Microbial Toxins BIOC 422 Fungal Toxins Dr Ayat

Microbial Toxins (BIOC 422) Fungal Toxins Dr. Ayat B. Al-Ghafari Associate Professor of Biomedical Sciences e. mail: abalghafari@kau. edu. sa

About fungi Fungi are not plants! • Fungi were first officially recognized as a distinct group in 1969. • more recently, using DNA sequences and comparisons of cell structure, we have learned that Fungi are in fact more closely related to animals than they are to plants. • Superficially, they remind us more of plants than animals because they don’t move, but this is the only thing they have in common. How do Fungi eat? • Unlike plants, which make their own food, Fungi are like miniature versions of our stomachs, turned inside-out. • Fungi 'eat' by releasing enzymes outside of their bodies that break down nutrients into smaller pieces that they can then absorb. • This feeding strategy means that Fungi always live in and on their food

About fungi Where does a fungus live? • Everywhere: Antarctica, the Amazon jungle, the Gobi desert, and even all over (and inside) you! • Fungi are amazingly well adapted to just about any condition on Earth. • More than 70, 000 species of fungi have been identified. • The organisms in kingdom fungi include: mushrooms, yeasts, and moulds.

Mould vs. Yeast Mould • Grows in multicellular filaments called hyphae. • Found in damp, dark, or steam-filled area. • Reproduction through small pores which can be either sexual or asexual. • Has a fuzzy appearance and can be orange, green, black, brown and can found in several shapes. • Some moulds are used in food production (e. g. penicillium is used in production of cheese). • Secrete hydrolytic enzymes that degrades biopolymers such as starch and cellulose. Yeast • Grows as a single cell • Found on fruits, in the stomach of mammals and on skin, among other places. • Most reproduce asexually through mitosis. • White and thready, usually oval in shape. • Ethanol production, baking, vitamins, study of cell cycle. • Convert carbohydrates to alcohol and carbon dioxide in anaerobic through fermentation.

About fungi Many Fungi are useful to humans: ØAlthough we often think of fungi as organisms that cause disease and rot food, fungi are important to human life on many levels. ØThey provide fundamental products including foods, medicines, and enzymes important to industry. üRecycling: Fungi, together with bacteria, are responsible for most of the recycling which returns dead material to the soil in a form in which it can be reused.

About fungi Many Fungi are useful to humans: § Food: üMany mushrooms are edible and different species are cultivated for sale worldwide. üStinky cheeses – blue cheese and camembert § Medicines: Penicillin, perhaps the most famous of all antibiotic drugs, is derived from a common fungus called Penicillium. § Yeasts: baking and brewing § Many organic acids are produced with the help of fungi ex. citric acid in Coke is made with a fungus

Fungal toxins Toxins from fungi are widely grouped into toxins from moulds ("mycotoxins") and toxins from higher fungi ("mushroom poisons"). https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/ I. Mycotoxins:

Fungal toxins I. Mycotoxins: There are many definitions for what a mycotoxin is: Ø It is a "natural products from moulds that evoke a toxic response when introduced in low concentrations to vertebrates". Ø Mycotoxins are products of a fungus' secondary metabolism, i. e. that part of fungal metabolism that is not essential for cell growth and maintenance of basic cell function. Why fungi produce such substances is not entirely clear, but they may, at least in part, be used for "chemical warfare" and thus provide some advantage to survive in the environment.

Fungal toxins I. Mycotoxins: ØMycoses vs Mycotoxicoses: Frank growth of fungi on animal hosts produces the diseases called MYCOSES, while dietary, respiratory, dermal, and other exposures to toxic fungal metabolites produce the diseases called MYCOTOXICOSES. § Mycoses range from merely annoying (e. g. , athlete's foot) to life-threatening (e. g. , invasive aspergillosis). § Mycotoxicoses are examples of “poisoning by natural means” and thus are similar to the pathologies caused by exposure to pesticides or heavy metal residues. In Summary: Mycosis is tissue invasion by a fungi and mycotoxicoses is toxin production by a fungi growing on foodstuffs which is then ingested causing disease. Ø The production of toxins (mycotoxins) depends on the surrounding intrinsic and extrinsic environments Ø The severity of the toxins varies greatly, depending on the organism infected and its susceptibility, metabolism, and defence mechanisms.

