extension
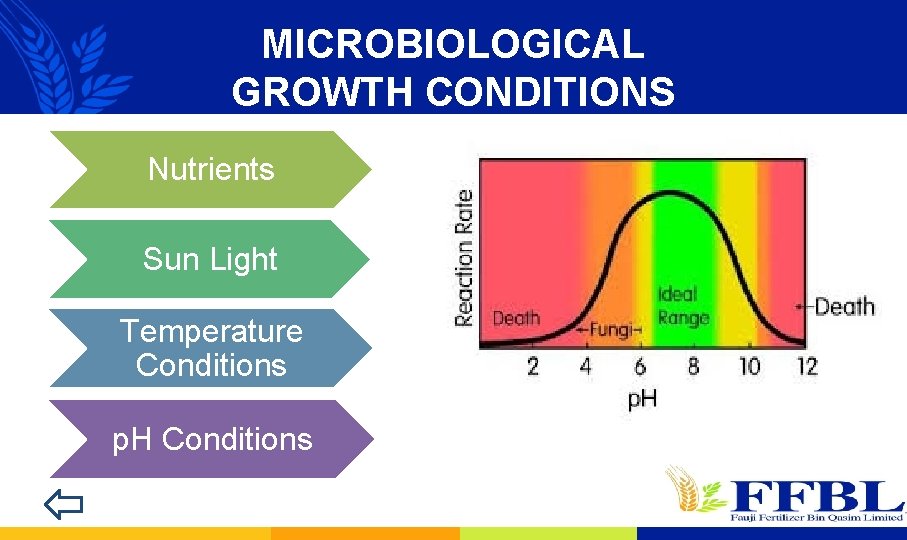
MICROBIOLOGICAL GROWTH CONDITIONS Nutrients Sun Light Temperature Conditions p. H Conditions
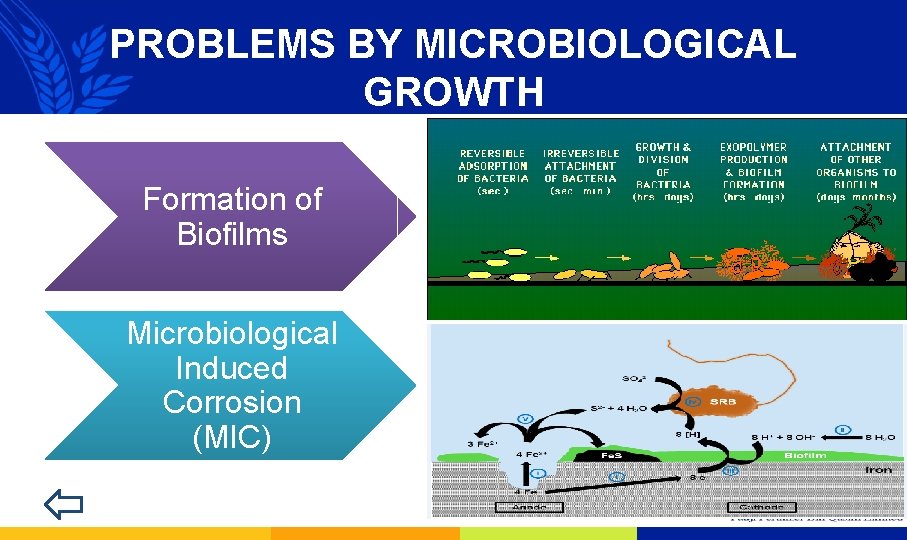
PROBLEMS BY MICROBIOLOGICAL GROWTH Formation of Biofilms Microbiological Induced Corrosion (MIC)

