MICROBIAL METABOLISM LAB Lab 4 MICROBIAL METABOLISM Microorganisms
MICROBIAL METABOLISM LAB Lab #4

MICROBIAL METABOLISM • Microorganisms Oxidize Inorganic & Organic Materials to Obtain Energy for Growth & Maintenance • Heterotrophic Organisms Use Portion of Organic Material for Energy, fe, which In Turn Used to Convert Remaining Portion of Organic Matter Into Cells, fs. • Autotrophic Organisms Oxidize Inorganic Compounds to Generate Energy, fe, Use Energy Released In Reaction to Reduce Carbon Dioxide to Form Cellular Materials, fs. • Whether Heterotrophic or Autotrophic, Portion of

BALANCED BIOCHEMICAL REACTIONS • Energy & Synthesis Reactions Combined, Accounting for what are Carbon Donors, Carbon Acceptors, and form of Nitrogen Available for Cell Synthesis • Overall Reaction Equation Given as R = fs. Rc + fe. Ra - Rd (1) where Rc represents the half reaction for synthesis of bacterial cells; Ra represents the half reaction for the electron acceptor; Rd represents the half reaction for the electron donor; fs and fe represent the portion of the


APPLICATION OF EQUATION 1 An anaerobic reactor is being used for the stabilization of high glucose content (1, 000 mg/L), ammonia containing wastewater. In this heterotrophic reaction, glucose is being used by anaerobic bacteria for energy and carbon for cell synthesis, and CO 2 is being used as the terminal electron acceptor. The expected end products of this anaerobic reaction are methane, CO 2, and cell biomass. Using the bioenergetics approach and the appropriate half-reactions and (fs)max values from Tables 1 and 2, write and balance the complete chemical equation describing microbial metabolism taking place in this reactor and determine the quantity of products generated from the conversion of 1, 000 L of this waste.

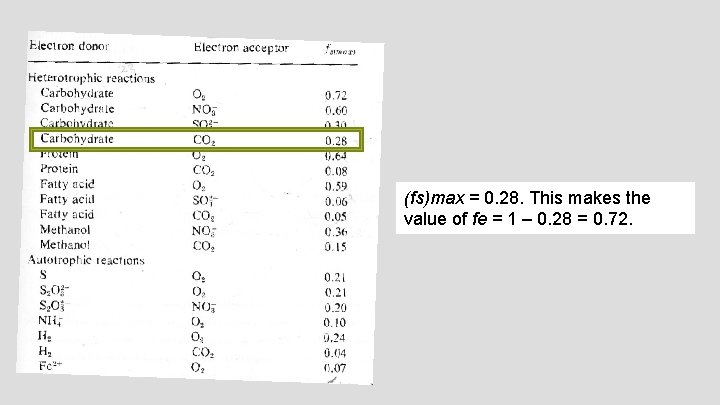
(fs)max = 0. 28. This makes the value of fe = 1 – 0. 28 = 0. 72.

APPLICATION OF EQUATION 1 R = fs. Rc + fe. Ra - Rd (1)

APPLICATION OF EQUATION 1 The final balanced equation indicates that for each mol of carbohydrate, 0. 056 mol ammonium are required to generated 0. 36 mol methane along with 0. 056 mol of microbial biomass (FW = 113 g/gmol). For 1, 000 L of waste treated, 1, 000 g of glucose is converted. The formula weight of glucose, C 6 H 12 O 6, is 12(6) +12 + 16(6) = 180 g/gmol. A total of (1, 000 g)/(180 g/gmol) = 5. 56 gmol glucose = 6 (5. 56) = 33. 36 gmol CH 2 O have been converted leading to the following: Ammonia-N needed = 0. 056 (33. 36) (14 g/gmol Ammonia-N) = 26. 15 g Methane generated = 0. 36 (33. 36) = 12 mol methane (22. 4 L/mol) = 269 L (STP) Biomass generated = 0. 056 (33. 36) (113 g/gmol) = 211 g cell biomass

QUESTIONS TO ANSWER FOR LAB 1. The autotrophic oxidation of ammonia to nitrate is an important reaction in wastewater engineering as it is the first step in the biological removal of nitrogen from wastewater. Determine the balanced equation for the ammonia to nitrate reaction (nitrification) using the (fs)max value from Table 2 and realizing that ammonia is available for use in cell synthesis. Determine the amount of oxygen required to oxidize 30 mg/L of ammonia and determine the biomass generated during this nitrification process.

QUESTIONS TO ANSWER FOR LAB 2. The second step in biological nitrogen removal in wastewater treatment is denitrification where the nitrate generated in the nitrification process is used as an electron acceptor by heterotrophic bacteria. Methanol can be used as the electron donor for this anoxic denitrification reaction. Using the (fs)max value from Table 2 and realizing that nitrate is used as the nitrogen source for cell synthesis, determine the balanced equation for denitrification with methanol as the carbon donor. If 100 mg/L of nitrate is denitrified in this reaction, determine the methanol dose required, as well as the cell biomass generated in the process.

QUESTIONS TO ANSWER FOR LAB 3. In anaerobic digesters, a first step in the stabilization of high strength waste is the conversion of complex organic materials to simpler fatty acids through fermentation. An example of this would be the anaerobic fermentation of glucose to acetic acid. Write the balanced equation for this anaerobic fermentation from glucose to acetate using an (fs)max value of 0. 10 and assuming ammonia is available for the production of microbial biomass. Determine the concentration of acetate generated in the fermentation of 1, 000 mg/L of glucose, and the concentration of cells produced during this fermentation process.
- Slides: 12