Fungal toxins I. Mycotoxins: Ø Major mycotoxins • Aflatoxins • Ochratoxin • Citrinin • Ergot Alkaloids • Fumonisins • Patulin https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/

I. Mycotoxins: Ø Major mycotoxins 1) Aflatoxins What are aflatoxins? Fungal toxins • Aflatoxins are a family of toxins produced by certain fungi that are found on agricultural crops such as maize (corn), peanuts, cottonseed, and tree nuts. • The main fungi that produce aflatoxins are Aspergillus flavus and Aspergillus parasiticus, which are abundant in warm and humid regions of the world. • There are four major aflatoxins: B 1, B 2, G 1, G 2 plus two additional metabolic products, M 1 and M 2 >> The metabolites, M 1 & M 2, are found in milk >> The B designation of aflatoxins B 1 and B 2 resulted from the exhibition of blue fluorescence under UV-light, while the G designation refers to the yellow-green fluorescence of the relevant structures under UV-light. https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/

Fungal toxins I. Mycotoxins: Ø Major mycotoxins 1) Aflatoxins Aflatoxin Occurrence: • Aflatoxins can contaminate corn, cereals, sorghum, peanuts and other oil-seed crops. Thus, food contamination by this group of mycotoxins has been implicated in both animal and human Aflatoxicosis. • Milk, cheese and other dairy products are at risk of contamination by aflatoxin M. • Aflatoxin-producing fungi can contaminate crops in the field, at harvest, and during storage. https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/

I. Mycotoxins: Ø Major mycotoxins 1) Aflatoxins Health effects: Fungal toxins • Aflatoxin is associated with both toxicity and carcinogenicity in human and animal populations • The diseases caused by aflatoxin consumption are loosely called aflatoxicoses. ü Acute aflatoxicosis results in death: § At high enough exposure levels, aflatoxins can cause acute toxicity, and potentially death, in mammals, birds and fish, as well as in humans. § The liver is the principal organ affected, but high levels of aflatoxin have also been found in the lungs, kidneys, brains and hearts of individuals dying of acute aflatoxicosis. *Acute conditions are severe and sudden in onset. **A chronic condition, is a long-developing syndrome. https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/

I. Mycotoxins: Ø Major mycotoxins 1) Aflatoxins Health effects: Fungal toxins ü Chronic aflatoxicosis results in cancer, immune suppression, and other “slow” pathological conditions. § Chronic toxicity is probably more important from a food safety point of view, certainly in more developed regions of the world. § Aflatoxin B 1 is a very potent carcinogen and a mutagen in many animals, and therefore potentially in humans, and the liver is again the main target organ. § Ingestion of low levels over a long period has been implicated in primary liver cancer, chronic hepatitis, jaundice, cirrhosis and impaired nutrient conversion § Less is known about the chronic toxicity of aflatoxin G 1 and M 1, but these are also thought to be carcinogens, though probably a little less potent than B 1. *Acute conditions are severe and sudden in onset. **A chronic condition, is a long-developing syndrome. https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/

Carcinogenic effect: Aflatoxin may exert its hepatocellular carcinogenic activity through p 53 dysregulation • In the body, After ingestion, aflatoxin is metabolized by cytochrome p 450 group of enzymes in the liver, >> where it is bio-transformed to various metabolites; especially the active AFB 1 -exo-8, 9 -epoxide (AFBO). {AFB 1 requires metabolic conversion to its exo-8, 9 -epoxide in order to damage DNA } • The AFB, AFBO and other metabolites interact with various biomolecules in the body including nucleic acids such as DNA and RNA and the various metabolic pathways such as protein synthesis, glycolytic pathway and electron transport chain involved in ATP production in body cells. • The AFB -DNA interaction forms AFB-DNA adduct >> causes a transversion (the substitution of a purine for a pyrimidine or vice versa, in DNA) at codon 249 in p 53 gene in liver >> this mutation will inactivate p 53 >> this inactivation gives cells a selective growth advantage >> which may lead to hepatic carcinoma. A DNA adduct is a segment of DNA bound to a cancer-causing chemical. This process could be the start of a cancerous cell, or carcinogenesis. What is p 53? p 53 is a transcriptional activator that has been shown to regulate the cell cycle, to play a role in the apoptosis pathway and to be involved with DNA repair >> therefore it is an important tumor-suppressor gene.

Fungal toxins I. Mycotoxins: Ø Major mycotoxins 1) Aflatoxins Health effects: • In general, little is known about the level of dietary exposure to aflatoxins necessary to affect health, especially in humans, and diagnosis of chronic toxicity is very difficult. • It is generally agreed that the best approach is to minimise the levels in all foods as far as is technically possible and to assume that any dietary exposure is undesirable. https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/

Fungal toxins I. Mycotoxins: Ø Major mycotoxins 1) Aflatoxins How are people exposed to aflatoxins? People can be exposed to aflatoxins by: • eating contaminated plant products (such as peanuts/corn) • consuming meat or dairy products from animals that ate contaminated feed. • Farmers and other agricultural workers may be exposed by inhaling dust generated during the handling and processing of contaminated crops and feeds https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/

Fungal toxins I. Mycotoxins: Ø Major mycotoxins 1) Aflatoxins Are they stable in foods? • Aflatoxins are quite stable compounds and survive at relatively high temperatures with little degradation. • Their heat stability is influenced by other factors, such as moisture level and p. H, but heating or cooking processes cannot be relied upon to destroy aflatoxins. For example, roasting green coffee at 180°C for 10 minutes gave only a 50% reduction in aflatoxin B 1 level. • The stability of aflatoxin M 1 in milk fermentation processes has also been studied and although appreciable losses do occur, significant quantities of the toxin were found to remain in both cheese and yoghurt.