MICROBIOLOGICAL TREATMENT Oxidizing Biocides Non oxidizing biocides Ozone Chlorine Dioxide Chlorine Bromine Monochloramines Isothiazoline Carbamates Quaternary amines
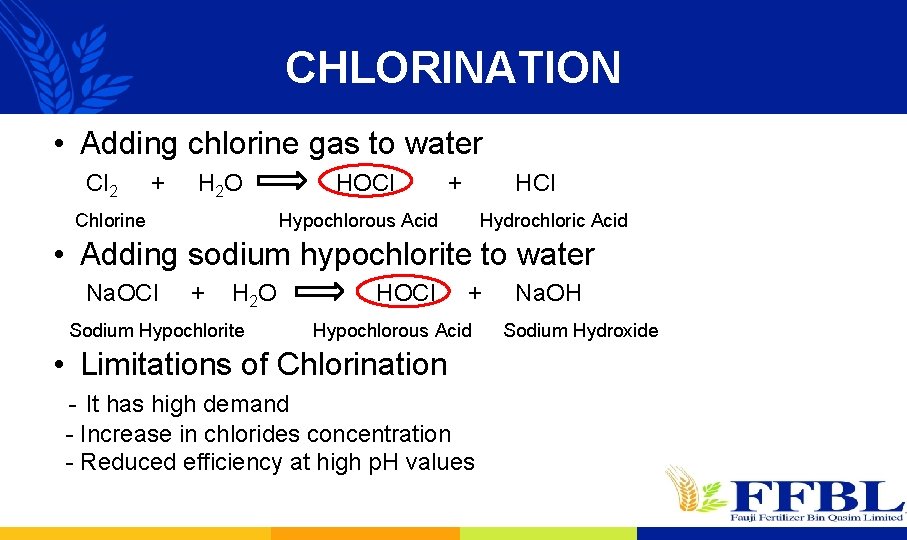
CHLORINATION • Adding chlorine gas to water Cl 2 + H 2 O Chlorine HOCl + HCl Hypochlorous Acid Hydrochloric Acid • Adding sodium hypochlorite to water Na. OCl + H 2 O Sodium Hypochlorite HOCl + Hypochlorous Acid • Limitations of Chlorination - It has high demand - Increase in chlorides concentration - Reduced efficiency at high p. H values Na. OH Sodium Hydroxide

BROMINATION • Sodium bromide reacts with hypochlorous acid Na. Br + Sodium Bromide HOCl Hypochlorous Acid HOBr + Hypobromous Acid • Benefits of Bromination over Chlorination. - Better effectiveness at high p. H than chlorine • Limitations of Bromination - Use with parallel of chlorine - Increase in chlorides concentration Na. Cl

MONOCHLORAMINES • Formation of Monochloramine: HOCl + NH 3 NH 2 Cl + H 2 O • Characteristics of monochloramines - It has Low demand - Not affected by suspended solids and organic compounds - Excellent biofilm penetration and removal - Effective at wide range of p. H - Effective up to and above temperature of 60 ºC

ADVANTAGES OF MONOCHLORAMINES • • • Elimination of Chlorine Reduction of Sodium Hypochlorite consumption Chlorides reduction Blow down reduction Reduction in Bulab chemical consumption

