Microbial Toxins BIOC 422 Bacterial Toxins Exotoxins Dr

Microbial Toxins (BIOC 422) Bacterial Toxins (Exotoxins) Dr. Ayat B. Al-Ghafari Associate Professor of Biomedical Sciences e. mail: abalghafari@kau. edu. sa

4 - Production of toxins (Toxigenicity) I. Exotoxins ØMain Characteristics: • They are poisonous proteins produced inside pathogenic bacteria as part of their growth and metabolism and released into host tissue. >> Produced mainly by gram-positive bacteria. >> Proteins: Enzymes that carry out specific reactions. • Soluble in body fluids, rapidly transported throughout body in blood or lymph. • Work by destroying particular parts of the host’s cells or by inhibiting certain metabolic functions.

4 - Production of toxins (Toxigenicity) I. Exotoxins ØExotoxin classification: There are different systems for classifying or identifying exotoxins including: • By organism generating the toxin • By organism susceptible to the toxin • By tissue target type susceptible to the toxin (neurotoxins affect the nervous system, cardiotoxins affect the heart, etc. ) • By structure (for example, AB 5 toxin) • By a letter, such as "A", "B", or "C", to communicate the order in which they were identified ØTherefore: the same exotoxin may have different names, depending of the field of research.

4 - Production of toxins (Toxigenicity) I. Exotoxins Ø Exotoxin Types (General classifications): Types of Exotoxins 1. Cytotoxins: exotoxins that kill cells. Cytotoxins e. g. shigella, vibrio 2. Neurotoxins: exotoxins that affect nerve transmission of the nerve Neurotoxins system and interfere with nerve impulses. e. g. Clostridium botulinum, Clostridium tetani 3. Enterotoxins: Effect cells lining of the G. I. Tract Enterotoxins e. g. E. coli , cholera, Shigella, Salmonella

• Usually, virulent strains of the bacterium produce the toxin while nonvirulent strains do not. • The toxin is the major determinant of virulence. • Because exotoxins are Protein Toxins they have protein properties including; • Denatured by heat, acid, and proteolytic enzymes • High biological activity • Most act catalytically • Highly specific in the substrate utilized. v According to the mode of Action, some exotoxins can; • Damage membranes by either act as lipases and digest the phospholipids or by form membrane pores. • Inhibit protein synthesis (N-glycosidase). • Activate 2 nd messenger pathways (ADP-ribosyltransferase). • Block nerve function By interfere in neurotransmission and thereby result in spastic or paralysis • Trigger specific host cell necrosis. • Activate immune response (super-antigen). • Protease (Zinc-metalloprotease called lethal toxin).

4 - Production of toxins (Toxigenicity) I. Exotoxins Ø Exotoxin Structure: Many exotoxins are composed of an A and a B subunit and are called A-B toxins Ø The A subunit is the active or enzymatic component Ø The B subunit is the binding component

I. Exotoxins Ø Mode of Action of Exotoxin This figure shows that exotoxins are secreted by Clostridium botulinum and are toxic to somatic cells. Somatic cells have antibodies on the cell surface to target exotoxins antibodies and bind to them, preventing the invasion of somatic cells. The binding of the exotoxin and antibody forms an Ag-AB interaction and the exotoxins are targeted for destruction by the immune system. If this interaction does not happen, the exotoxins happen bind to the exotoxin receptors that are on the cell surface and causes death of the host cell by inhibiting protein synthesis. This figure also shows that the application of heat or chemicals to exotoxins can result in the deactivation of exotoxins. The deactivated exotoxins are called toxoids and they are not harmful to somatic cells.

4 - Production of toxins : I. Exotoxins 1)- Cytotoxins : Ø A cytotoxin is any substance which has a toxic effect on cells. ØCytotoxins typically attack only a specific type of cell or organ, rather than an entire body. ØDifferent kinds of cytotoxins work in different ways: ØSome cytotoxins make holes in the cell membrane, so the inside of the cell is cytotoxins make holes in the cell membrane not protected from the outside. ØOther cytotoxins turn on a program in the cell that causes it to self-destruct. This is called apoptosis
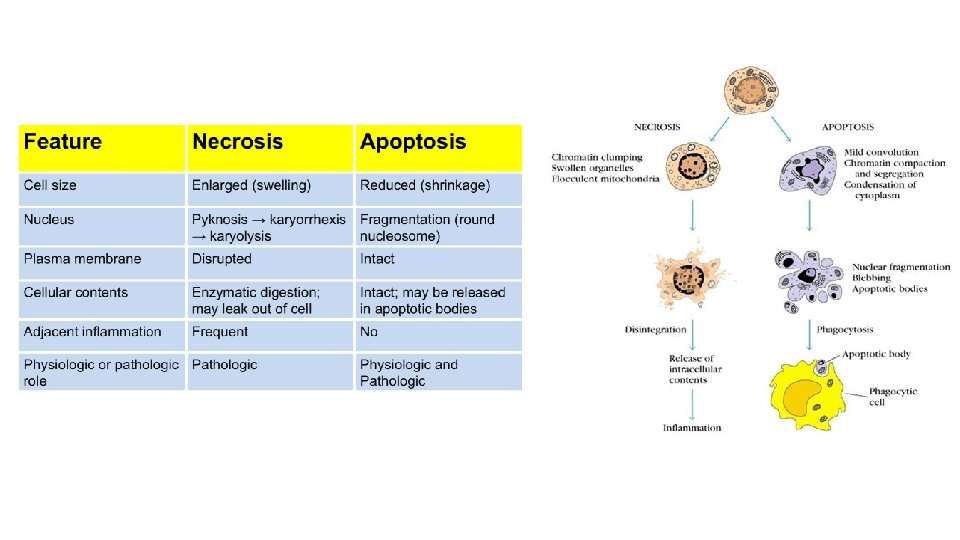

4 - Production of toxins : I. Exotoxins 1)- Cytotoxins : Ø A cytotoxin is any substance which has a toxic effect on cells. Ø Cytotoxins typically attack only a specific type of cell or organ, rather than an entire body. EXAMPLES: i. Diphtheria Toxin >> Diphtheria • The best known and studied bacterial toxin. • It is an exotoxin secreted by Corynebacterium diphtheria, causing Diphtheria, which produce diphtheria toxin (exotoxin), a binary toxin consisting of two molecular components termed fragments A and B. (Aminoff's Neurology and General Medicine (Fifth Edition), 2014).

i. Diphtheria Toxin What is Corynebacterium diphtheria? Biological Features: C. diphtheriae is an aerobic gram-positive bacillus. • Rod shaped • Non-capsulated • Non-spore-forming • Non-motile (show no motion) • Produces AB exotoxin

i. Diphtheria Toxin What is Diphtheria? • Diphtheria is a highly-contagious disease caused by Corynebacterium diphtheriae. • It is generally an upper respiratory tract illness characterized by sore throat, low-grade fever, and an adherent membrane (a pseudomembrane) on the tonsil(s), pharynx, and/or nose. • A milder form of diphtheria can be limited to the skin. • Diphtheria was named in 1826 and takes its name from the Greek word for “leather, ” >>The name alludes to the leathery, sheath-like membrane that grows on the tonsils, throat, and in the nose.

