Microbial Metabolism 1 Metabolism Metabolism is all the

Microbial Metabolism 1

Metabolism • • • Metabolism is all the chemical reactions to sustain life. Metabolism = Catabolism + Anabolism Driven by energy, ATP 2

Metabolism • • Metabolism = Catabolism + Anabolism Catabolism makes ATP. Anabolism consumes ATP. Where does ALL this energy come from? 3

REVIEW Where does ALL energy on earth originate? A. plant life B. fossil fuels TIME IS UP !! C. the sun D. volcanic forces E. it’s man made. 10 seconds left !! 4

Groups based on metabolism Autotroph 1 - can make own food from CO 2. Heterotroph - needs organic source of C. Phototroph - uses light for energy. Chemotroph- uses chemicals for energy. 1 Troph means “to feed or to eat”. 5

Groups based on metabolism 1. Photoautotroph - can make own food from CO 2 using light. ex. plants, algae • Chemoautotroph - can make own food from CO 2 using inorganic chemicals. ex. sulfur and nitrogen bacteria • Photoheterotroph - uses light for energy needs + organic source of C. ex. green and purple nonsulfur bacteria • Chemoheterotroph - uses chemicals for both energy and source of C. ex. most organisms here -animals, fungi, bacteria, protozoa. 6
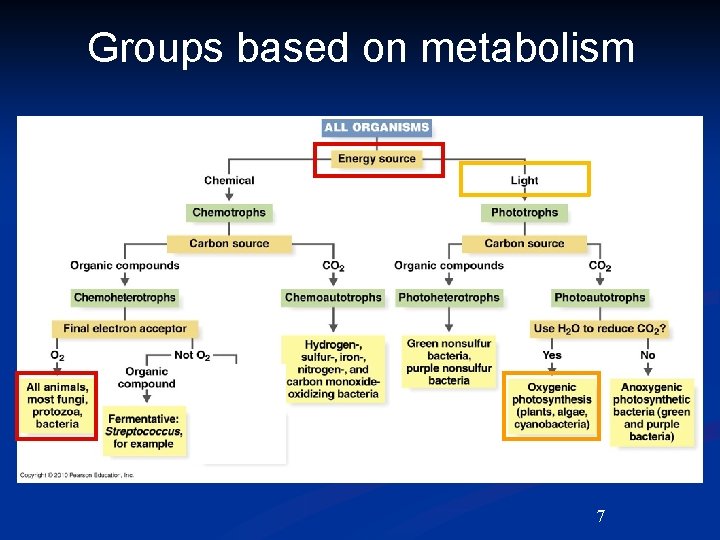
Groups based on metabolism 7
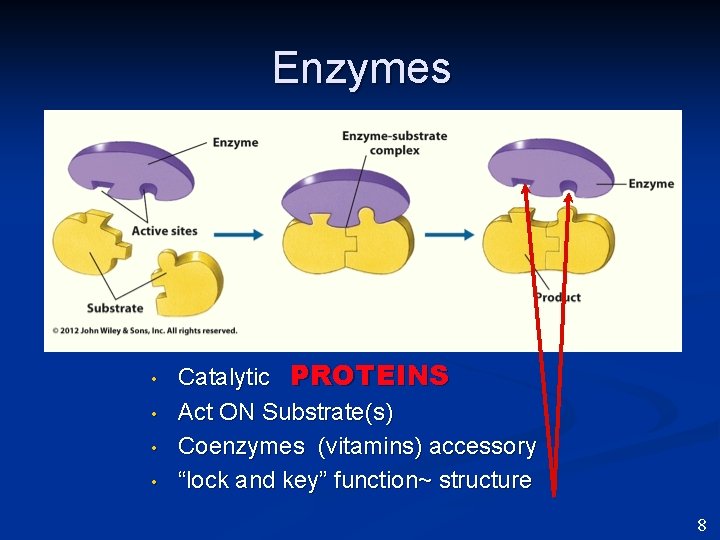
Enzymes • • Catalytic PROTEINS Act ON Substrate(s) Coenzymes (vitamins) accessory “lock and key” function~ structure 8
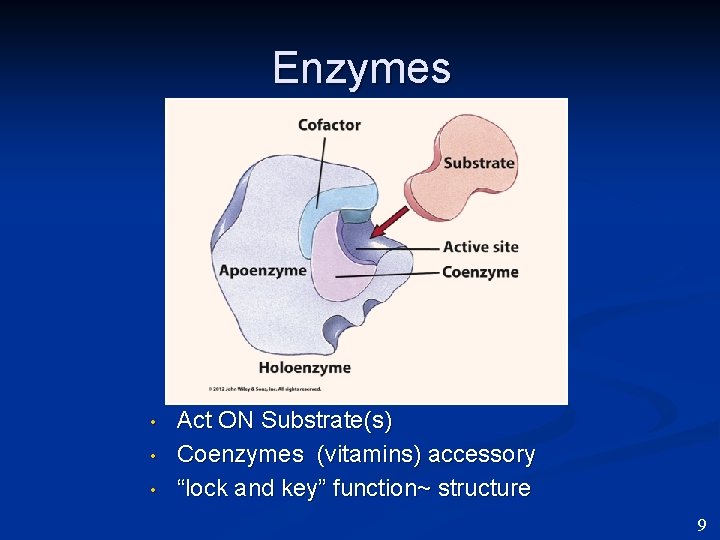
Enzymes • • • Act ON Substrate(s) Coenzymes (vitamins) accessory “lock and key” function~ structure 9
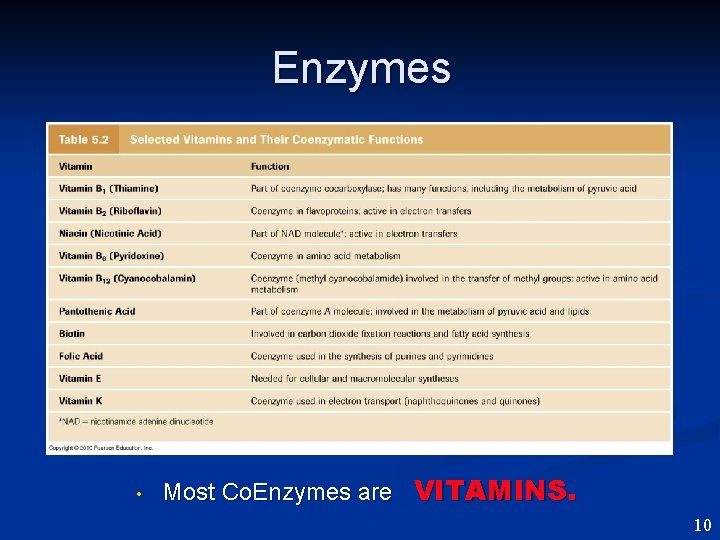
Enzymes • Most Co. Enzymes are VITAMINS. 10
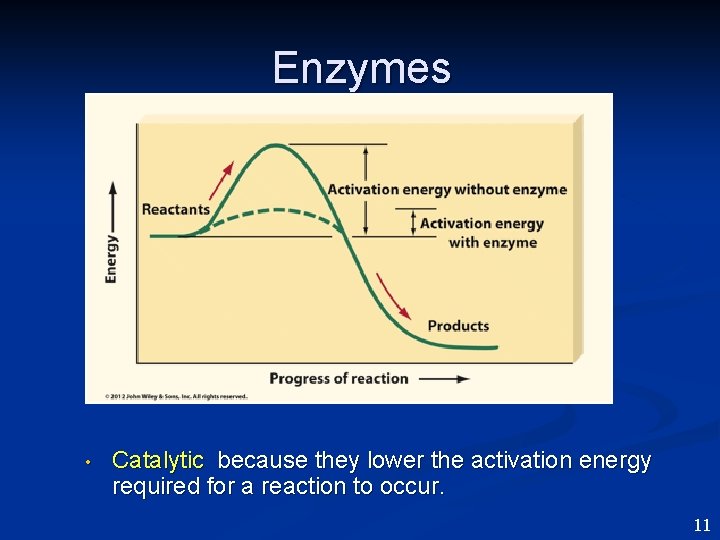
Enzymes • Catalytic because they lower the activation energy required for a reaction to occur. 11

