Microbial conversion of inorganic substances BioGeoChemical Cycles Anaerobic

Microbial conversion of inorganic substances (Bio-Geo-Chemical Cycles) Anaerobic respirations of inorganic electron acceptors Aerobic oxidation of the endproducts of anaerobic respirations Cycles (C, N, S, Fe) Industrially and environmentally relevant reactions 1

How Can Life without Oxygen Work? O 2 = principal electron acceptor of aerobic life. Without O 2 a different electron acceptor needs to be found. Bacteria are the only life forms capable of using electron acceptors other than O 2 (anaerobic respiration). The use of alternative electron acceptors dramatically changes the chemistry of the environment – usually to the worse. 2

What are the Typical Electron Accepting Reactions? O 2 + 4 e- H 2 O (aerobic respiration) SO 42 - + 8 e- H 2 S (sulfate reducing bacteria) Fe 3+ + 1 e- Fe 2+ (iron reducing bacteria) S + 2 e- H 2 S (sulfur reducing bacteria) NO 3 - + 5 e- N 2 (denitrifying bacteria) NO 3 - + 8 e- NH 3 (nitrate ammonification) CO 2 + 8 e- CH 4 (methane producing bacteria) 3

What Happens with the Electron Acceptors after Accepting Electrons? By accepting electrons, the acceptors they turn into reduced species. Reduced species are reducing agents that dramatically change the chemistry of the environment If in contact with O 2 , reduced species can become electron donors for specialised lithotrophic bacteria The continued cycle of electron acceptors to reduced species and back to electron acceptors is a typical part of biogeochemical cycling. The S, N, Fe cycle are typical examples. 4

Classification of Microbial Metabolic Types Examples: §Algae: Photo-Litho-Autotroph, §Bacteria, Fish: Chemo-organo-heterotroph §Thiobacillus: Chemo-litho-autotroph §Photosynthetic bacteria: Photo-Organo-heterotroph 5

Life without O 2: Alternative Electron Acceptors 1 Electron Acceptors O 2 NO 3 - Fe 3+ SO 42 - So CO 2 Hum. Ox H 2 O N 2 NH 4 Fe 2+ H 2 S CH 4 Acetate Hum. Red Measure for Energy Released with H 2 as Electron Donor (log K) 21 21 15 8 5 3 2 ? ? adapted from Stumm & Morgan Observation: There is a sequence of use of electron acceptors which is according to the energetic usefulness (redox potential) O 2 , nitrate, humic acids, ferric iron, sulfate, CO 2 6

Electron Acceptors are Reduced and can become e- Donors Electron Acceptors organic O 2 H 2 S So NO 3 - So SO 42 - NO 3 - NH 4+ Fe 2+ Fe 3+ Fe 2+ NO 2 - NO 3 - SO 42 - H 2 S H 2 H+ So H 2 S CO CO 2 Acetate NH 4 NO 2 - CO 2 CH 4 Hum. Ox Hum. R H 2 O N 2 Hum. R 7

Interconnection Between Different Electron Donors and Acceptors Electron Donors H 2 H+ Electron Acceptors CO 2 CH 4 So H 2 S CH 4 CO 2 SO 42 - H 2 S So Fe 3+ Fe 2+ SO 42 - NO 3 - NH 4 Fe 2+ Fe 3+ NO 3 - N 2 NO 2 - NO 3 - O 2 H 2 O So 8
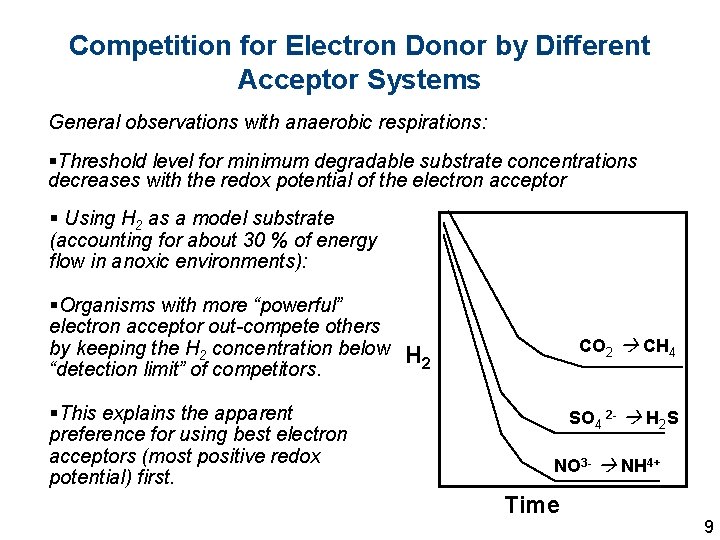
Competition for Electron Donor by Different Acceptor Systems General observations with anaerobic respirations: §Threshold level for minimum degradable substrate concentrations decreases with the redox potential of the electron acceptor § Using H 2 as a model substrate (accounting for about 30 % of energy flow in anoxic environments): §Organisms with more “powerful” electron acceptor out-compete others by keeping the H 2 concentration below H 2 “detection limit” of competitors. §This explains the apparent preference for using best electron acceptors (most positive redox potential) first. CO 2 CH 4 SO 4 2 - H 2 S NO 3 - NH 4+ Time 9