Diphtheria
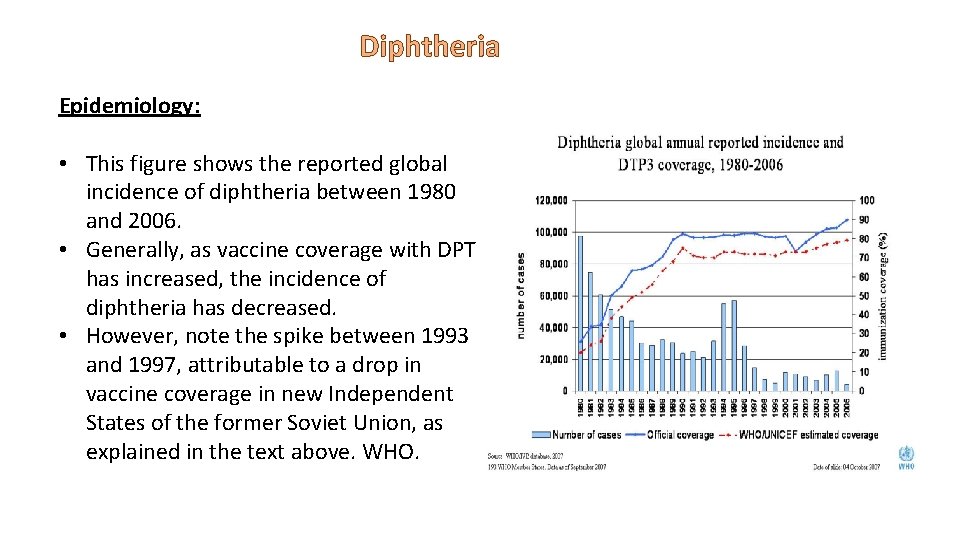
Diphtheria Epidemiology: • This figure shows the reported global incidence of diphtheria between 1980 and 2006. • Generally, as vaccine coverage with DPT has increased, the incidence of diphtheria has decreased. • However, note the spike between 1993 and 1997, attributable to a drop in vaccine coverage in new Independent States of the former Soviet Union, as explained in the text above. WHO.

Pathogenesis: Diphtheria • The transfer of the gene for the formation of diphtheria toxin carried by bacteriophage (a virus that attacks and infects bacteria). • The toxin causes the disease diphtheria in humans by gaining entry into the cell cytoplasm and inhibiting protein synthesis. • It is produced as single polypeptide chain of 535 amino acids consisting of two subunits linked by disulfide bridges, known as an A-B toxin. • “A” (Red) Active part: is the catalytic domain; • “B” (yellow) is the binding domain which displays the receptor for cell attachment; • “T” (blue) is the hydrophobic domain responsible for insertion into the endosome membrane to secure the release of A.
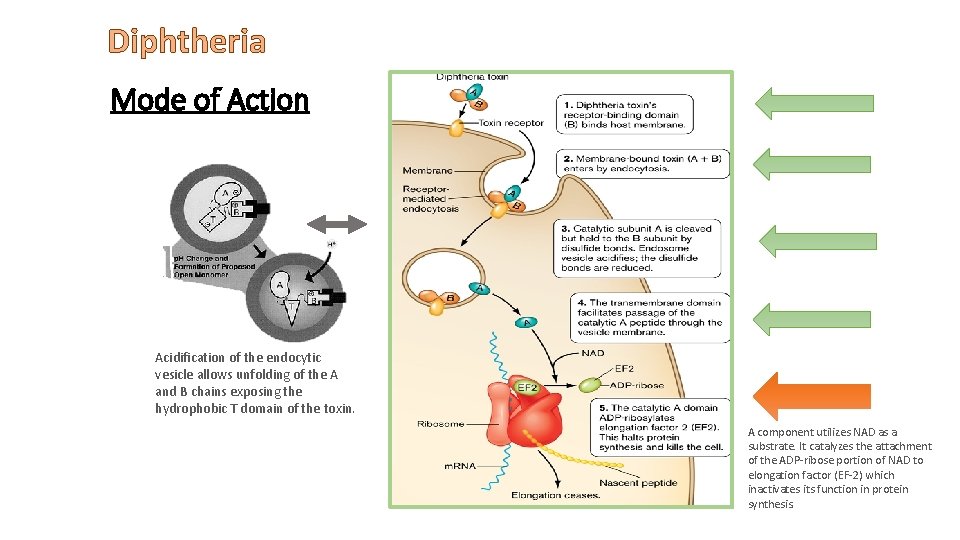
Diphtheria Mode of Action Acidification of the endocytic vesicle allows unfolding of the A and B chains exposing the hydrophobic T domain of the toxin. A component utilizes NAD as a substrate. It catalyzes the attachment of the ADP-ribose portion of NAD to elongation factor (EF-2) which inactivates its function in protein synthesis.

Signs and symptoms Diphtheria • The lethal dose for humans is about 0. 1 μg of toxin per kg of bodyweight. • A massive release of toxin into the body will likely cause lethal necrosis of the heart and liver. • The respiratory form has an incubation period of two to five days and the onset of disease is usually gradual. • Symptoms include : > Local infection symptoms • Severe inflammatory reaction • Severe swelling in back of neck • Sore throat, nausea, vomiting • Formation of pseudomembrane > Systemic symptoms • Toxemia as toxin is absorbed from throat and carried by blood to target organs • Heart and nervous system

Diphtheria Transmission: solely among humans spread by droplets, secretions & direct contact Risk Factors: 1 - Poor nutrition 2 - Crowded or unhealthy living conditions 3 - Low vaccine coverage among infants and children 4 - Immunity gaps in adults Diagnosis: The laboratory criteria for diagnosis The clinical criteria for diagnosis § Isolation of Corynebacterium diphtheriae from a clinical specimen; § Histopathologic diagnosis of diphtheria. § Upper respiratory tract illness with sore throat; § Low-grade fever; § An adherent membrane of the tonsil(s), pharynx, and/or nose. .