Enzymes • Catalytic Proteins • • Re-useable - NOT consumed Active site “lock and key” function The turnover number is generally 1 -10, 000 molecules per second. Once they reach maximum rate, they won’t go higher. 12
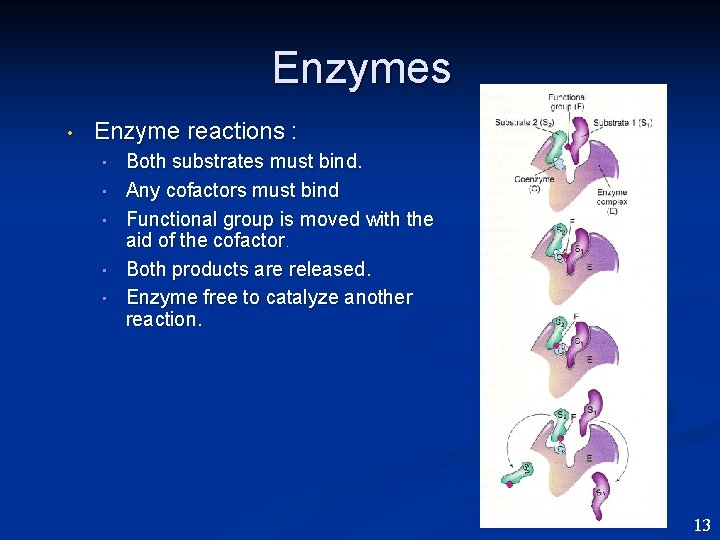
Enzymes • Enzyme reactions : • • • Both substrates must bind. Any cofactors must bind Functional group is moved with the aid of the cofactor. Both products are released. Enzyme free to catalyze another reaction. 13
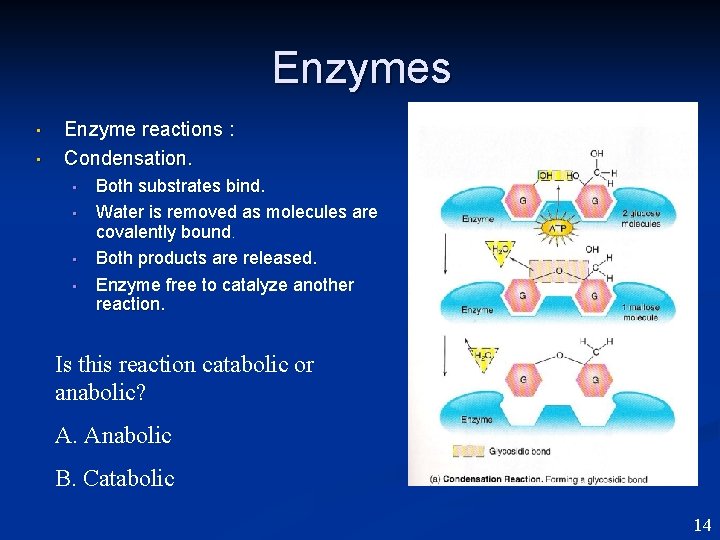
Enzymes • • Enzyme reactions : Condensation. • • Both substrates bind. Water is removed as molecules are covalently bound. Both products are released. Enzyme free to catalyze another reaction. Is this reaction catabolic or anabolic? A. Anabolic B. Catabolic 14

Enzymes • • Enzyme reactions : Hydrolysis. • • Single substrate and H 2 O binds. Water is added as covalent bond is broken. Both products are released. Enzyme free to catalyze another reaction. Is this reaction catabolic or anabolic? A. Anabolic B. Catabolic 15
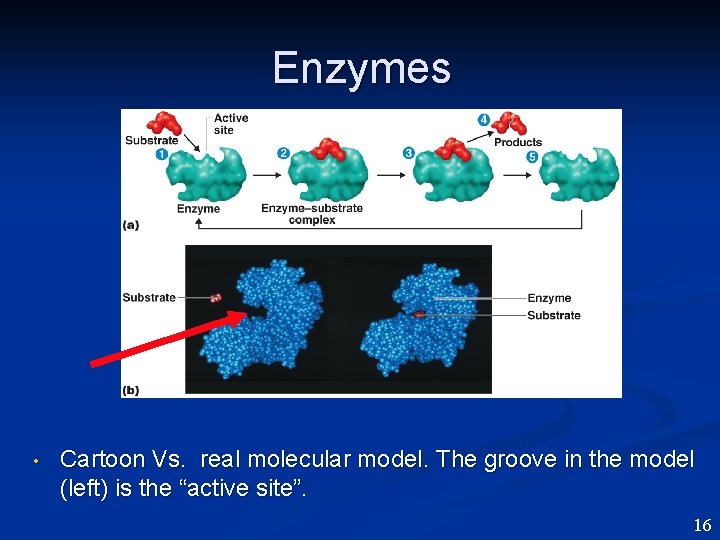
Enzymes • Cartoon Vs. real molecular model. The groove in the model (left) is the “active site”. 16