Sulfate Reduction (SRB) Sulfate is a suitable and abundant alternative electron acceptor Typical reactions: 4 H 2 + SO 42 - + H+ CH 3 -COO- + SO 42 - HS- + 4 H 2 O HS- + 2 HCO 3 - Organisms: Sulfate Reducing Bacteria (SRB), strictly anaerobes, Desulfovibrio, Desulfobacter, etc. Electron donors: small molecules (breakdown products from fermentations, or geologically formed, e. g. H 2, acetate, organic acids, alcohols) Reduce also elemental sulfur, sulfite and thiosulfate to H 2 S Ubiquitous 10

Bacterial Sulfate Reduction When Does it Occur ? §In the presence of organic substances , after depletion of oxygen nitrate and ferric iron sulfate reduction is next §Initially in sediments, Rates from 0. 01 to 10 m. M/day § Typically in sediments but also on surfaces (ships) underneath biofilms §Within flocs or intestines of marine animals §Sulfide reacts chemically as a reducing agent (e. g. with O 2 or Fe 3+) elemental sulfur formation §Formation of Fe. S and Fe. S 2 black color of sediment §In typical reduced sediments (e. g. mangroves, estuaries SR may be higher than O 2 use) Thiosulfate disproportionation (SO 32 -) into sulfate and sulfide 11

Dissimilatory Sulfate Reduction by SRB § Organisms: Sulfate Reducing Bacteria (SRB), strictly anaerobe § Desulfovibrio, Desulfobacter, etc. § Use of small compounds (H 2, acetate, other organic acids alcohols but not polymers, proteins, carbohydrates, fats) § Cooperation with fermentative bacteria needed to degrade detritus § End product sulfide (H 2 S HS- + H+) is toxic, reactive, explosive § Typical reactions: 4 H 2 + SO 42 - + H+ HS- + 4 H 2 O CH 3 -COO- + SO 42 - HS- + 2 HCO 3 - Reduce also elemental sulfur, sulfite and thiosulfate to H 2 S 12

SRB Significance in Marine Environments Ecologically: playing a major role in sulfur cycle and sediment activities sulfide = O 2 scavenger “negative oxygen concentration” responsible for sulfur deposits (H 2 S + O 2 S + H 2 O) P-release from sediments Economically: End product H 2 S: poisonous, explosive, corrosive, malodorous Corrosion of submersed steel structures (e. g. platforms, bridges) Corrosion of oil pipelines (inside and outside) Lethal gas emissions on offshore platforms Petroleum degradation (burning sour gas: H 2 S + O 2 SO 2) 13