Diphtheria Treatment: The disease may remain manageable, but in more severe cases: Ø lymph nodes in the neck may swell, causing breathing and swallowing to become more difficult. >>People in this stage may require intubation (placing a tube in the trachea) to open a direct airway through an cut in the trachea. Ø In addition, an increase in heart rate may cause cardiac arrest Ø can cause paralysis in the eye, neck, throat, or respiratory muscles. >> Individuals with severe cases are put in a hospital intensive care unit (ICU) and given a diphtheria antitoxin. >> Since antitoxin does not neutralize toxin that is already bound to tissues, delaying its administration is associated with an increase in mortality risk… . . Therefore, the decision to administer diphtheria antitoxin is based on clinical diagnosis, and does not have to await laboratory confirmation. Vaccine: Prevention is done by immunization with toxoid vaccine,

4 - Production of toxins : Exotoxins 2)- Neurotoxins: Ø are toxins that are poisonous or destructive to nerve tissue, causing neurotoxicity. Ø This can eventually disrupt or even kill neurons, key cells that transmit and process signals in the brain and other parts of the nervous system. ØCommon examples of neurotoxins include botulinum toxin, tetrodotoxin and tetanus toxin. ØGenerally, neurotoxins inhibit neuron control over ion concentrations across the cell membrane, or communication between neurons across a synapse. cell membrane communication between neurons across a synapse EXAMPLES: i. Tetanospasmin >> Tetanus • Tetanospasmin is an extremely potent neurotoxin produced by Clostridium tetani, causing tetanus

i. Tetanospasmin Toxin What is Clostridium tetani ? Biological Features: # Gram positive bacilli # Rod shaped # Non capsulated # heat-sensitive # exhibits flagellar motility # strict anaerobe: cells killed from O 2 exposure >> However, as the bacterium matures, it develops a terminal spore, ( Spores survive in long periods of O 2) giving it a drumstick appearance >> C. tetani spores are extremely hardy as they are resistant to heat and most antiseptics # Habitat: Live in all anaerobic habitats including soil, feces & intestinal tracts of various animals.

i. Tetanospasmin Toxin What is tetanus? • From a Greek word: “to stretch” • First described by Hippocrates (Greek-Father of Modern Medicine, 460 BC) & Susruta (an ancient Indian physician, has been called "the first plastic surgeon, 600 B. C. ). • Tetanus is an acute, often fatal, disease caused by Tetanospasmin exotoxin that is produced by the bacterium Clostridium tetani. • Tetanus is often called “lockjaw or trismus” because the jaw muscles tighten, and the person cannot open their mouth • It affects the nervous system, leading to painful muscle contractions, particularly of your jaw and neck muscles. • Tetanus is very dangerous. It can cause difficulty breathing and paralysis. Even with intensive care, 10%– 20% of people with tetanus die