Fungal toxins I. Mycotoxins: Ø Major mycotoxins 1) Aflatoxins How can aflatoxin exposure be reduced? • You can reduce your aflatoxin exposure by buying only major commercial brands of nuts and nut butters. • Discard nuts that look mouldy, discoloured, or shrivelled. • To date, no outbreak of human illness caused by aflatoxins has been reported in the United States, but such outbreaks have occurred in some developing countries.

• https: //www. youtube. com/watch? v=47 r. QYo. ETWm 4
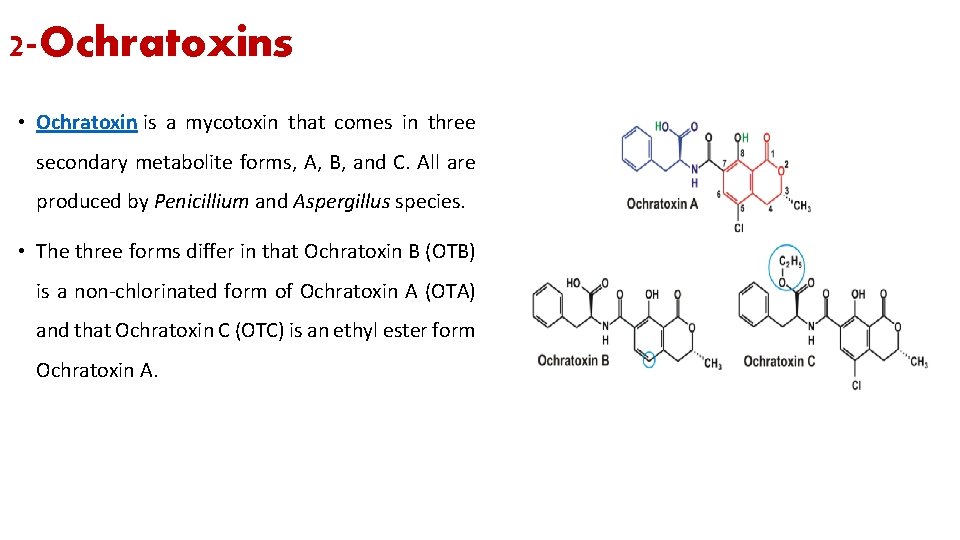
2 -Ochratoxins • Ochratoxin is a mycotoxin that comes in three secondary metabolite forms, A, B, and C. All are produced by Penicillium and Aspergillus species. • The three forms differ in that Ochratoxin B (OTB) is a non-chlorinated form of Ochratoxin A (OTA) and that Ochratoxin C (OTC) is an ethyl ester form Ochratoxin A.

2 - Ochratoxins • Ochratoxin A is the most prevalent and relevant fungal toxin of this group, while ochratoxins B and C are of lesser importance. • The principal source of ochratoxins is cereals, coffee, dried fruit especially raisins, and red wine which can cause ochratoxicosis if you ingested it. • The literature does not contain case reports of acute toxicity caused by ochratoxin in humans. However, it has been shown to be a potent nephrotoxin (acute toxicity in mammalian kidneys) in all animal species tested. Additionally, animal studies have shown that ochratoxin is hepatotoxic, an immunosuppressant, a teratogen, and a carcinogen.

2 - Ochratoxins • Geographically, ochratoxins are found in regions having temperate climates, with the northern European countries, the Balkans, and Canada being most affected. • Optimal conditions for ochratoxin A production are a moisture content of 19– 22% and a temperature of 24°C. • LD 50 of ochratoxin A about 20– 25 mg kg causing kidney damage, damage including degeneration of the proximal tubule in many animal species. Liver necrosis and enteritis were also observed.

2 - Ochratoxins • OTA or its metabolites; 4 -hydroxyochratoxin inhibits protein synthesis or cell growth >> by inhibiting the aminoacylation of phenylalanine t. RNA catalyzed by phenylalanyl-t. RNA synthetase/ligase >> which ultimately inhibits RNA synthetase activity by binding competitively at the site for phenylalanine. • It was suggested that supplementation with L-phenylalanine in OTA-exposed animals revealed a protective effect against its toxic effect.