Enzyme Catalysis • Some reactions are reversible, some NOT. 1. Glucose + ATP 2. 2 H 2 O 2 hexokinase Glucose-6 -phosphate + ADP catalase 2 H 2 O + O 2 ALL substrates must be present for reaction to occur. IF you have 5 µmoles of glucose and 10 µmoles of ATP, reaction #1 will proceed to produce 5 µmoles of glucose 6 -phosphate and then stop. 17
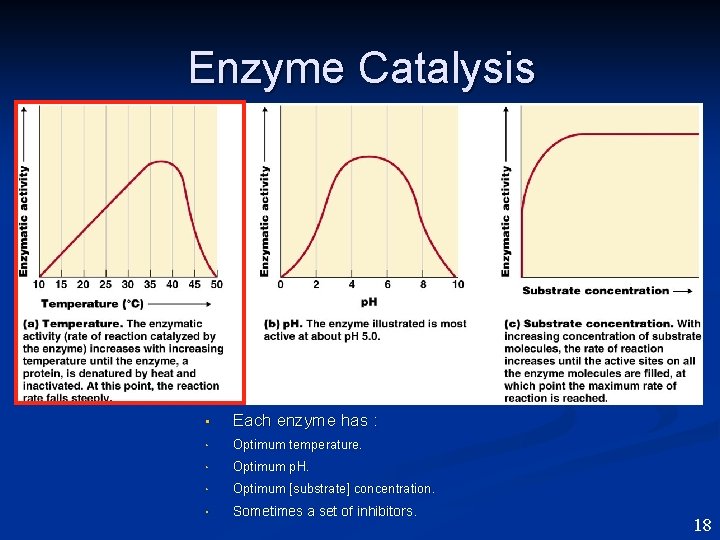
Enzyme Catalysis • Each enzyme has : • Optimum temperature. • Optimum p. H. • Optimum [substrate] concentration. • Sometimes a set of inhibitors. 18
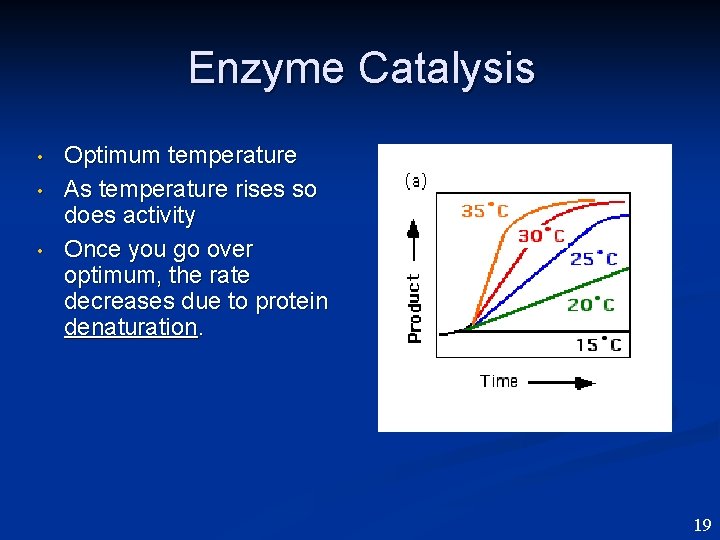
Enzyme Catalysis • • • Optimum temperature As temperature rises so does activity Once you go over optimum, the rate decreases due to protein denaturation. 19
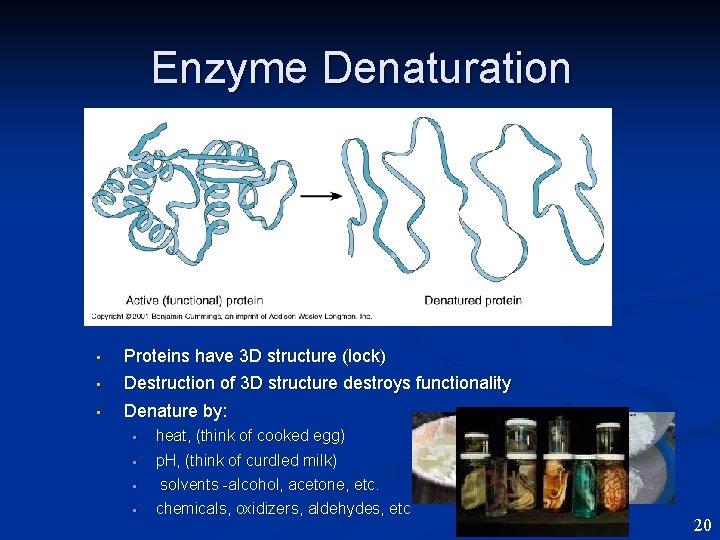
Enzyme Denaturation • Proteins have 3 D structure (lock) Destruction of 3 D structure destroys functionality • Denature by: • • • heat, (think of cooked egg) p. H, (think of curdled milk) solvents -alcohol, acetone, etc. chemicals, oxidizers, aldehydes, etc 20
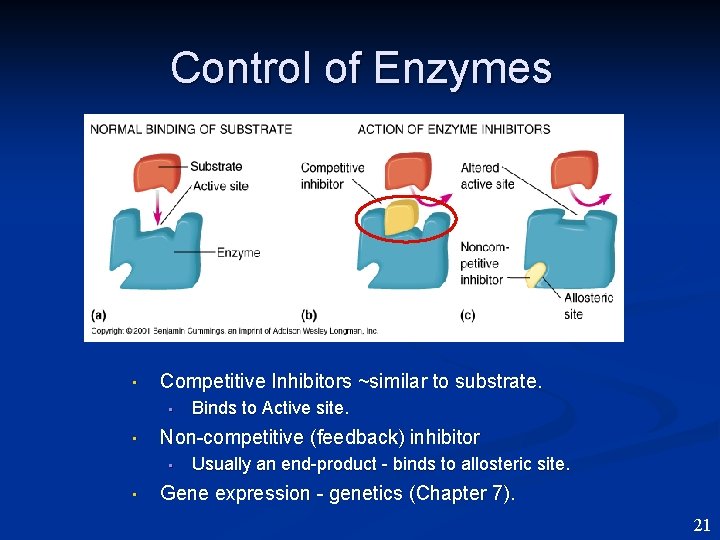
Control of Enzymes • Competitive Inhibitors ~similar to substrate. • • Non-competitive (feedback) inhibitor • • Binds to Active site. Usually an end-product - binds to allosteric site. Gene expression - genetics (Chapter 7). 21
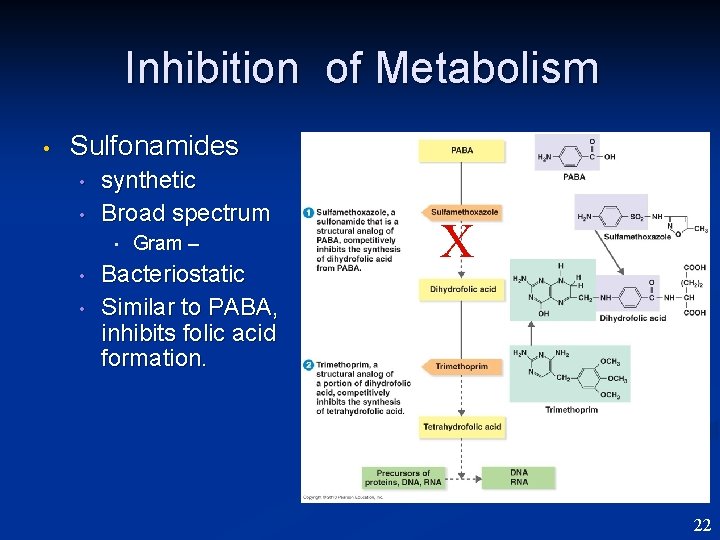
Inhibition of Metabolism • Sulfonamides • • synthetic Broad spectrum • • • Gram – Bacteriostatic Similar to PABA, inhibits folic acid formation. X 22