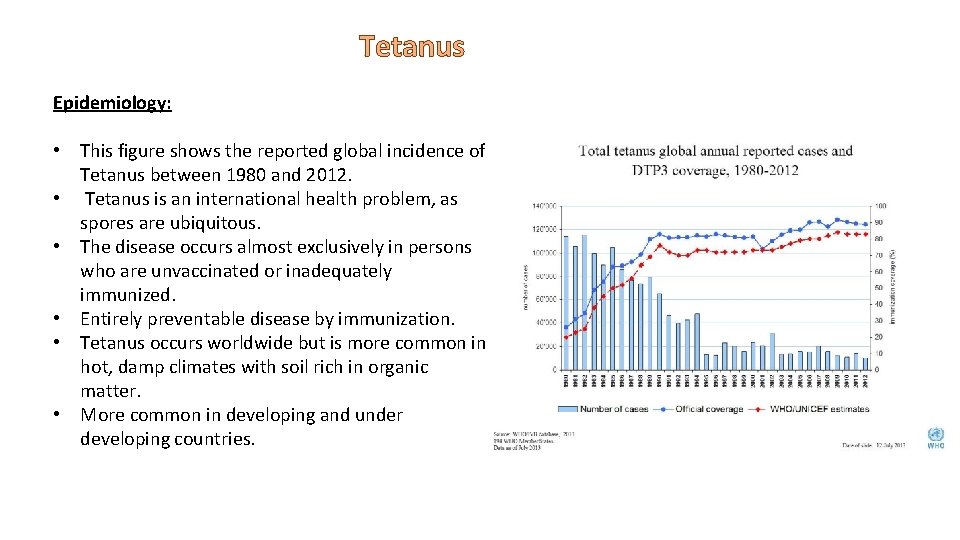
Tetanus Epidemiology: • This figure shows the reported global incidence of Tetanus between 1980 and 2012. • Tetanus is an international health problem, as spores are ubiquitous. • The disease occurs almost exclusively in persons who are unvaccinated or inadequately immunized. • Entirely preventable disease by immunization. • Tetanus occurs worldwide but is more common in hot, damp climates with soil rich in organic matter. • More common in developing and under developing countries.
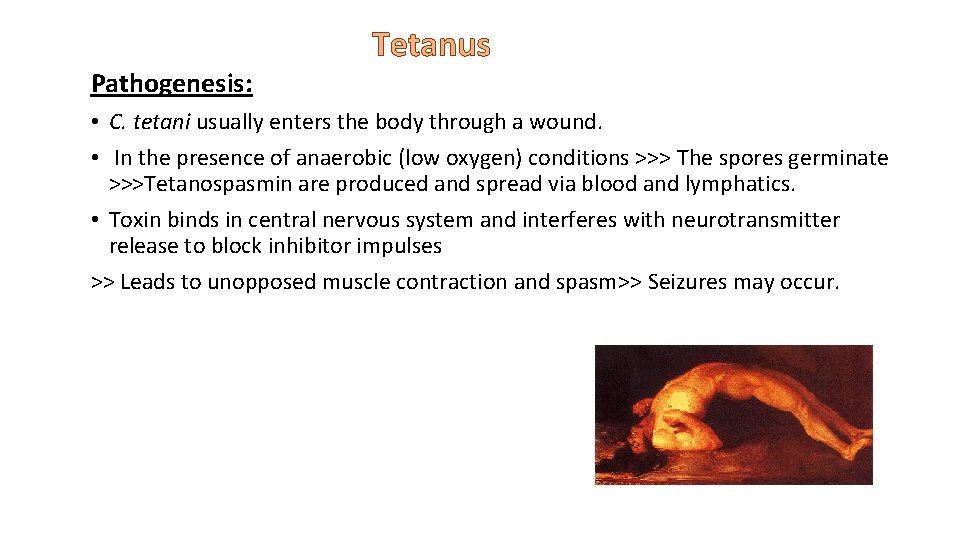
Tetanus Pathogenesis: • C. tetani usually enters the body through a wound. • In the presence of anaerobic (low oxygen) conditions >>> The spores germinate >>>Tetanospasmin are produced and spread via blood and lymphatics. • Toxin binds in central nervous system and interferes with neurotransmitter release to block inhibitor impulses >> Leads to unopposed muscle contraction and spasm>> Seizures may occur.
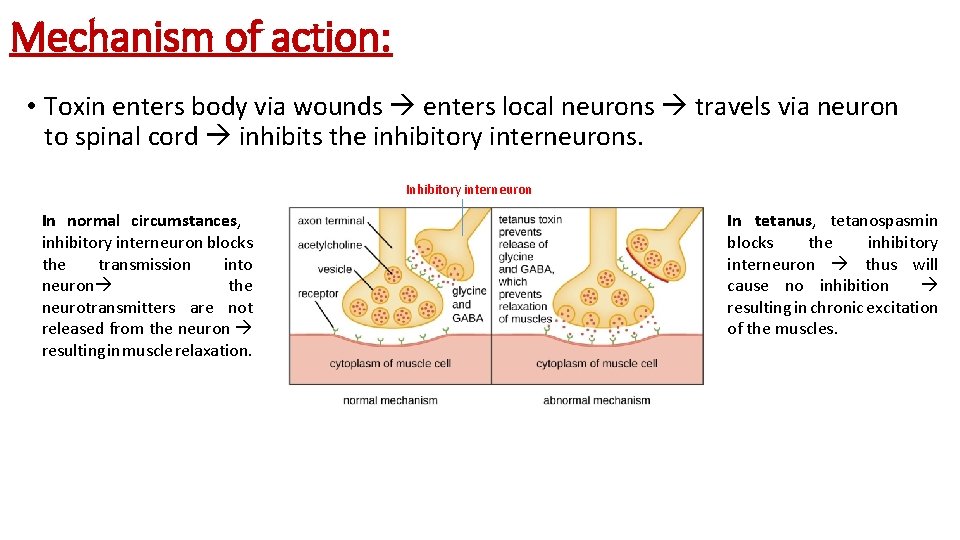
Mechanism of action: • Toxin enters body via wounds enters local neurons travels via neuron to spinal cord inhibits the inhibitory interneurons. Inhibitory interneuron In normal circumstances, inhibitory interneuron blocks the transmission into neuron the neurotransmitters are not released from the neuron resulting in muscle relaxation. In tetanus, tetanospasmin blocks the inhibitory interneuron thus will cause no inhibition resulting in chronic excitation of the muscles.
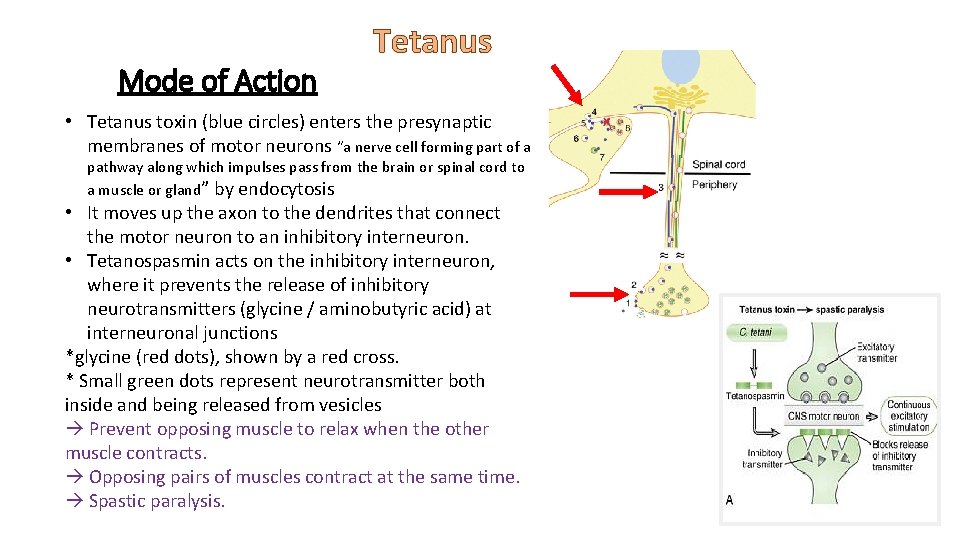
Mode of Action Tetanus • Tetanus toxin (blue circles) enters the presynaptic membranes of motor neurons “a nerve cell forming part of a pathway along which impulses pass from the brain or spinal cord to a muscle or gland” by endocytosis • It moves up the axon to the dendrites that connect the motor neuron to an inhibitory interneuron. • Tetanospasmin acts on the inhibitory interneuron, where it prevents the release of inhibitory neurotransmitters (glycine / aminobutyric acid) at interneuronal junctions *glycine (red dots), shown by a red cross. * Small green dots represent neurotransmitter both inside and being released from vesicles Prevent opposing muscle to relax when the other muscle contracts. Opposing pairs of muscles contract at the same time. Spastic paralysis.

Tetanus Signs and symptoms • Tetanus symptoms usually emerge about 7 -10 days after initial infection, however, this can vary from 4 days to about 3 weeks, and, in some cases, it may take months. • In general, the further the injury site is from the central nervous system, the longer the incubation period. “Patients with shorter incubation times tend to have more severe symptoms”. Ø Common signs include: # Spasms and stiffness in jaw muscles (trismus), (a rigid smile) # Stiffness of neck muscles # Difficulty swallowing # Stiffness of abdominal muscles Ø Possible other signs include: Fever Sweating Rapid heart rate(Tachycardia) Diarrhea: Bloody stools (feces)

Tetanus Transmission: • Tetanus is not transmitted from person to person. • A person usually becomes infected with tetanus when dirt enters a wound or cut. Tetanus germs are likely to grow in deep puncture wounds caused by dirty nails, knives, tools, wood splinters, and animal bites. .

Tetanus Risk Factors: • Anyone who experiences a wound that is exposed to contaminated soil or manure can get tetanus • The only people who are at little to no risk of catching tetanus are people who have been vaccinated against it. • Tetanus is a standard childhood vaccine, and pregnant women can also pass their immunity onto their children in the womb. • Adults need booster shots approximately every five to seven years. People who are not sure of their vaccination status should get a booster immediately after becoming injured. Diagnosis: Doctors diagnose tetanus based on a physical exam, medical and immunization history, and the signs and symptoms of muscle spasms, stiffness and pain. Laboratory tests generally aren't helpful for diagnosing tetanus.