3 - Citrinin • Citrinin is a mycotoxin that was first isolated from Penicillium citrinum, but has been identified in over a dozen species of Penicillium and several species of Aspergillus. • Some of these species are used to produce human foodstuffs such as cheese (Penicillium camemberti), miso (paste made from fermented soybeans and barley or rice or malt, used in Japanese cooking), and soy sauce (Aspergillus oryzae). • Research has shown that the kidney is the main target organ of citrinin. It shows change in histopathology and mild morbidity of the rat's kidney, disruption of the renal function in rats. Recent studies show that the mitochondria respiratory system is another target of citrinin. Citrinin can interfere with the electron transport system, Ca 2+ fluxes and membrane permeability • Citrinin induces different toxic effects, such as nephrotoxicity, hepatotoxicity, carcinogenicity and genotoxicity (explained in next slide). it is mainly found in long-stored grains, grains but sometimes also in fruits and other plant products. • Citrinin is associated with yellow rice disease in Japan and acts as a nephrotoxin in all animal species tested. • Although it is associated with many human foods (wheat, rice, corn, barely, oats, rye), its full significance for human health is unknown. • It has been shown that citrinin can also act synergistically with Ochratoxin A to depress RNA synthesis in murine kidneys.

3 - Citrinin • Acute toxicity • The acute toxicity of citrinin depends on the route of administration and on the species used for the research • Oral administration required the highest dose for lethality and the LD 50 of this administration route is 134 mg/kg bodyweight (b. w. ) for rabbit (Hanika, C; Carlton, WW; Tuite, J; 1983). Intravenous administration required the lowest dose for lethality and LD 50 is 19 mg/kg b. w. for rabbit (Ambrose, AM; De. Eds, F; 1946). • Nephrotoxicity and carcinogenicity • In a study with male rats, it was found that the rats showed an increased ratio of kidney weight to body weight after an exposure of 70 mg citrinin/kg b. w. for 32 weeks and an increase in the ratio of liver weight to body weight after an exposure of 80 weeks. After an exposure of 40 weeks to citrinin the rats also showed small adenoma (abenign tumor of epithelial tissue with glandular origin; Arai, M; Hibino, T (1983). • Genotoxicity • In mammalian cells in vitro, citrinin did not induce DNA single-strands breaks, oxidative DNA damage but induced micronuclei, aneuploidy (is the presence of an abnormal number of chromosomes in a cell) and chromosomal abberations (is a missing, extra, or irregular portion of chromosomal DNA). In vivo it induced chromosome abnormalities and hypodiploidy (having slightly fewer than the diploid number of chromosomes) in the bone marrow of mice. This indicates that citrinin is mutagenic.
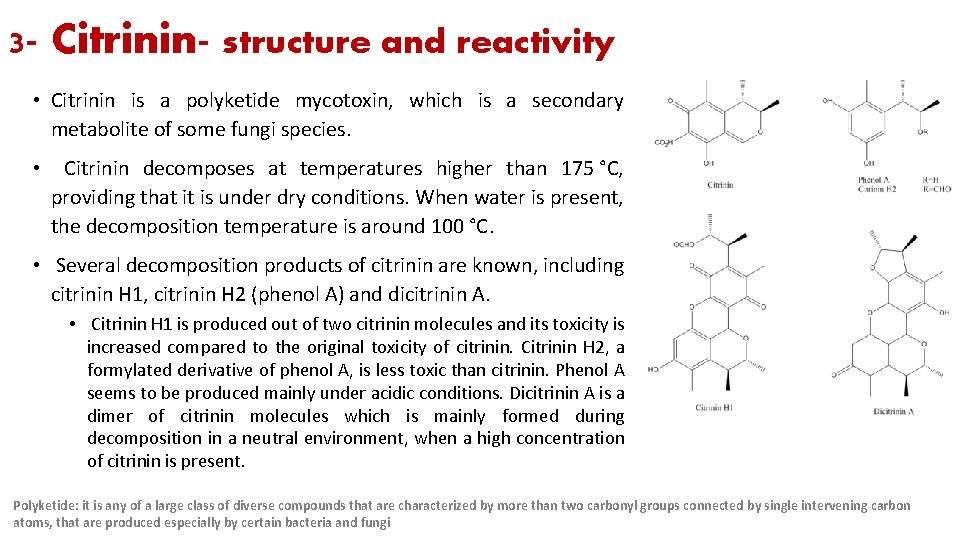
3 - Citrinin- structure and reactivity • Citrinin is a polyketide mycotoxin, which is a secondary metabolite of some fungi species. • Citrinin decomposes at temperatures higher than 175 °C, providing that it is under dry conditions. When water is present, the decomposition temperature is around 100 °C. • Several decomposition products of citrinin are known, including citrinin H 1, citrinin H 2 (phenol A) and dicitrinin A. • Citrinin H 1 is produced out of two citrinin molecules and its toxicity is increased compared to the original toxicity of citrinin. Citrinin H 2, a formylated derivative of phenol A, is less toxic than citrinin. Phenol A seems to be produced mainly under acidic conditions. Dicitrinin A is a dimer of citrinin molecules which is mainly formed during decomposition in a neutral environment, when a high concentration of citrinin is present. Polyketide: it is any of a large class of diverse compounds that are characterized by more than two carbonyl groups connected by single intervening carbon atoms, that are produced especially by certain bacteria and fungi