S-ox: Volcanic Sulfur Springs E. g. New Zealand Yelllowstone National Park 14
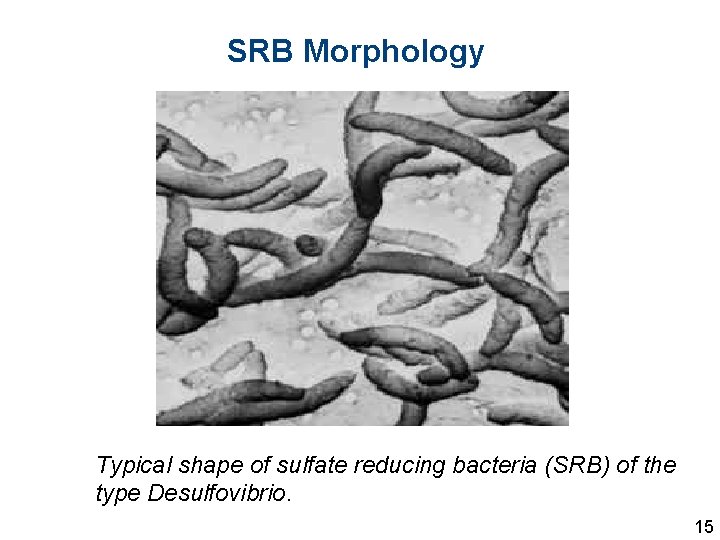
SRB Morphology Typical shape of sulfate reducing bacteria (SRB) of the type Desulfovibrio. 15
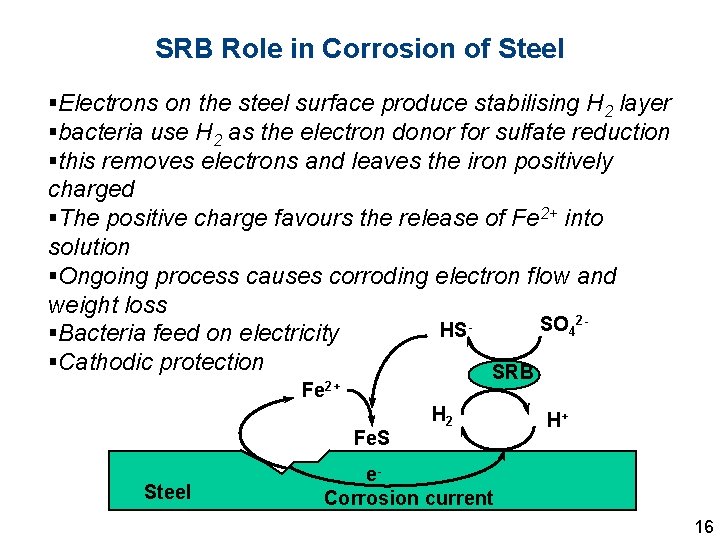
SRB Role in Corrosion of Steel §Electrons on the steel surface produce stabilising H 2 layer §bacteria use H 2 as the electron donor for sulfate reduction §this removes electrons and leaves the iron positively charged §The positive charge favours the release of Fe 2+ into solution §Ongoing process causes corroding electron flow and weight loss 2 SO 4 HS §Bacteria feed on electricity §Cathodic protection SRB Fe 2+ Fe. S Steel H 2 H+ e. Corrosion current 16

SRB Damage to Pipeline Microbially influenced corrosion of marine oil pipeline showing typical pitting corrosion. 17

“SRB in Petroleum Industry” Research at Murdoch Q: Where do SRB in oil pipelines come from? A: Mostly as a biofilm attached inside the pipes. Method SRB monitoring during pig runs. Q: Are SRB supported by corrosion ? A: SRB can grow on corroding iron. Cathodic protection enhances their growth. Q: Are treatments effective against SRB? A: SRB can degrade organic biocides. 18

Dissimilatory Nitrate Reducing Bacteria § Dentrification (nitrate to N 2) typically involves the aerobic bacteria §Organic electron donor + NO 3 - N 2 § Bacteria use complex substrates §Further details in lecture on N-cycle §In sediments nitrate ammonification can play important role §Organic electron donor + NO 3 - NH 3 §Nitrate ammonification is due to anaerobic bacteria (e. g. SRB) 19

Dissimilatory Iron Reducing Bacteria § Organisms: Various anaerobic bacteria, no specific group § e. g. Geobacter § e- donors: mainly small compounds § Typical reaction: H 2 + 2 Fe 3+ 2 Fe 2+ + 2 H+ § Reaction results in lowering of redox potential § Reduce also Manganese, elemental sulfur and other metals (e. g. uranium) § End product is magnetite (Fe 3 O 4) and other compounds (black precipitates) § Significance of iron reduction is still being underestimated §Recent research: electricity production using ferric iron reducing bacteria 20

CO 2 or HCO 3 - Reduction (Methane Producing Bacteria) § CO 2 is even more abundant than sulfate but difficult to use § By Methane Producing Bacteria (Archeae) § Strictly anaerobic requiring a redox potential of less than -350 m. V § Highly oxygen sensitive: 4 H 2 + HCO 3 - + H+ CH 4 + 3 H 2 O § Very limited substrate spectrum (H 2, acetate, methanol) § Syntrophic associations are formed with fermenting bacteria §Because of poor solubility (bubble formation) some methane from sediments escapes into atmosphere (greenhouse gas) True removal of BOD from water. 21