Feedback of Enzymes • • • Occurs in a metabolic pathway Pathway is usually anabolic. End products of pathway inhibit enzymes at the beginning (feedback). Prevent over-production. Conserve energy, resources. STOPPED 23

Oxidation - Reduction • • Reduction- gain of electrons Oxidation - loss of electrons Must have both. One chemical gains - one loses e-. 24
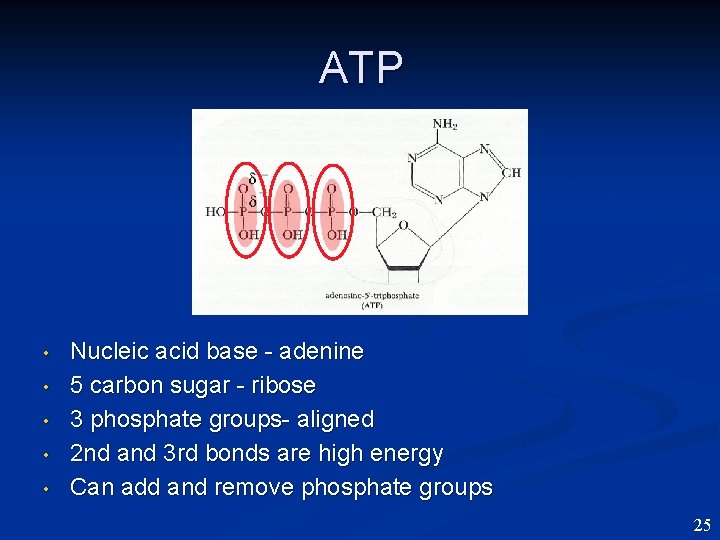
ATP • • • Nucleic acid base - adenine 5 carbon sugar - ribose 3 phosphate groups- aligned 2 nd and 3 rd bonds are high energy Can add and remove phosphate groups 25
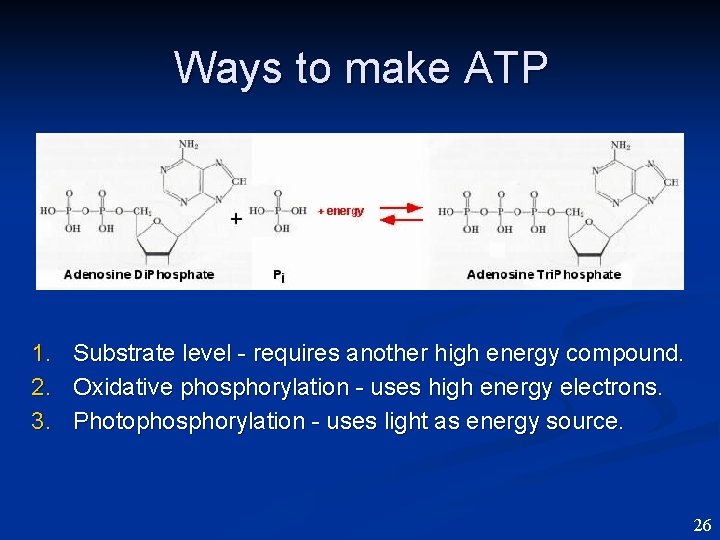
Ways to make ATP 1. Substrate level - requires another high energy compound. 2. Oxidative phosphorylation - uses high energy electrons. 3. Photophosphorylation - uses light as energy source. 26
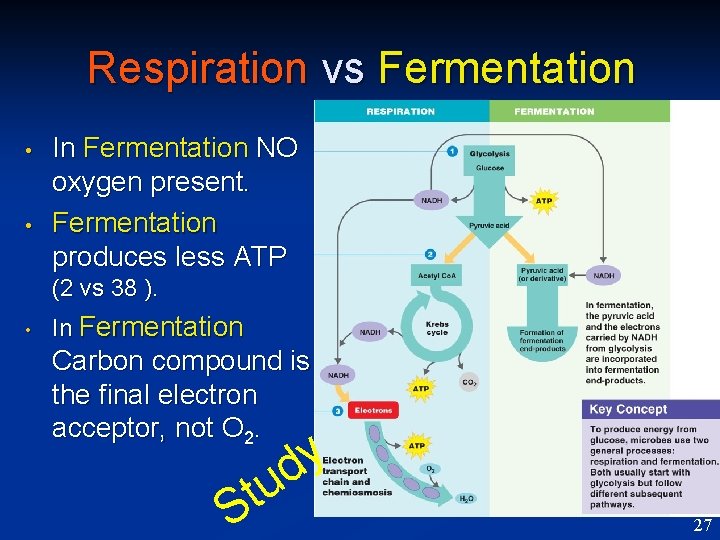
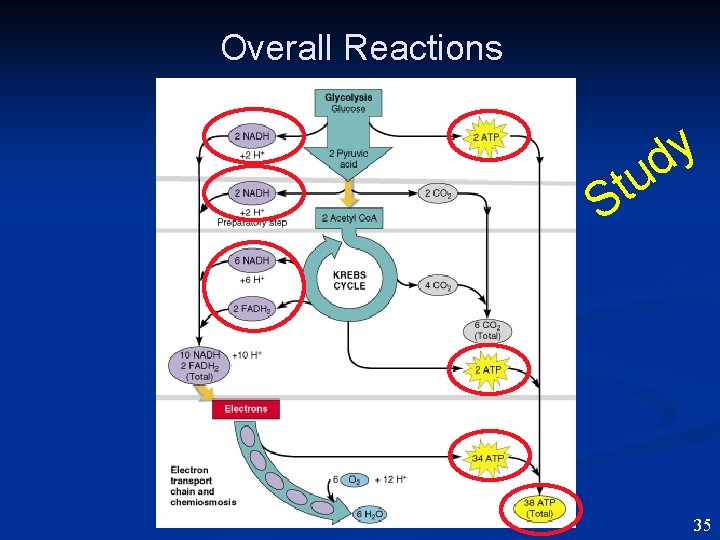
Respiration vs Fermentation • • In Fermentation NO oxygen present. Fermentation produces less ATP (2 vs 38 ). • In Fermentation Carbon compound is the final electron acceptor, not O 2. u t S y d 27
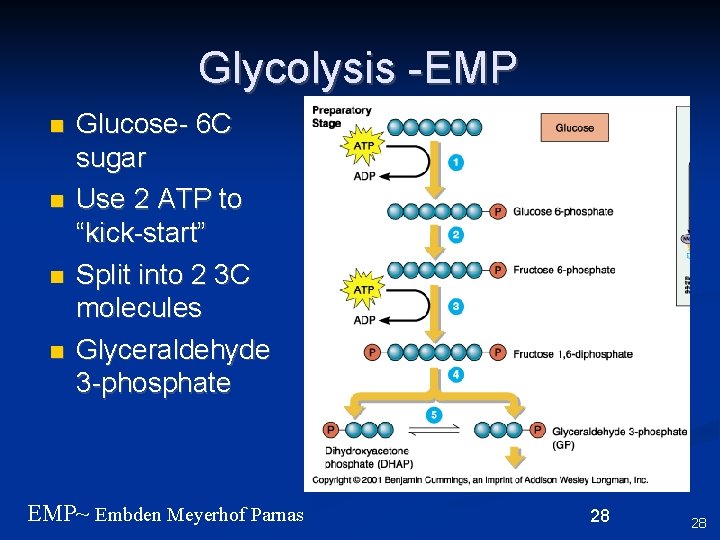
Glycolysis -EMP n n Glucose- 6 C sugar Use 2 ATP to “kick-start” Split into 2 3 C molecules Glyceraldehyde 3 -phosphate EMP~ Embden Meyerhof Parnas 28 28
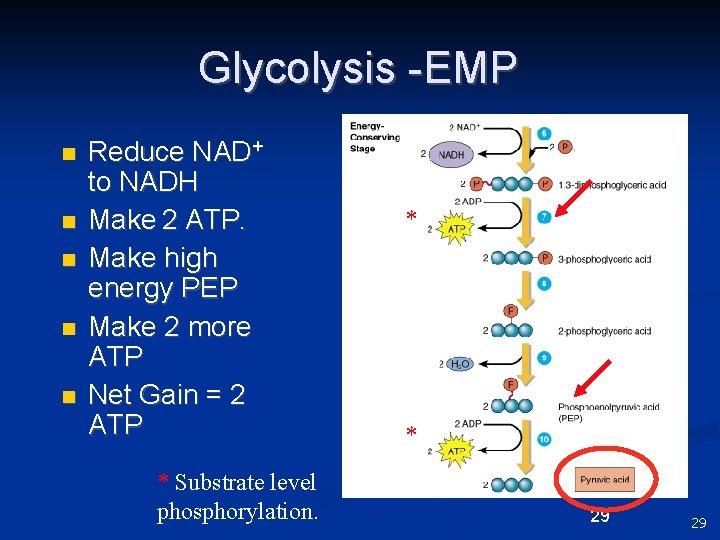
Glycolysis -EMP n n n Reduce NAD+ to NADH Make 2 ATP. Make high energy PEP Make 2 more ATP Net Gain = 2 ATP * Substrate level phosphorylation. * * 29 29