Treatment: Tetanus If tetanus does develop, seek hospital treatment immediately. Treatment includes: • wound care, • a course of antibiotics, • and an injection of tetanus antitoxin. • Patient may receive medications such as chlorpromazine or diazepam to control muscle spasms Vaccine: § Prevention is done by immunization with toxoid vaccine. § The baby is injected with the DTa. P vaccine which is 3 inactive toxins in one injection. § DTa. P protects against diphtheria, pertussis, and tetanus. >> This vaccine is safer than the previously used DTP. Another option for infants is DT which is a vaccine that is a combination of diphtheria and tetanus vaccines.

Exotoxins (Enterotoxins) • An enterotoxin is a protein exotoxin released by a microorganism that targets the intestines • The enterotoxin is a common cause of foodborne bacterial poisoning in the United States, the United Kingdom, and probably across the globe. • They are often heat-stable, and are of low molecular weight and water-soluble. heat-stable low molecular weight water-soluble • Enterotoxins are frequently cytotoxic and kill cells by altering the apical membrane permeability of the mucosal epithelial cells of the intestinal wall. • They are mostly pore- forming toxins (mostly chloride pores), secreted by bacteria, that assemble to form pores in cell membrane. This causes the cells to die. • The action of enterotoxins leads to increased chloride ion permeability of the apical membrane of intestinal mucosal cells >> These membrane pores are activated either by increased c. AMP or by increased calcium ion concentration intracellularly >> Increased chloride permeability leads to leakage into the lumen followed by sodium and water movement >> This leads to a secretory diarrhoea within a few hours of ingesting enterotoxin. Several microbial organisms contain the necessary enterotoxin to create such an effect, such as Staphylococcus aureus and E. coli.

3)- Enterotoxin EXAMPLES: i. Disease: Cholera ii. Toxin: Cholera toxin iii. Bacteria: vibrio cholerae
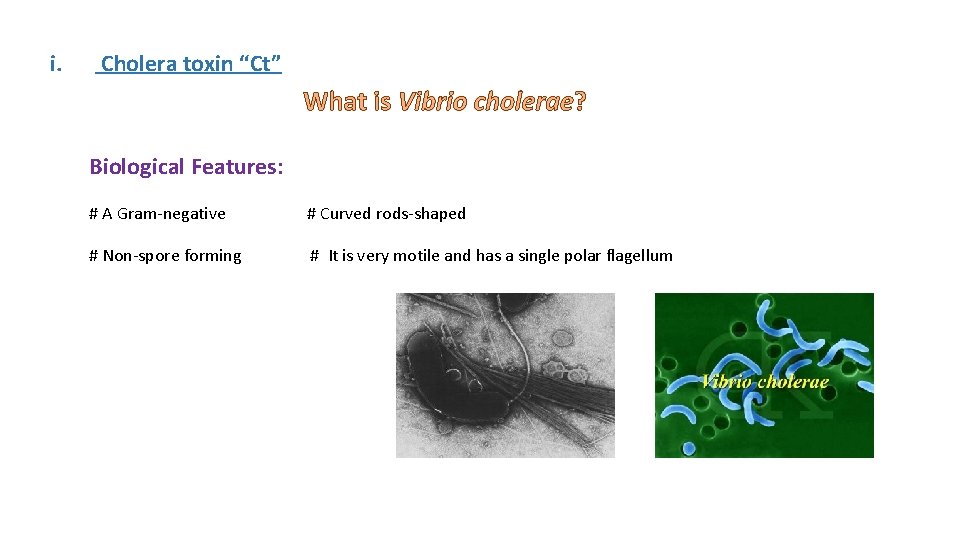
i. Cholera toxin “Ct” What is Vibrio cholerae? Biological Features: # A Gram-negative # Curved rods-shaped # Non-spore forming # It is very motile and has a single polar flagellum

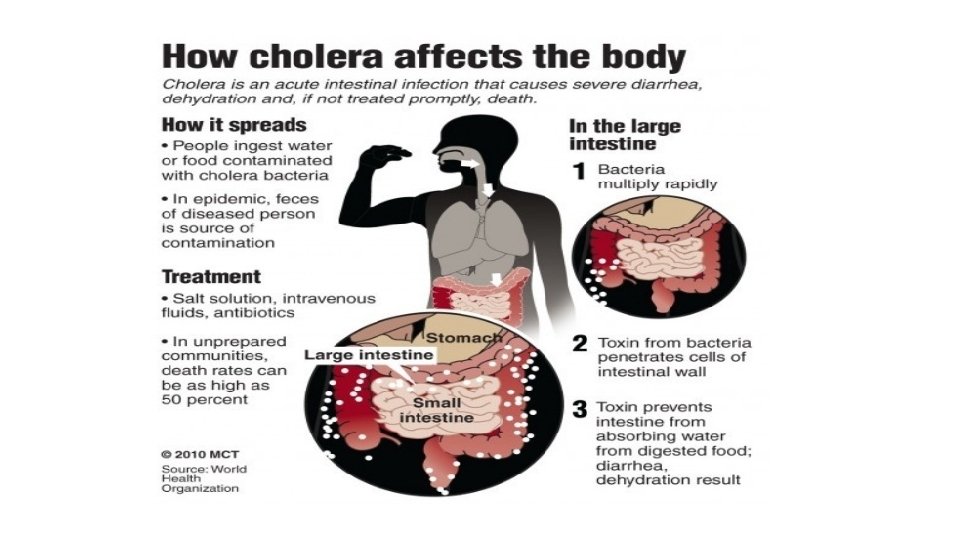
i. Cholera toxin “Ct” What is cholera? • Cholera is an extremely virulent disease/infection of the small intestine by some strains of the bacterium Vibrio cholerae. • Symptoms may range from none, to mild, to severe. • It is one of the most devastating human diseases, with a rapid onset of diarrhoea and vomiting within several hours to 2 -3 days of ingestion and leg cramps. • Severe diarrhea and vomiting lead to massive fluid losses (~ 15 -20 liters/day), leads to dehydration, electrolyte imbalance and shock. • Cholera affects both children and adults and can kill within hours if untreated. • The untreated mortality rate is in excess of 60%. However, rehydration therapy brings the mortality rate to below 1%. • According to WHO, a global strategy on cholera control with a target to reduce cholera deaths by 90% was launched in 2017

i. Cholera toxin “Ct” What is cholera? • The main cause of cholera is infected/contaminated water >> then Provision of safe water and sanitation is critical to control the transmission of cholera and other waterborne diseases. • Most of those infected will have no or mild symptoms >> can be successfully treated with oral rehydration solution. • Severe cases will need rapid treatment with intravenous fluids and antibiotics.