3 - Citrinin- Coexposure with ochratoxin A • Citrinin often occurs together with other mycotoxins like ochratoxin A or aflatoxin B 1 because they are produced by the same fungi species. • The combination which is observed most often is citrinin with ochratoxin A and this is also the most studied combination. The effects of co-occurrence of these mycotoxins are either additive or synergistic The nephrotoxic effects of ochratoxin A and citrinin, for example, are increased synergistic when exposure to both takes place. • Next to that, the co-exposure of these compounds is expected to be involved in the pathogenesis of a human kidney disease, called Balkan Endemic Nephropathy. The interaction of both substances might also influence apoptosis and necrosis in hepatocytes.

3 - Citrinin- mechanism of action Various in vitro studies have revealed • the involvement of citrinin toxicity in reduced cytokine production • inhibition of RNA and DNA synthesis • induction of oxidative stress • inhibition of nitric oxide gene expression • increase in reactive oxygen species (ROS) production • activation of apoptotic cell death via signal transduction pathways and the caspase-cascade system
What are alkaloids? § Alkaloid is an active nitrogen containing basic compounds of plant origin. § They are produced within various organisms including bacteria, fungi, plants, and animals as a secondary metabolite. § Alkaloids have a wide range of pharmacological activities including antiasthma, anticancer, analgesic, antihyperglycemia. § The plants use alkaloids as defense chemical agents against herbivores and pathogens.

I. Mycotoxins: Ø Major mycotoxins 4). Ergot Alkaloids Fungal toxins What are Ergot Alkaloids? Ø Ergot alkaloids, in general, are mycotoxins produced by several species of fungi in the genus Claviceps. Ø Ergot fungus contains a number of harmful substances collectively called the ergot alkaloids. What is alkaloids? § Alkaloid is an active nitrogen containing basic compounds of plant origin. § They are produced within various organisms as a secondary metabolite. Secondary metabolites are most commonly produced in plants as a defence system. The alkaloids produced by fungi are often toxic. The plants use alkaloids as defense chemical agents against herbivores and pathogens. The fungi produce alkaloids such as Ergotamine, Ergometrine, Ergonaline and various other Clavine alkaloids, are often toxic. . http: //www. apsnet. org/edcenter/intropp/topics/Mycotoxins/Pages/Ergot. Alkaloids. aspx https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/

Fungal toxins 4). Ergot Alkaloids What are Ergot Alkaloids? What is Ergot? • Ergot “or ergot fungi” refers to a group of fungi of the genus “Claviceps”. ü The most prominent member of this group is (Claviceps purpurea): ü Claviceps purpurea contaminates rye and wheat >> it produces *alkaloids substances called *ergotamines https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 164220/ http: //www. apsnet. org/edcenter/intropp/topics/Mycotoxins/Pages/Ergot. Alkaloids. aspx

Fungal toxins 4). Ergot Alkaloids What are Ergot Alkaloids? What is Ergotamine? • Ergotamine is one of the alkaloids that is produced by Claviceps purpurea • It has the biological activity as a vasoconstrictor. . (which means it constricts the blood vessels and thus the blood flow) • Ergotamine, in a proper dose, has been used in medicine: Ø For the treatment of acute migraine attacks. Ø They have also been used and misused to induce abortion. Ø It has been used to prevent post-partum hemorrhage (bleeding after child birth). • In excess, ergotamine can cause>> Ergot poisoning in humans and domestic animals that is known as Ergotism
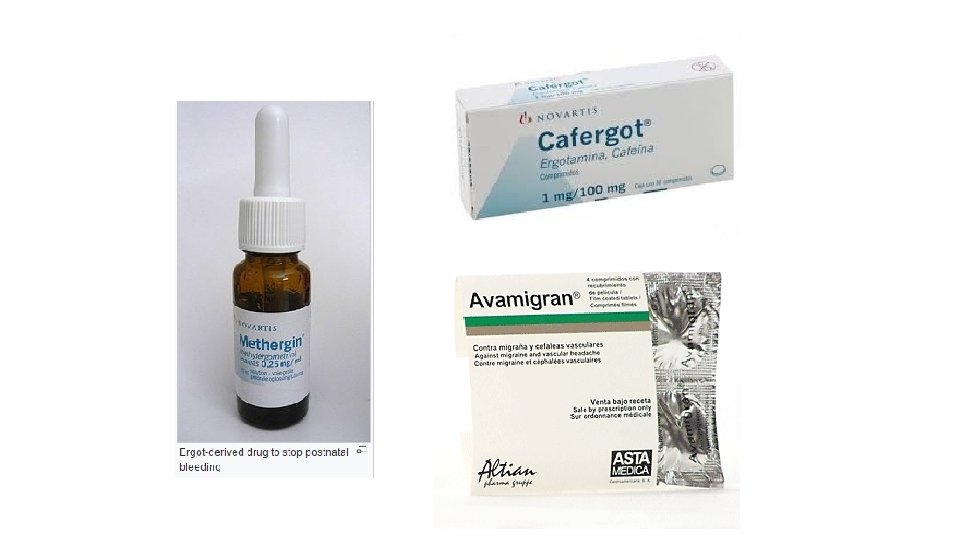