Alternatives to Glycolysis n Pentose Phosphate n Makes pentoses for: n n n Nucleic acids Some Amino acids Entner Doudoroff 2 NADPH + ATP Used for biosynthesis NADPH + ATP 30 30
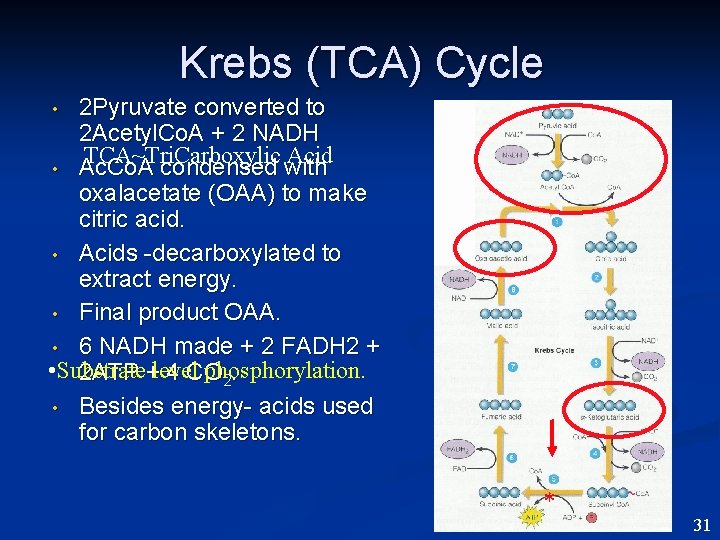
Krebs (TCA) Cycle 2 Pyruvate converted to 2 Acetyl. Co. A + 2 NADH TCA~Tri. Carboxylic Acid • Ac. Co. A condensed with oxalacetate (OAA) to make citric acid. • Acids -decarboxylated to extract energy. • Final product OAA. • 6 NADH made + 2 FADH 2 + • Substrate phosphorylation. 2 ATP +level 4 CO 2. • Besides energy- acids used for carbon skeletons. • * 31
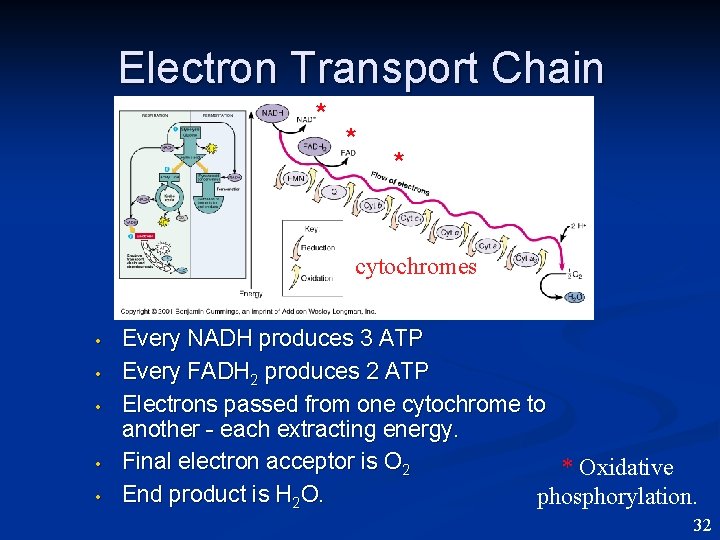
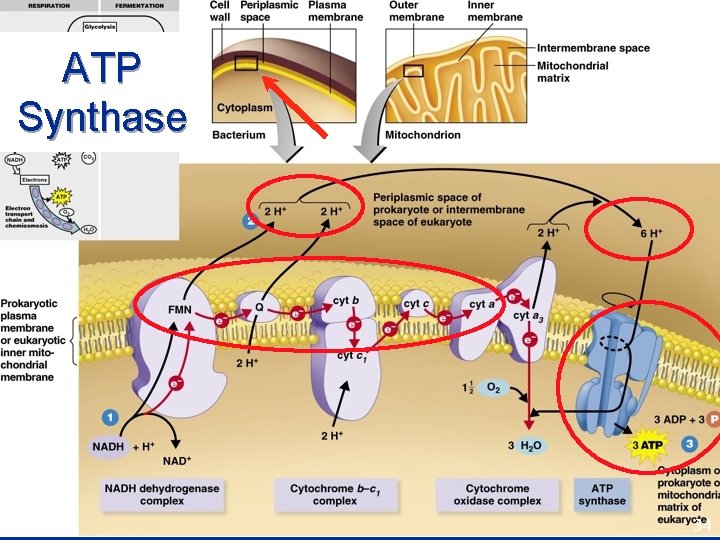
Electron Transport Chain * * * cytochromes • • • Every NADH produces 3 ATP Every FADH 2 produces 2 ATP Electrons passed from one cytochrome to another - each extracting energy. Final electron acceptor is O 2 * Oxidative End product is H 2 O. phosphorylation. 32