Cholera History: • During the 19 th century, The first 2 cholera pandemic occurred in the Bengal region of India, near Calcutta starting in 1817 through 1824. • The disease spersed from India by trade 3 routes (land sea) to Southeast Asia, the Middle East, Europe, Eastern Africa… and from Europe to North America and the rest of the world. 1 4 5 6 7 8
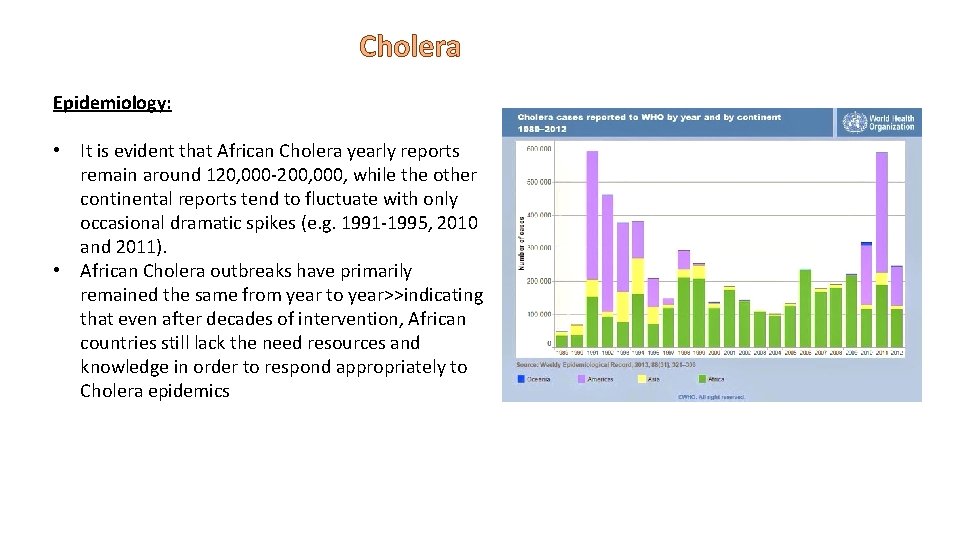
Cholera Epidemiology: • It is evident that African Cholera yearly reports remain around 120, 000 -200, 000, while the other continental reports tend to fluctuate with only occasional dramatic spikes (e. g. 1991 -1995, 2010 and 2011). • African Cholera outbreaks have primarily remained the same from year to year>>indicating that even after decades of intervention, African countries still lack the need resources and knowledge in order to respond appropriately to Cholera epidemics
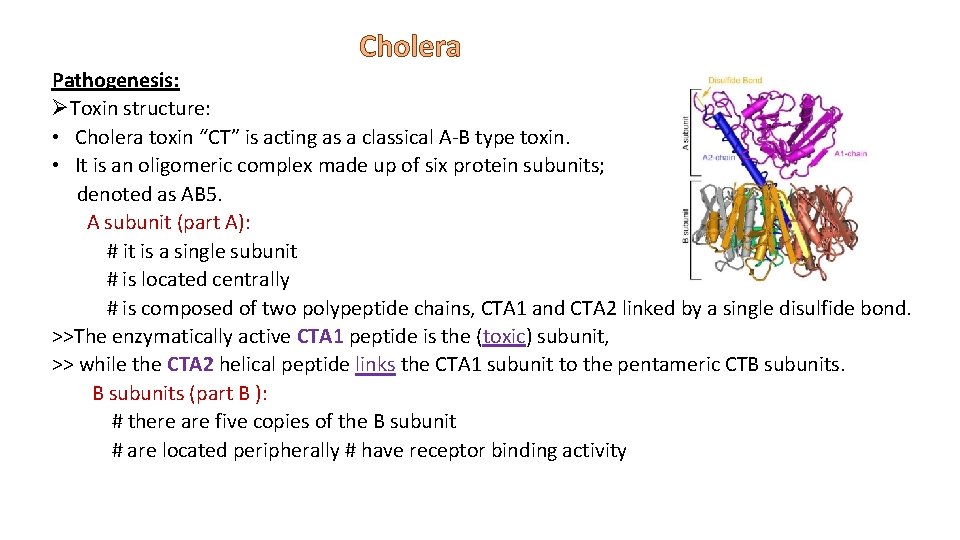
Cholera Pathogenesis: ØToxin structure: • Cholera toxin “CT” is acting as a classical A-B type toxin. • It is an oligomeric complex made up of six protein subunits; denoted as AB 5. A subunit (part A): # it is a single subunit # is located centrally # is composed of two polypeptide chains, CTA 1 and CTA 2 linked by a single disulfide bond. >>The enzymatically active CTA 1 peptide is the (toxic) subunit, >> while the CTA 2 helical peptide links the CTA 1 subunit to the pentameric CTB subunits. B subunits (part B ): # there are five copies of the B subunit # are located peripherally # have receptor binding activity
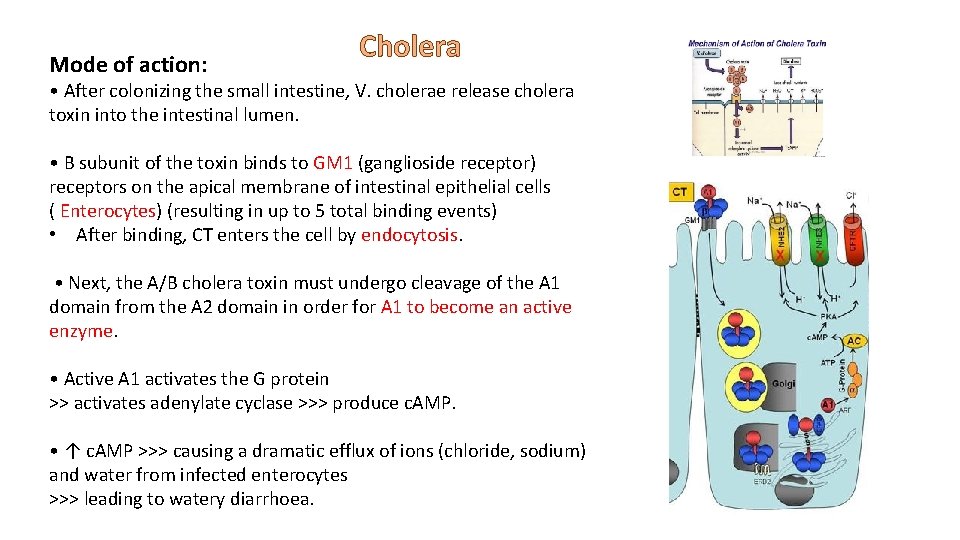
Mode of action: Cholera • After colonizing the small intestine, V. cholerae release cholera toxin into the intestinal lumen. • B subunit of the toxin binds to GM 1 (ganglioside receptor) receptors on the apical membrane of intestinal epithelial cells ( Enterocytes) (resulting in up to 5 total binding events) • After binding, CT enters the cell by endocytosis. • Next, the A/B cholera toxin must undergo cleavage of the A 1 domain from the A 2 domain in order for A 1 to become an active enzyme. • Active A 1 activates the G protein >> activates adenylate cyclase >>> produce c. AMP. • ↑ c. AMP >>> causing a dramatic efflux of ions (chloride, sodium) and water from infected enterocytes >>> leading to watery diarrhoea.