Fungal toxins 4). Ergot Alkaloids What are Ergot Alkaloids? What is Ergotism? • Ergotism is a severe reaction to ergot-contaminated food • A condition that develops from ergotamines, substances produced due to fungal contamination of grains. ** It is the effect of long-term (chronic) ergot poisoning, traditionally due to the ingestion of the alkaloids produced by the Claviceps purpurea fungus “ergotamines” that infects rye and other cereals, and more recently by the action of a number of ergotmines-based drugs.

Historical Significance Ergotism Because of the burning pain (dry gangrene), it was known as (holy fire) and (hell's fire), and was one of the causes of St. Anthony's fire. http: //www. apsnet. org/edcenter/intropp/topics/Mycotoxins/Pages/Ergot. Alkaloids. aspx

Historical Significance/ Ergotism http: //botit. botany. wisc. edu/toms_fungi/oct 99. html Anthony’s popularity as a saint reached its height in the Middle Ages. The Order of Hospitallers of St. Anthony was founded near Grenoble, France (c. 1100), and this institution became a pilgrimage centre for persons suffering from the disease known as St. Anthony’s fire (or ergotism).

Historical Significance/ Ergotism http: //homepages. rpi. edu/~caporl/home/Notes_files/Satan%20 Loosed. pdf

Fungal toxins 4). Ergot Alkaloids What are Ergot Alkaloids? What is Ergotism? • Before this disease was understood, the ergots were ground up along with rye grains and ingested when the flour was used for baking. • The symptoms can be roughly divided into: i. Convulsive symptoms: include seizures and effects on the central nervous system that range from hallucinations to psychotic episodes. ii. Gangrenous symptoms: are a result of vasoconstriction induced by the alkaloids. Peripheral systems, such as fingers and toes, are typically affected.

4). Ergot Alkaloids Fungal toxins What are Ergot Alkaloids? What is Ergotism? • Ergotism (ergot poisoning) is currently very rare, but was more common in centuries past, as the ergot fungus grows well on grain, particularly rye, which is kept in moist conditions. • The fungus is not destroyed by milling or cooking (although it is destroyed by refining), so cooked whole grain products can also be contaminated. • Persons who ingest ergot suffer similar hallucinogenic effects of LSD “lysergic acid diethylamide”, plus because the dose is typically higher than what is used for recreational purposes, it also results in heightened physical symptoms which often vary greatly depending on the patient. • Such symptoms can result in the death of the patient if the dose is large and the symptoms are severe. • Antidepressants can suppress the effect of the LSD, but the symptoms will disappear in time if the patient is not exposed further.

Fungal toxins 4). Ergot Alkaloids What are Ergot Alkaloids? What is Ergotism? What is the possible side effect of using medicine Ergot-derivative? • There is a high risk of poisoning, and it can be fatal. • Ergot poisoning can progress to gangrene, vision problems, confusion, spasms, convulsions, unconsciousness, and death. • Some people have extra reasons not to use it: ü Pregnancy and breast-feeding: It’s UNSAFE to use ergot. Ergot has many effects that can be harmful during pregnancy and breast-feeding. ü Heart disease: Ergot can narrow blood vessels and make heart disease worse. ü Kidney disease: People with kidney problems are not able to flush ergot out of their bodies well enough. This can cause ergo to build up, and that increases the risk of ergot poisoning. ü Liver disease: People with liver problems are not able to remove ergot from their bodies well enough. This can cause ergo to build up, and that increases the risk of ergot poisoning.

5 - Patulin • Patulin is a mycotoxin produced by a variety particular, Aspergillus and Penicillium and Byssochlamys. of molds, in • Most commonly found in rotting apples, in general the amount of patulin in apple products is viewed as a measure of the quality of the apples used in production. In addition, patulin has been found in other foods such as grains, fruits, and vegetables. • While not considered a particularly potent toxin, a number of studies have shown patulin to be genotoxic (still need more researches/proofs), which has led some to theorize that it may be a carcinogen, although animal studies have remained inconclusive. • Patulin has shown antimicrobial properties against some microorganisms. Several countries have instituted patulin restrictions in apple products.

5 - Patulin • Patulin was originally used as an antibiotic against Gram-positive and Gram-negative bacteria, but after several toxicity reports, it is no longer used for that purpose. • It was specifically trialed to be used against the common cold. • It is also used as a potassium-uptake inhibitor in laboratory applications. • It has been reported that patulin stimulates suicidal erythrocyte death under physiological concentrations.