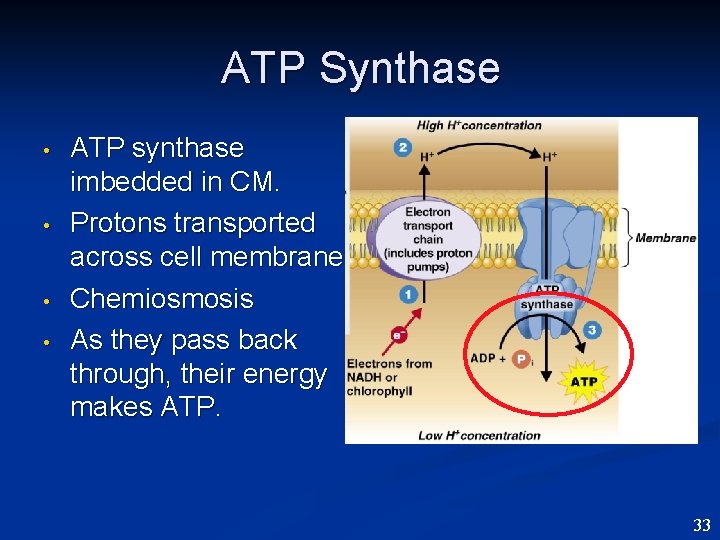
ATP Synthase • • ATP synthase imbedded in CM. Protons transported across cell membrane Chemiosmosis As they pass back through, their energy makes ATP. 33
ATP Synthase 34
Overall Reactions y d tu S 35
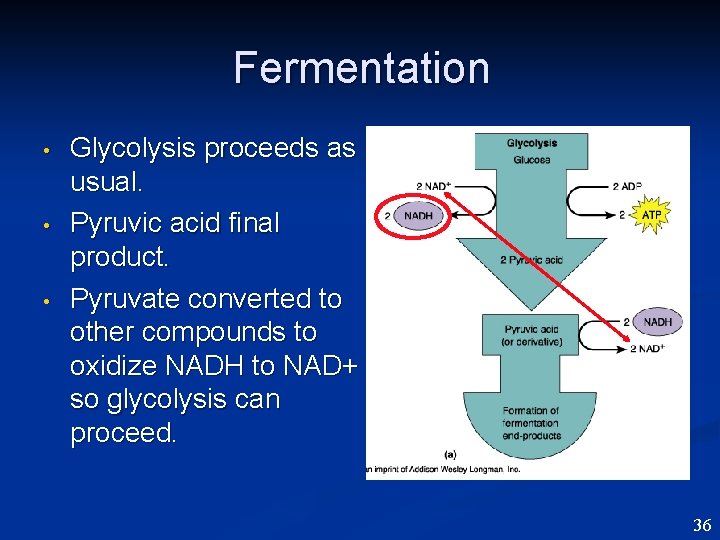
Fermentation • • • Glycolysis proceeds as usual. Pyruvic acid final product. Pyruvate converted to other compounds to oxidize NADH to NAD+ so glycolysis can proceed. 36
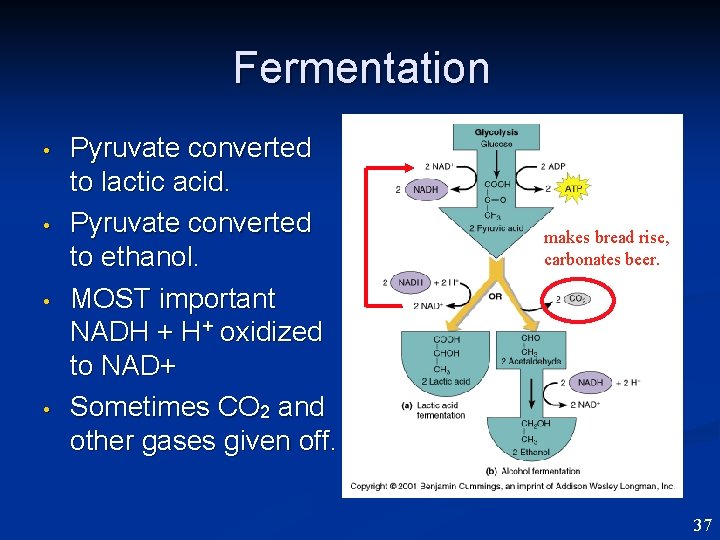
Fermentation • • Pyruvate converted to lactic acid. Pyruvate converted to ethanol. MOST important NADH + H+ oxidized to NAD+ Sometimes CO 2 and other gases given off. makes bread rise, carbonates beer. 37