Cholera Signs and symptoms • Cholera is one of the most devastating human diseases, with a rapid onset of diarrhoea and vomiting within several hours to 2 -3 days of ingestion. • Due to massive fluid losses (15 -20 litres/day), the untreated mortality rate is in excess of 60%. However, rehydration therapy brings the mortality rate to below 1%. .

Cholera Transmission: • It is spread mostly by water and food that has been contaminated with human faeces containing the bacteria. • Insufficiently cooked seafood is a common source. • Humans are the only animal affected. Risk Factors: • People living in unsafe water and sanitation conditions are at risk as well as those with lower immunity, including from autoimmune disorders such as HIV/AIDS. Treatment: Consuming large amounts of water mixed with oral rehydration salts or, for cases of severe dehydration, administering intravenous fluids in addition to oral rehydration salts can effectively treat the majority of cholera cases. Antibiotics can also be administered along with a rehydration program.

Cholera Vaccine: Cholera has been a burden to human health for centuries and has spread globally in the past 200 years. For the past 15 years, the World Health Organization has called for the development of oral cholera vaccines. While previously, researchers focused on creating vaccines using inactivated CT, it is now understood through animal model and human studies that anti-CT immunity that developed from treatment by the vaccine is not nearly as effective at protecting the patient from cholera. Prevention: It involves improved sanitation and access to clean water.

4 - Production of toxins : Exotoxins 3)- Enterotoxin EXAMPLES: ii. E. coli toxins >> E. Coli Food poisoning

ii. E. coli toxin: Biological Features: What is E. Coli ? • E. coli was first discovered in 1885 by Theodor Escherichia, a German bacteriologist. • It is a gram-negative, rod-shaped bacterium. Motile by peritrichous flagella and fimbriae. https: //www. youtube. com/watch? v=PD 30 t. MWHj. Jo • The bacterium can be grown easily and inexpensively in a laboratory setting, and has been intensively investigated for over 60 years. • E. coli is the most widely studied prokaryotic model organism, and an important species in the fields of biotechnology and microbiology, where it has served as the host organism for the majority of work with recombinant DNA.

ii. E. coli toxin: What is E. Coli ? Biological Features: • It is commonly found in the intestines of warm-blooded animals, including humans. • It is expelled into the environment within faecal matter. • E. coli consists of a diverse group of bacteria. The vast majority of its strains are harmless, or even harmless beneficial. For instance, E. coli produces vitamins K and B 6, and maintains a protective space in your gut for other beneficial bacteria.

ii. E. coli toxin: Pathogenesis of E. coli: • Pathogenic E. coli strains are categorized into pathotypes. strains • Six pathotypes are associated with DIARRHEA and collectively are referred to as diarrheagenic E. coli. 1. Enterohemorrhagic E. Coli (EHEC) 2. Enterotoxigenic E. Coli (ETEC) 3. Enteropathogenic E. coli (EPEC) 4. Enteroaggregative E. coli (EAEC) 5. Enteroinvasive E. coli (EIEC) 6. Diffusely adherent E. coli (DAEC)

ii. E. coli toxin (Shiga toxin): Shiga toxin Pathogenesis of E. coli: ØE. coli O 157: H 7 is within the class enterohemorrhagic (EHEC): E. coli O 157: H 7 • E. coli O 157: H 7 makes Verotoxin 1 (Vt 1) and Verotoxin 2 (Vt 2) these two toxins are also referred to as: Shiga toxin 1(Stx 1) and Shiga toxin 2 (Stx 2), respectively. Shiga toxin is known as a Shiga toxin-producing E. coli (STEC). • The ability of these strains of E. coli to produce Shiga toxin was acquired from a bacteriophage carrying the Shiga toxin gene from the Gram negative (G-) pathogenic bacteria Shigella.

Pathogenesis of E. coli : Shiga toxin-producing E. coli (STEC). • The most commonly heard about in the news in association with foodborne outbreaks. • E. coli O 157: H 7 >> { (EHEC) serotype} is the worst type of E. coli: >> it causes bloody diarrhea >> it can sometimes cause kidney failure and even death. • STEC can grow in temperatures ranging from 7 °C to 50 °C, with an optimum temperature of 37 °C. Some STEC can grow in acidic foods, down to a p. H of 4. 4. • STEC is destroyed by thorough cooking of foods until all parts reach a temperature of 70 °C or higher. • An estimated 73, 000 cases of these E. coli infections occur in the U. S. every year, according to the CDC. Verotoxin= Shiga toxin E. coli O 157: H 7 = Shiga toxin-producing E. coli (STEC)> Enterohemorrhagic (EHEC)

Shiga toxin-producing E. coli (STEC) >> Food Poisoning • When two or more people get the same illness from the same contaminated food or drink, the event is called a foodborne disease outbreak. • E. coli O 157: H 7 is a deadly foodborne pathogen! • Annually in the United States, EHEC is responsible for an estimated 73, 000 illnesses, 1800– 3600 hospitalizations and from 61 to 541 deaths (Rangel et al. , 2005) (www. cdc. gov • E. coli O 157: H 7 is transmitted to humans primarily through consumption of contaminated foods

Shiga toxin-producing E. coli (STEC). Shiga toxin (from E. coli O 175: H 7 bacteria strain) structure: • It consists of one A-subunit and five B-subunits. • The B-subunit binds to endothelial cells surface Gb 3 receptor, and this specificity determines where Shiga toxin mediates its pathophysiology. • The A-subunit contains the catalytic domain, which is active when the A 1 fragment is released upon cleavages by the intracellular protease. >> Shiga toxin (A-subunit) are ribosome-inactivating proteins with RNA-N- glycosidase activity >> it remove a specific adenine from 28 S RNA >> inhibits host protein synthesis and induces apoptosis • • • Ribosomes are a cell structure that makes protein. Ribosomes consist of two major components: the small ribosomal subunit, which reads the RNA, and the large subunit, which joins amino acids to form a polypeptide chain. In Eukaryotic: 28 S r. RNA is part of the large subunit

ii. E. coli toxin: Shiga toxin-producing E. coli (STEC). Mode of Action: 1) EHEC inject Shiga Toxin into epithelial cells. 2) B subunit of the toxin binds to GB 3 receptors on the surface of eukaryotic cells 3) Shiga toxin is then internalized by endocytosis 4) It is transport to the Golgi apparatus >> Then transported to the endoplasmic reticulum (ER). 4) In the endoplasmic reticulum (ER), Shiga toxin encounters its target, the ribosome and inactivates it: (The A subunit has Nglycosidase activity that cleaves an adenosine residue from 28 S ribosomal RNA>>INACTIVE 28 S r. RNA) 5) As a consequence, Shiga toxin inhibits protein synthesis, causing cell death by apoptosis >> leads to destruction of intestinal epithelial cells, producing bloody diarrhea 6) Shiga toxins might get into the blood stream and targets the kidney. It can cause renal failure by destroying the glomerular endothelial >> hemolytic uremic syndrome (HUS).