Aerobic Re-oxidation Processes 1 - Sulfide and Fe 2+ §Contact of reduced (black sediments) with O 2 : bacterial oxidation of sulfide and Fe 2+ occurs. § Beggiatoa: 2 H 2 S + O 2 2 S 0 + H 2 O (white algae) §Thiobacillus: H 2 S + 2 O 2 H 2 SO 4 (sulfuric acid) very low local p. H values of <1. Further microbial pipeline corrosion. § Also insoluble species are re-oxidized e. g. pyrite (Fe. S 2) bio-leaching of minerals). § Elemental sulfur is often produced as intermediate (white precipitate (“white smoker”, “white algae”) 22

Aerobic Re-oxidation Processes 2 - Ammonia NH 4+ oxidation is energetically difficult and slow and requires oxygen as electron acceptor. Organisms: Nitrosomonas, Nitrobacter, two step process. NH 4 uptake also possible by assimilation of phytoplankton. 23

Fate of Sulfide in the Presence of Oxygen Contact of reduced sulfide (H 2 S or Fe. S) with air spontaneous oxidation (H 2 S) to insoluble S Microbial Oxidation: (a) Beggiatoa: 2 H 2 S + O 2 2 So + H 2 O (“white algae”) (b) Thiobacillus: H 2 S + 2 O 2 H 2 SO 4 (sulfuric acid) very low local p. H values of <1. Further microbial pipeline corrosion. Also insoluble species are re-oxidized e. g. pyrite (Fe. S 2) (bio-leaching of minerals). Elemental sulfur is often produced as intermediate (white precipitate (“white smoker”, “white algae”) Together, sulfate reduction and sulfide oxidation can close the sulfur cycle. 24
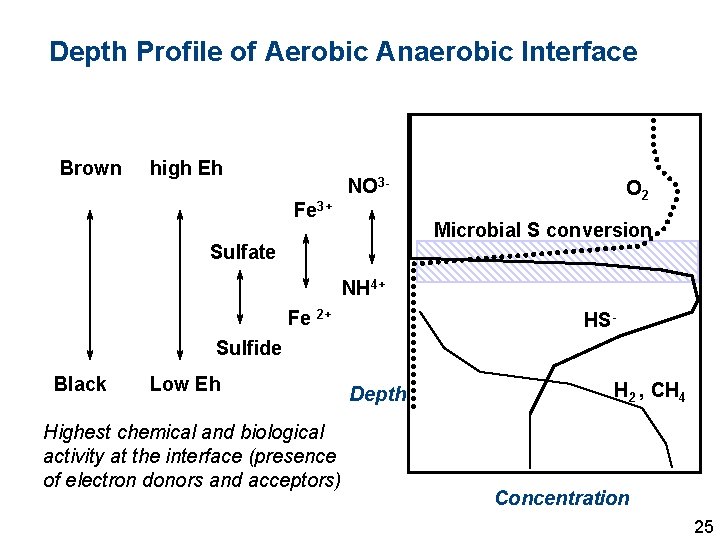
Depth Profile of Aerobic Anaerobic Interface Brown high Eh NO 3 Fe 3+ O 2 Microbial S conversion Sulfate NH 4+ Fe 2+ HS- Sulfide Black Low Eh Highest chemical and biological activity at the interface (presence of electron donors and acceptors) Depth H 2 , CH 4 Concentration 25
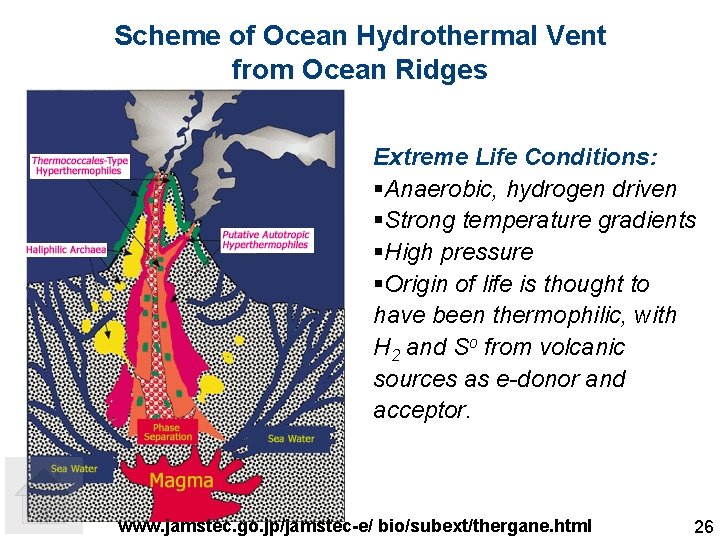
Scheme of Ocean Hydrothermal Vent from Ocean Ridges Extreme Life Conditions: §Anaerobic, hydrogen driven §Strong temperature gradients §High pressure §Origin of life is thought to have been thermophilic, with H 2 and So from volcanic sources as e-donor and acceptor. www. jamstec. go. jp/jamstec-e/ bio/subext/thergane. html 26
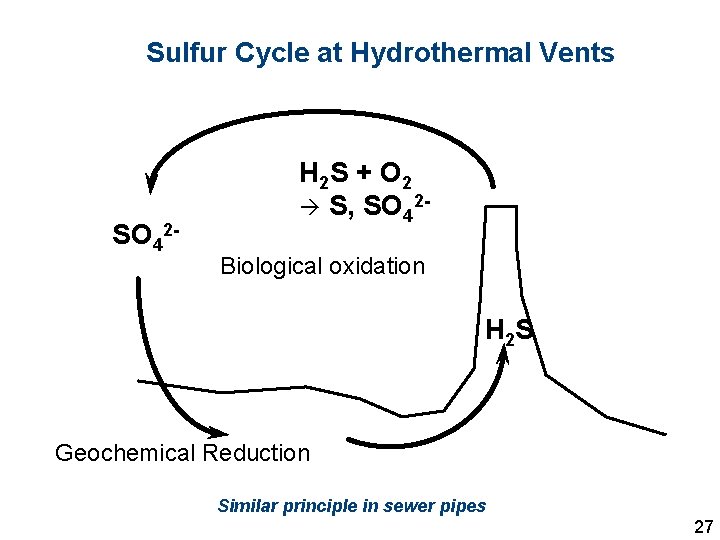
Sulfur Cycle at Hydrothermal Vents SO 42 - H 2 S + O 2 S, SO 42 Biological oxidation H 2 S Geochemical Reduction Similar principle in sewer pipes 27