Aerobic Respiration Vs. Fermentation • • • Aerobe - Glycolysis + Kreb’s + Electron Transport. Anaerobe - only glycolysis and fermentation. Facultative anaerobe - both. Aerobic respiration Fermentation Pseudomonas E. coli 38
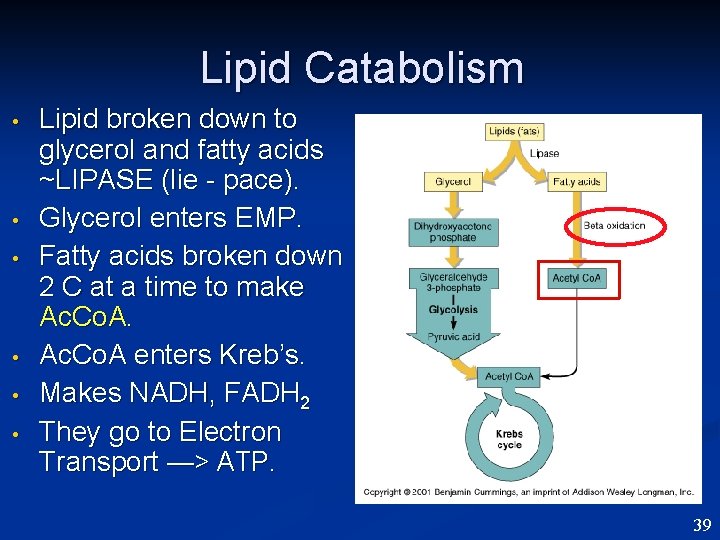
Lipid Catabolism • • • Lipid broken down to glycerol and fatty acids ~LIPASE (lie - pace). Glycerol enters EMP. Fatty acids broken down 2 C at a time to make Ac. Co. A enters Kreb’s. Makes NADH, FADH 2 They go to Electron Transport —> ATP. 39
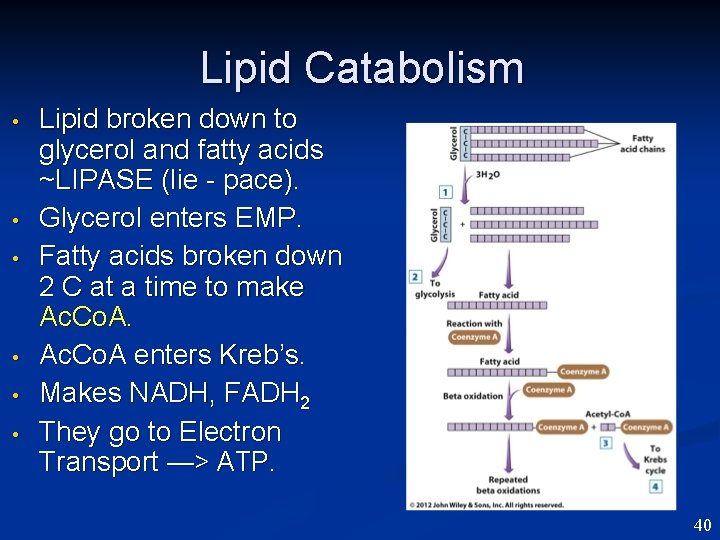
Lipid Catabolism • • • Lipid broken down to glycerol and fatty acids ~LIPASE (lie - pace). Glycerol enters EMP. Fatty acids broken down 2 C at a time to make Ac. Co. A enters Kreb’s. Makes NADH, FADH 2 They go to Electron Transport —> ATP. 40
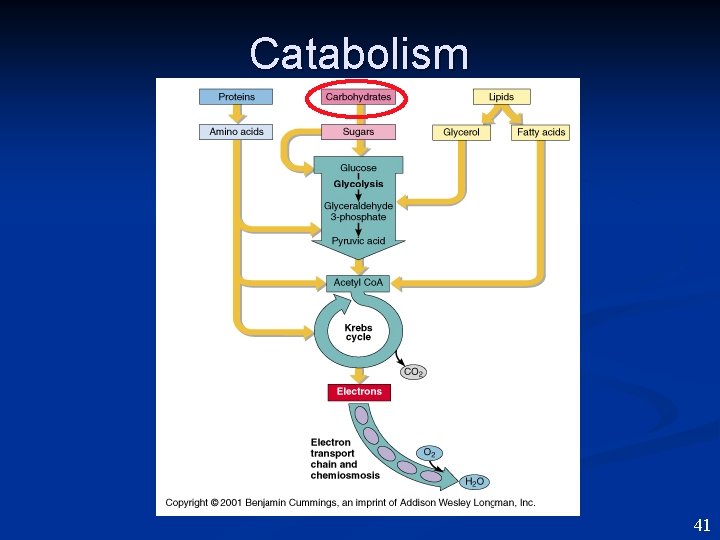
Catabolism 41
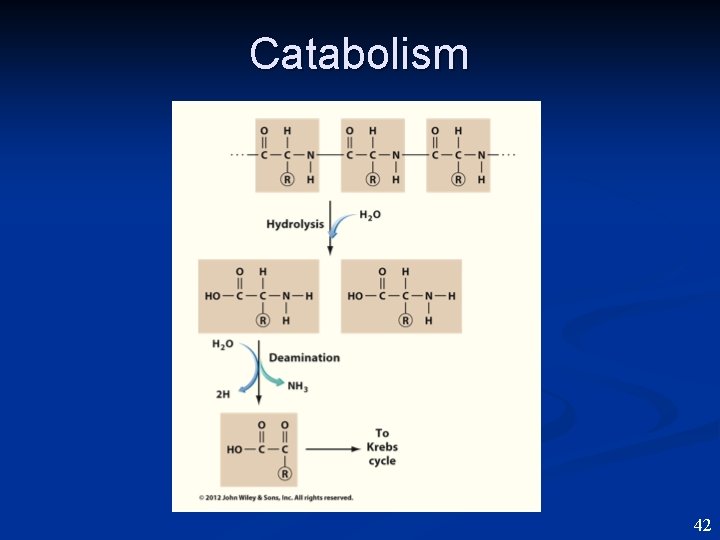
Catabolism 42