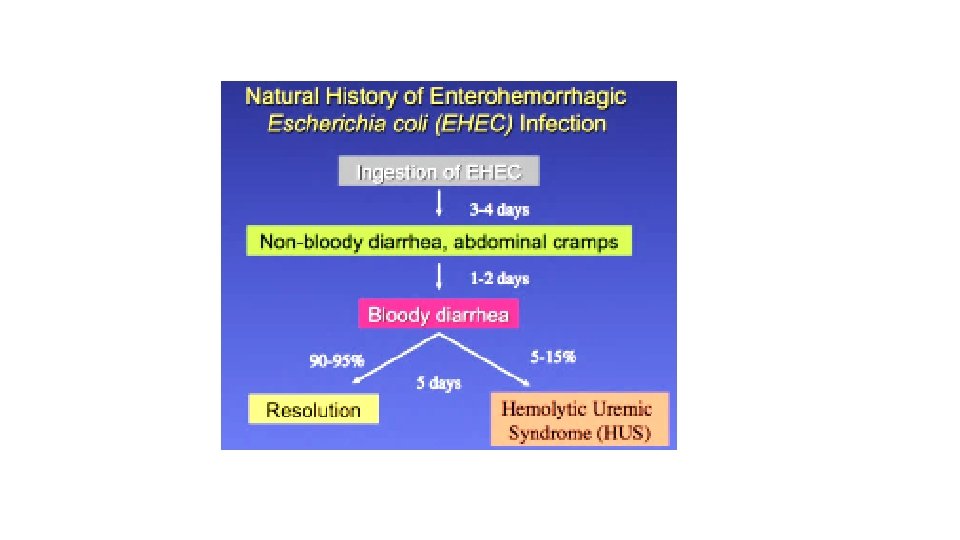
Shiga toxin-producing E. coli (STEC). Signs and symptoms • The incubation period can range from 3 to 8 days, with a median of 3 to 4 days. • Persons who experience bloody diarrhea or severe abdominal cramps should seek medical care. Ø A RARE BUT SERIOUS COMPLICATION • Most patients recover within 10 days, but in a small proportion of patients (10%: particularly young children and the elderly), the infection may lead to a life-threatening disease, such as haemolytic uraemic syndrome (HUS). • HUS occurs when Shiga toxins get into the bloodstream and cause the part of the kidney that filters toxins out of the blood to break down, causing kidney injury and sometimes kidney failure. (HUS is characterized by acute renal failure, haemolytic anaemia and thrombocytopenia (low blood platelets). • Some HUS patients also suffer damage to the pancreas and central nervous system impairment. • Antibiotics are not part of the treatment of patients with STEC disease and may possibly increase the risk of subsequent HUS.

Shiga toxin-producing E. coli (STEC). DIAGNOSIS • Shiga toxin-producing E. coli infection can be diagnosed in a doctor’s office or hospital by laboratory analysis of a stool sample. • Bacteria isolated from patients’ stool samples can be compared through laboratory analysis, helping to match strains of E. coli to the food or other source it came from, a process called “fingerprinting. ” .

Shiga toxin-producing E. coli (STEC). Transmission: 1. Through food or water contaminated with human or animal feces. Typical sources of contamination include: • Ground beef • Unpasteurized dairy products and juices, including apple juice • Produce exposed to water runoff from cattle farms • Well water or open water (lakes, rivers) frequented by animals 2. People can also pass E. coli to you if they touch your food or you (and you don't wash your hands before eating). 3. Additionally, you may get E. coli in your system if you ingest water while swimming in a contaminated lake, river, or swimming pool.

Shiga toxin-producing E. coli (STEC). Risk Factors: E. coli can affect anyone who is exposed to the bacteria. But some people are more likely to develop problems than are others. Ø Age. Young children and older adults are at higher risk of experiencing illness caused by E. coli and more-serious complications from the infection. Ø Weakened immune systems. People who have weakened immune systems — from AIDS or drugs to treat cancer or prevent the rejection of organ transplants — are more likely to become ill from ingesting E. coli. Ø Eating certain types of food. Riskier foods include undercooked hamburger; unpasteurized milk, apple juice or cider; and soft cheeses made from raw milk. Ø Time of year. Though it's not clear why, the majority of E. coli infections in the U. S. occur from June through September. Ø Decreased stomach acid levels. Stomach acid offers some protection against E. coli. If you take medications to reduce your levels of stomach acid, you may increase your risk of an E. coli infection.

Treatment: Shiga toxin-producing E. coli (STEC). For illness caused by E. coli, no current treatments can cure the infection, relieve symptoms or prevent complications. For most people, treatment includes: • Rest • The best treatment is oral fluid and electrolyte replacement • Avoid taking an anti-diarrheal medication — this slows your digestive system down, preventing your body from getting rid of the toxins. • Antibiotics generally aren't recommended because they can increase the risk of serious complications. • If you have a serious E. coli infection that has caused hemolytic uremic syndrome “HUS”, you'll be hospitalized and given supportive care, including IV fluids, blood transfusions and kidney dialysis.

Prevention: Shiga toxin-producing E. coli (STEC). Any food that you eat has the potential to be contaminated with E. coli bacteria. This is why it is important to take precautions in preparing food and before eating at restaurants. • Wash hands thoroughly before and after eating and after going to the bathroom • Sanitize all fruits and vegetables before eating by skinning them if possible and washing them before eating • Check with your local department of health to find out which restaurants in your area have had recent problems with sanitation • Avoid allowing raw meats to come into contact with other foods while cooking • Do not allow children to share bath water with anyone who has diarrhoea or symptoms of stomach flu • Wash hands thoroughly after any contact with farm animals • Wear disposable gloves when changing diapers of children with diarrhoea • Make sure ground meat (such as hamburger patties) reaches an internal temperature of at least 160°F • Avoid drinking any non-chlorinated water
- Slides: 60