“Black Smokers” releasing reduced sufur and iron (e. g. Fe. S) as potential electron donors for bacteria. 28

• S oxidising Bacteria as primary producers White “snowblower” producing suspended sulfur bacteria in snow flake type aggregates during a volcanic eruption Woods Hole Oceanographic Institute East Pacific Rise. 29

Tubeworms (Riftia) living in association with sulfur oxidising bacteria 30

Tubeworms (Riftia) living in association with sulfur oxidising bacteria Micheal Degruy Dark food-chain Independent of Sunlight ? 31

Deep-sea mussel Bathymodiolus thermophilus using symbiotic sulfur bacteria. Photo by Richard A. Lutz 32

Galatheid crabs lining a fissure at a hot vent on the East Pacific Rise feeding on sulfur bacteria. Courtesy Woods Hole Oceanographic Institution 33

Tubeworms (Riftia) living in association with sulfur oxidising bacteria 34

Anaerobic Oxidation of Sulfide There are principally two conditions allowing sulfur cycle in the absence of oxygen: 1. Presence of light and phototrophic bacteria: Very colorful, play a role in microbial mats Can use light that is not suitable for algae Green sulfur bacteria (S outside, Chlorobium) Purple sulfur bacteria (S inside, Chromatium) 2. Presence of other “powerful e-acceptors (e. g. nitrate, Fe 3+) are available Thiomargareta a recent discovery 35
Thiomargareta namibiensis 36

Thiomargareta namibiensis 37

Aerobic Re-oxidation Processes 2 - Methane § CH 4 is a highly energetic e-donor (fuel) in aerobic areas § With electron acceptors O 2, Nitrate, Fe 3+ methane can be re-oxidized by methylotrophic bacteria Recently evidence has been found of CH 4 oxidation linked to sulfate reduction. §Those electron acceptors are usually made available by bioturbation, thus CH 4 usually does not reach the water column §Where benthic macrofauna has been killed methane production forms large CH 4 bubbles that can escape the water column (does not occur in the open ocean) Under pressure, methane forms hydrates on the ocean floor around continental shelfes. These hydrates can be used as electron donors for aerobic bacteria food chain. 38

Gas Hydrate Molecules (dusk. geo. orst. edu/ oceans/lec 14. html) 39

Methane hydrate mount under flashlight 40

Methane hydrate outcrop from continental shelf. approximately 250 miles east of Charleston, S. C courtesy Carolin Ruppel 41