Photophosphorylation • • • Sunlight excites chlorophyll Energized chlorophyll passes high energy electrons down electron transport. ATP made. 43
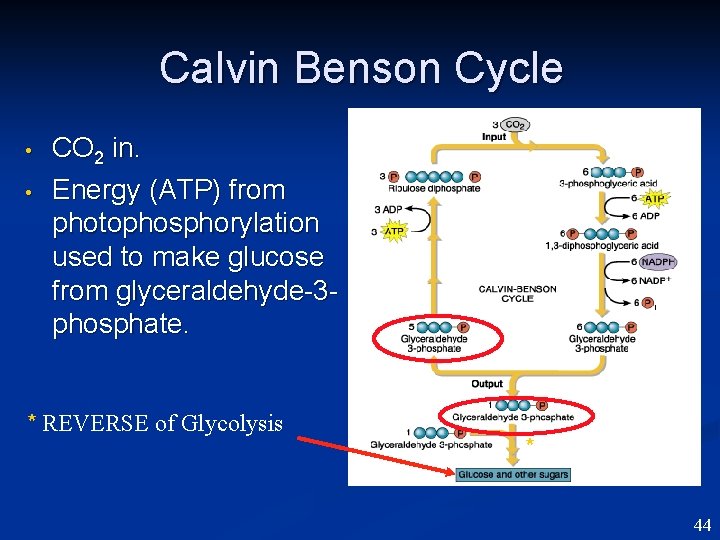
Calvin Benson Cycle • • CO 2 in. Energy (ATP) from photophosphorylation used to make glucose from glyceraldehyde-3 phosphate. * REVERSE of Glycolysis * 44
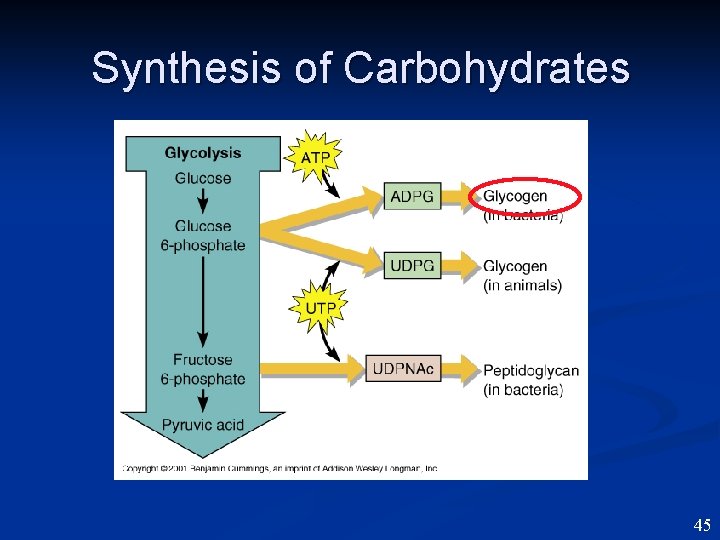
Synthesis of Carbohydrates 45
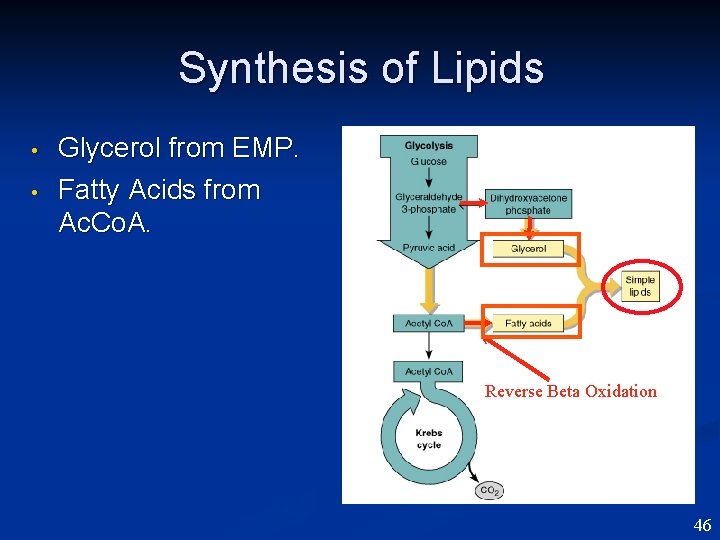
Synthesis of Lipids • • Glycerol from EMP. Fatty Acids from Ac. Co. A. Reverse Beta Oxidation 46
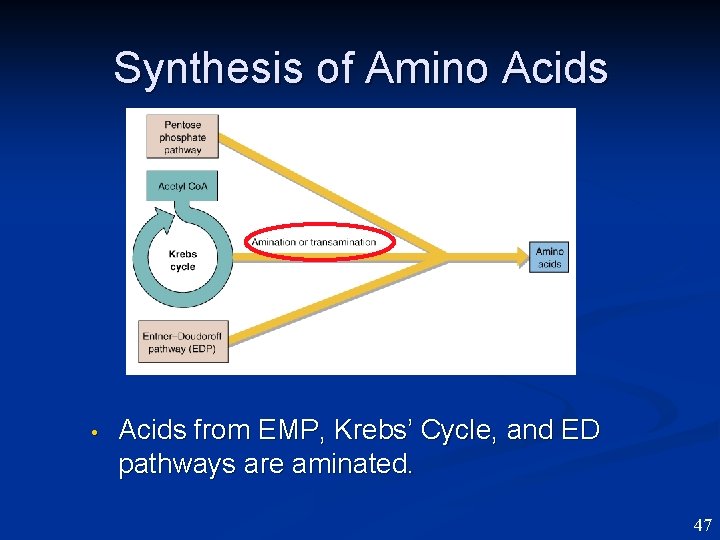
Synthesis of Amino Acids • Acids from EMP, Krebs’ Cycle, and ED pathways are aminated. 47
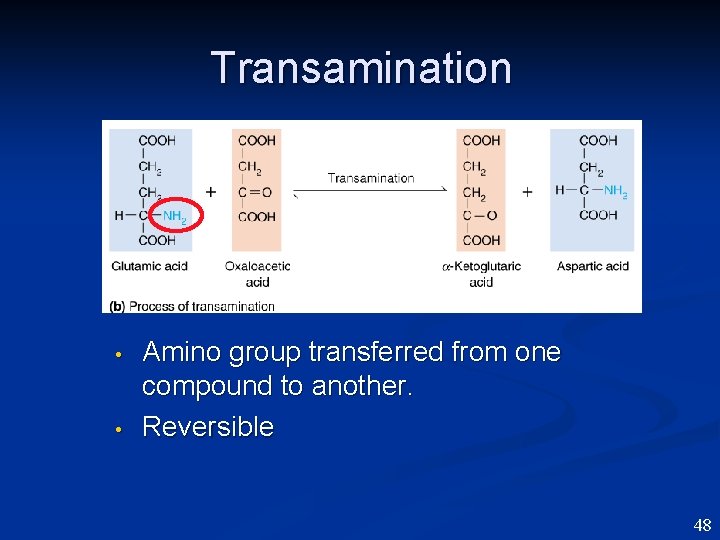
Transamination • • Amino group transferred from one compound to another. Reversible 48
Synthesis of Nucleotides DO NOT Memorize. 49
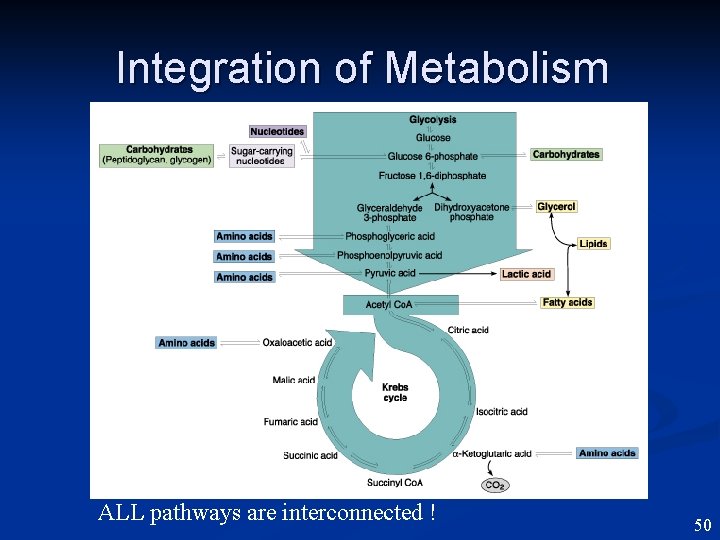
Integration of Metabolism ALL pathways are interconnected ! 50

What to remember ? • • • EVERYTHING ! (just kidding) Basic characteristics of Enzymes & reactions. Major Pathways - purpose. EMP Bridging reaction Catabolic or anabolic ? Krebs Cycle How are they controlled? Electron Transport Pentose Phosphate Entner Duodoroff Calvin Benson Beta Oxidation 51

THE END 52
- Slides: 52