5 - Patulin • Patulin is toxic primarily through affinity to sulfhydryl groups (SH), which results in inhibition of enzymes. Major acute toxicity findings include gastrointestinal problems, neurotoxicity (i. e. , convulsions), pulmonary congestion, and edema. • There are some reproduction studies in rat proved that Patulin decreased sperm count and altered sperm morphology. Also, it resulted in abortion of rats and mice after i. p. injection. • Patulin was found to be immunotoxic in a number of animal and even human studies via • • reduced cytokine secretion oxidative burst in macrophages increased splenic T lymphocytes increased neutrophil numbers • Human health • Although there are only very few reported cases and epidemiological data, the FDA has set an action limit of 50 ppb in cider due to its potential toxic effects. In humans, it was tested as an antibiotic intranasally for use against the common cold with few significant adverse effects, yet also had negligible or no beneficial effect.

Fungal toxins Toxins from fungi are widely grouped into: Ø toxins from moulds ("mycotoxins") Øtoxins from higher fungi ("mushroom poisons").


2 - Mushroom Poisoning • There actually many different types of mushrooms, though. Some have stems and caps and some don't. • Some mushrooms are called toadstools. >> This nickname probably came from the fact that they looked just like a perfect spot for a toad to sit! • Some people believe that the term “toadstool" means a mushroom that is poisonous. >> This belief may have come from the fact that many toads were considered highly poisonous. >> However, there actually many misconceptions out there about toadstools. “Not all toadstools are poisonous and not all mushrooms are safe to eat. ”

2 - Mushroom Poisoning • Mushroom poisoning (also known as mycetism/Mycetismus ) refers to harmful effects from ingestion of toxic substances present in a mushroom. Mycetismus = Ingestion of toxic fungus (poisonous mushrooms) Mycotoxicosis = Ingestion of a TOXIN produced in food by a fungus (contaminated grains) • There are thousands of species of mushrooms, but only about 100 species of mushrooms cause symptoms when eaten by humans, and only 15 -20 mushroom species are potentially lethal when ingested. • The toxicity of different mushrooms within the same genus may vary greatly. >> For instance, genus Amanita contains seven sections: https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 4057534/

2 - Mushroom Poisoning • Poisonous mushrooms contain a variety of different toxins that can differ markedly in toxicity. • Symptoms of mushroom poisoning may vary from slight gastrointestinal discomfort to lifethreatening organ failure resulting in death. • Serious symptoms do not always occur immediately after eating, often not until the toxin attacks the kidney or liver, sometimes days or weeks later. https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 4057534/
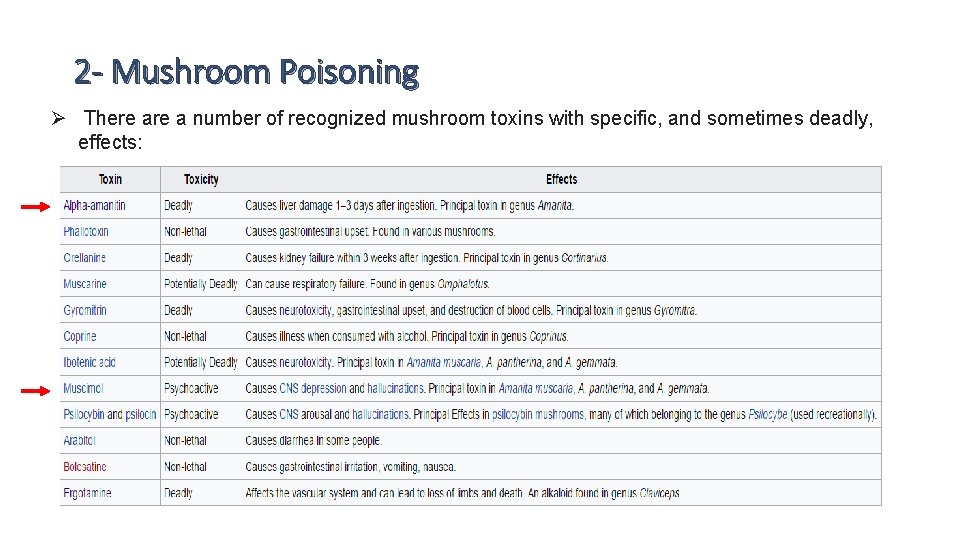
2 - Mushroom Poisoning Ø There a number of recognized mushroom toxins with specific, and sometimes deadly, effects:

2 - Mushroom Poisoning I. α-(Alpha)amanitin • α-Amanitin is a bicyclic octapeptide which belongs to a large group of protoplasmic mushroom toxins known as amatoxins. 90% of deaths from mushroom ingestions are due to AMATOXIN, which possesses a fatality rate of up to 25% when ingested. • α-Amanitin is produced by different mushroom species: ü the green death cap mushroom* (Amanita phalloides) ü Fool’s Mushroom*Amanita verna, ü The Destroying Angel* (Amanita virosa) • These mushroom species, and other not listed, produce α-amanitin in amounts sufficient to poison an adult person with liver damage and fatal outcome (LD 50, p. o. humans; 0. 1 mg/kg). http: //adcreview. com/adc-university/adcs-101/cytotoxic-agents/%CE%B 1 -amanitin/

2 - Mushroom Poisoning I. α-(Alpha)amanitin q Mode of Action: • Alpha amanitin is known as potent inhibitor RNA polymerase >> The toxin shows remarkable binding affinity for eukaryotic RNA polymerase II, slightly binds to RNA polymerase III, and shows no activity on RNA polymerase I {RNA polymerase: is an enzyme that produces primary transcript RNA polymerase II: catalyzes the transcription of DNA to synthesize precursors of m. RNA} v α-Amanitin binds on the back side of RNA polymerase, away from the active site and away from the binding site for the DNA and RNA. http: //adcreview. com/adc-university/adcs-101/cytotoxic-agents/%CE%B 1 -amanitin/

2 - Mushroom Poisoning I. α-(Alpha)amanitin q Mode of Action: v This binding does not physically block the active site, like most inhibitors, but instead jams the mechanism of the enzyme: >> RNA polymerase is a highly mobile enzyme, that flexes and changes shape as it performs the sequential steps of binding to DNA, unwinding it, and then building the RNA strand >> the poison binds between two subunits of the protein, gluing them together and blocking these essential motions. >> this will reduce the rate of polymerase translocating on DNA from several thousand to a few nucleotides per minute https: //www. youtube. com/watch? v=J 3 HVVi 2 k 2 No http: //adcreview. com/adc-university/adcs-101/cytotoxic-agents/%CE%B 1 -amanitin/

2 - Mushroom Poisoning I. α-(Alpha)amanitin q Mode of Action: v the poison binds between two subunits of the protein, gluing them together and blocking these essential motions. v this will reduce the rate of polymerase translocating on DNA from several thousand to a few nucleotides per minute! v Since the rate of transcription is significantly slowed to this extent, the cell cannot produce m. RNA transcripts at an efficient rate, ultimately destroying the cell. http: //adcreview. com/adc-university/adcs-101/cytotoxic-agents/%CE%B 1 -amanitin/

2 - Mushroom Poisoning I. α-(Alpha)amanitin q Symptoms: v Diarrhea and cramps are the first symptoms, but those pass, giving a false sign of remission. v Typically, on the 4 th to 5 th day, the toxin starts to have severe effects on the liver and kidneys, leading to total system failure in both. v Death usually takes place around a week from ingestion. http: //adcreview. com/adc-university/adcs-101/cytotoxic-agents/%CE%B 1 -amanitin/

2 - Mushroom Poisoning I. α-(Alpha)amanitin q Use in medicine: v The trial drug has shown the outstanding activity of amanitin-based ADCs in therapy-resistant tumor cells. *(Antibody-drug conjugates or ADCs are an important class of highly potent biopharmaceutical drugs designed as a targeted therapy for the treatment of people with cancer. Unlike chemotherapy, ADCs are intended to target and kill only the cancer cells and spare healthy cells. ADCs are complex molecules composed of an antibody linked to a biologically active cytotoxic (anticancer) payload or drug). v α-Amanitin seems to be a suitable toxic payload for use in an Antibody-drug Conjugate or ADC because of the unique mode of action and the molecular characteristics of the toxin http: //adcreview. com/adc-university/adcs-101/cytotoxic-agents/%CE%B 1 -amanitin/

2 - Mushroom Poisoning Examples of erroneous folklore "rules" include: Ø "Poisonous mushrooms are brightly colored. " Some choice edible species are brightly colored, while most poisonous species are brown or white. Ø "Insects/animals will avoid toxic mushrooms. " Fungi that are harmless to invertebrates can still be toxic to humans; the death cap, for instance, is often infested by insect larvae. Ø "Poisonous mushrooms blacken silver. " – None of the known mushroom toxins have a reaction with silver. Ø "Poisonous mushrooms taste bad. " – People having eaten the deadly Amanitas reported that the mushrooms tasted quite good. Ø "All mushrooms are safe if cooked/parboiled/dried/pickled/etc. " – While it is true that some otherwise-inedible species can be rendered safe by special preparation, many toxic species cannot be made toxin-free. Ø "Poisonous mushrooms will turn rice red when boiled. " – this misconception cost at least one person her life. Ø "Poisonous mushrooms have a pointed cap. Edible ones have a flat, rounded cap. " – The shape of the mushroom cap does not correlate with presence or absence of mushroom toxins

- Slides: 58