“Chemosynthetic mussel from methane hydrate Tubeworms (Riftia) living in association with sulfur oxidising bacteria Tube worms Methane hydrate outcrop Micheal Degruy 42

Methane hydrate sample 43

“Chemosynthetic mussel from methane hydrate Tubeworms (Riftia) living in association with sulfur oxidising bacteria Tubeworm collected from gas hydrate seepage areas Micheal Degruy 44

Tubeworms (Riftia) living in Spider crab looking for food between the tubeworms growing on association with sulfur a methane hydrate outcrop oxidising bacteria Micheal Degruy 45

“Chemosynthetic mussel from methane hydrate Tubeworms (Riftia) living in association with sulfur oxidising bacteria Mussels with bacterial slime living on methane hydrate Micheal Degruy 46
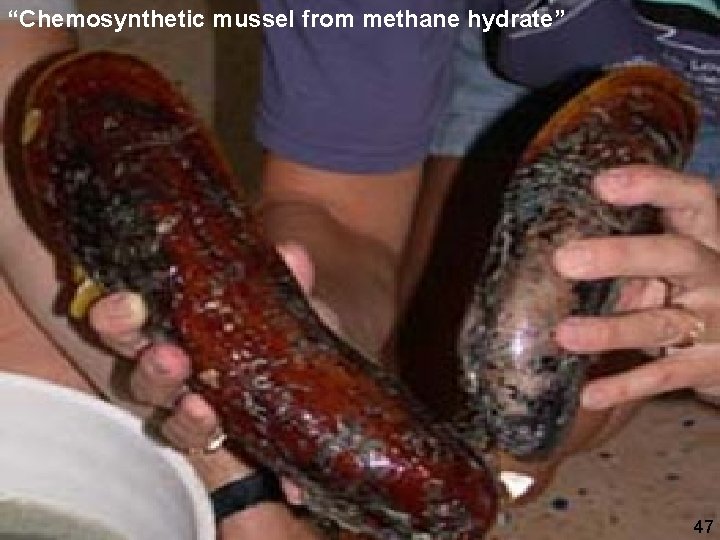
“Chemosynthetic mussel from methane hydrate” Tubeworms (Riftia) living in association with sulfur oxidising bacteria Micheal Degruy 47
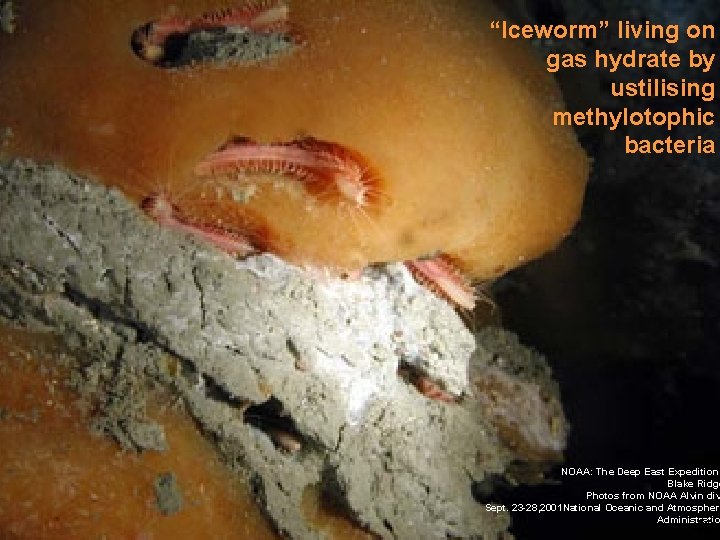
“Chemosynthetic mussel from methane hydrate “Iceworm” living on Tubeworms (Riftia) living in gas hydrate by association with sulfur ustilising oxidising bacteria methylotophic bacteria Micheal Degruy NOAA: The Deep East Expedition Blake Ridge Photos from NOAA Alvin div Sept. 23 -28, 2001 National Oceanic and Atmospheri Administratio 48

“Chemosynthetic mussel from methane hydrate Tubeworms (Riftia) living in association with sulfur oxidising bacteria Methane hydrate with ice worms Micheal Degruy 49

“Chemosynthetic mussel from methane hydrate Tubeworms (Riftia) living in association with sulfur oxidising bacteria Micheal Degruy 50