DISADVANTAGES OF MONOCHLORAMINES The only disadvantage of Monochloramines is the formation of Dichloramines and Trichloramines. NH 2 Cl + HOCl NHCl 2 + H 2 O NHCl 2 + HOCl NCl 3 + H 2 O
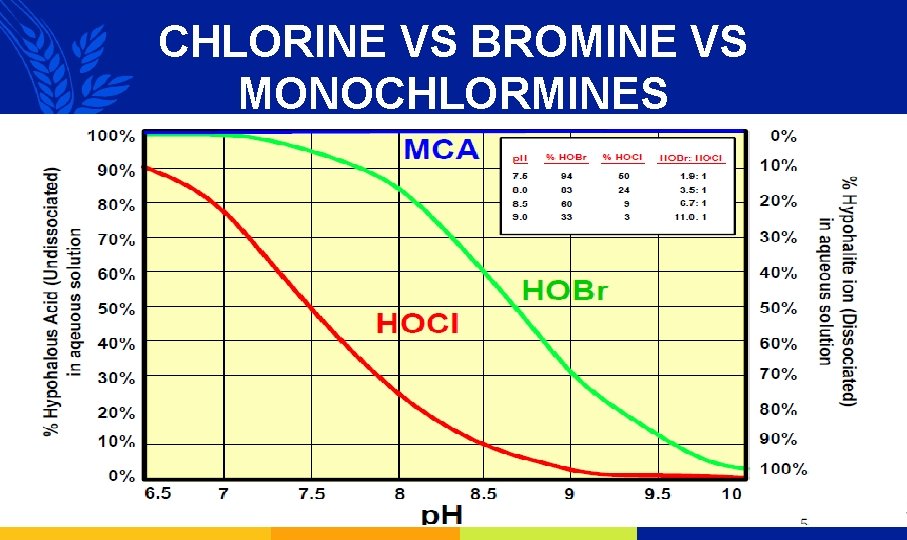
CHLORINE VS BROMINE VS MONOCHLORMINES
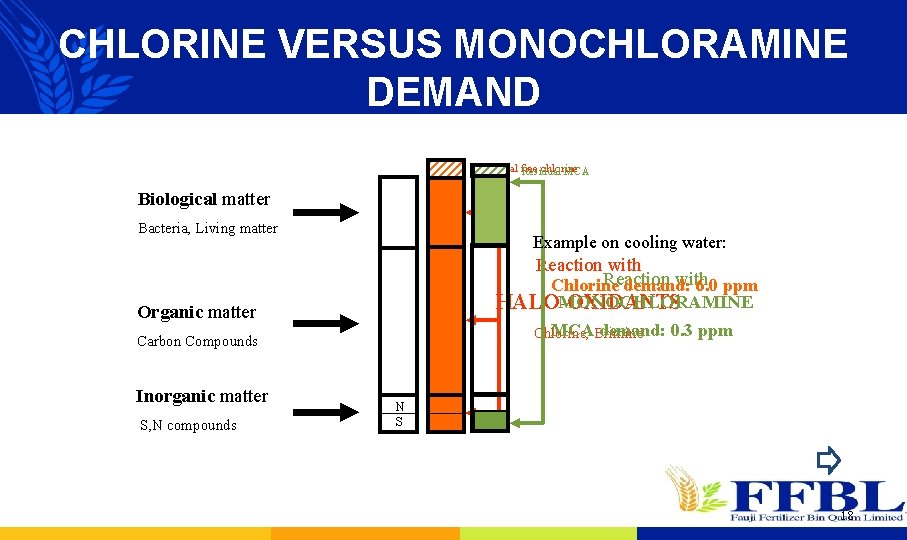
CHLORINE VERSUS MONOCHLORAMINE DEMAND Residual free chlorine Residual MCA Biological matter Bacteria, Living matter Example on cooling water: Reaction with Chlorine demand: 6. 0 ppm MONOCHLORAMINE HALO-OXIDANTS Organic matter MCABrimine demand: 0. 3 ppm Chlorine, Carbon Compounds Inorganic matter S, N compounds N S 18
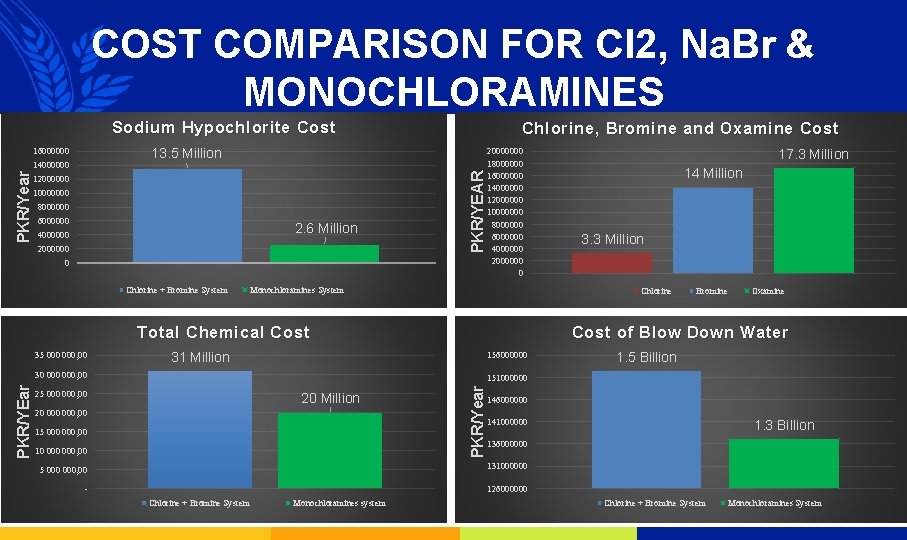
COST COMPARISON FOR Cl 2, Na. Br & MONOCHLORAMINES Sodium Hypochlorite Cost 14000000 12000000 10000000 8000000 6000000 2. 6 Million 4000000 2000000 PKR/YEAR 13. 5 Million 16000000 PKR/Year Chlorine , Bromine and Oxamine Cost 0 Chlorine + Bromine System 20000000 18000000 16000000 14000000 12000000 10000000 8000000 6000000 4000000 2000000 0 Monochloramines System 3. 3 Million Bromine Oxamine Cost of Blow Down Water 31 Million 156000000 30 000, 00 1. 5 Billion 151000000 25 000, 00 20 Million 20 000, 00 15 000, 00 10 000, 00 PKR/Year PKR/YEar 14 Million Chlorine Total Chemical Cost 35 000, 00 17. 3 Million 146000000 141000000 136000000 5 000, 00 131000000 - 126000000 Chlorine + Bromine System Monochloramines system 1. 3 Billion Chlorine + Bromine System Monochloramines System

CONCLUSION From the comparison we can say that Monochloramines system is a better choice for the microbiological treatment as: “It is a more efficient and more economical system”

Questions are Welcome ? ? ?
- Slides: 21