Gas Hydrate at the Surface Under Atmospheric Conditions: gas hydrate separates into flammable CH 4 and water (here cooling hands) 51
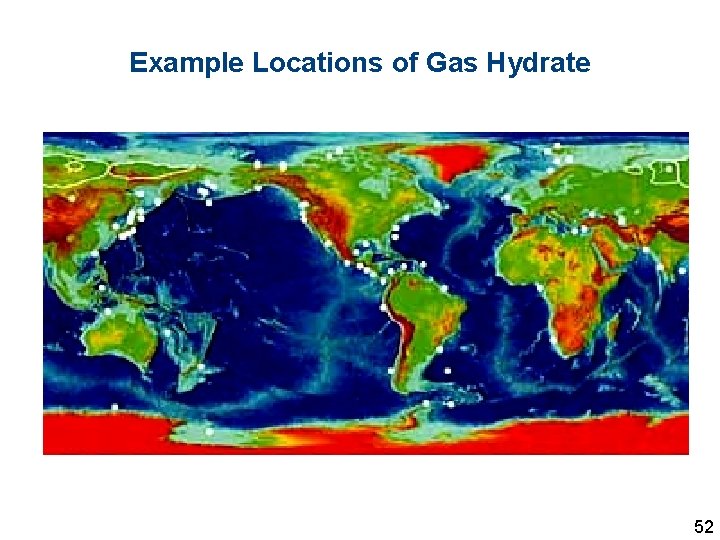
Example Locations of Gas Hydrate 52

53

Methane Hydrate (Summary) § Methane hydrate, a curiosity or a significant global phenomenon? § Needed formation: low temperature and high pressure § Why are hydrates mainly on the continental shelfes ? § Deep oceans lack organic material § High biologic productivity (CH 4) § Rapid sedimentation rates (bury the organic matter) 54

Aspects of microbial sulfide oxidation § Sulfide oxidation § Needed formation: low temperature and high pressure § Why are hydrates mainly on the continental shelfes ? § Deep oceans lack organic material § High biologic productivity (CH 4) § Rapid sedimentation rates (bury the organic matter) 55
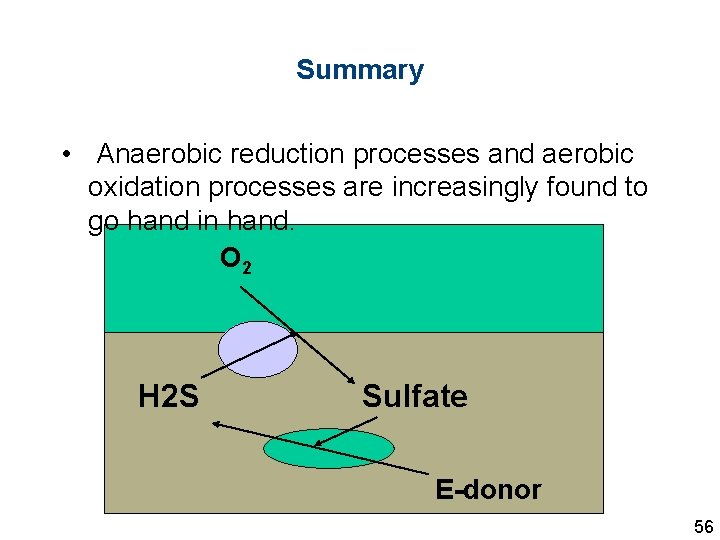
Summary • Anaerobic reduction processes and aerobic oxidation processes are increasingly found to go hand in hand. O 2 H 2 S Sulfate E-donor 56
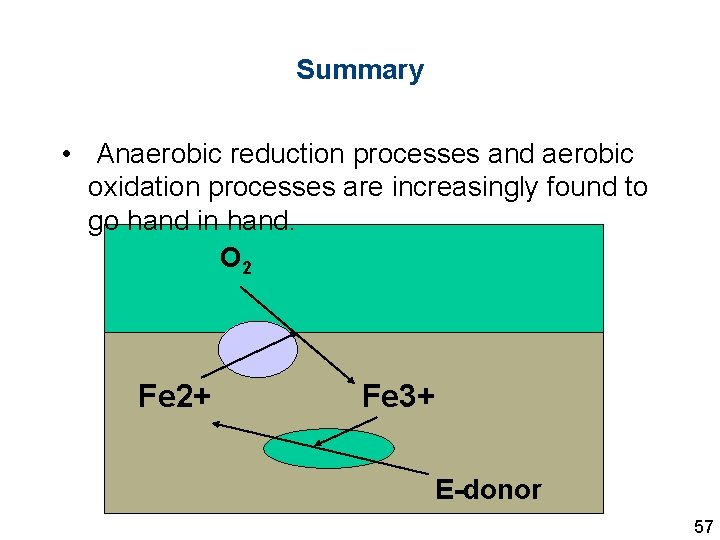
Summary • Anaerobic reduction processes and aerobic oxidation processes are increasingly found to go hand in hand. O 2 Fe 2+ Fe 3+ E-donor 57
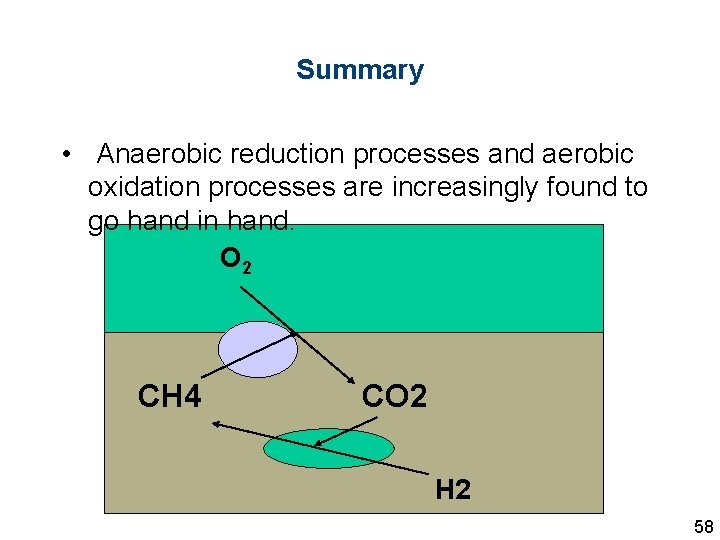
Summary • Anaerobic reduction processes and aerobic oxidation processes are increasingly found to go hand in hand. O 2 CH 4 CO 2 H 2 58
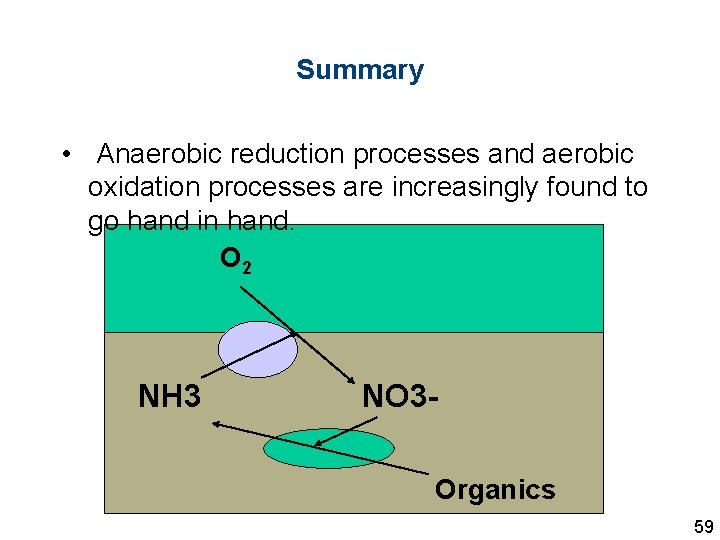
Summary • Anaerobic reduction processes and aerobic oxidation processes are increasingly found to go hand in hand. O 2 NH 3 NO 3 Organics 59

Iron reducing bacteria flowing electrons to ferric iron • Organisms: Geobacter and other anaerobic bacteria, no specific group • E- donors: mainly small organic compounds • Typical reaction: H 2 + 2 Fe 3+ --> 2 Fe 2+ + 2 H+ • Reaction results in lowering of redox potential. • Reduce also Manganese, elemental sulfur and other metals (e. g. . . uranium). • Endproduct is magnetite (Fe 3 O 4) and other compounds (black precipitates) • Significance of iron reduction is still being underestimated. • Recent research: electricity production using ferric iron reducing bacteria 60

Magnetite formation as the endproduct of iron reduction 61

How do IRB transfer electrons to insoluble iron ? ? Ideas: • solubilise iron (complexing, acid dissolving, etc. ) • physically attach to ferric iron • excrete electron shuttling species (mediators) use e-carriers present in environment humic acids (quinone, analogue to NADH) 62
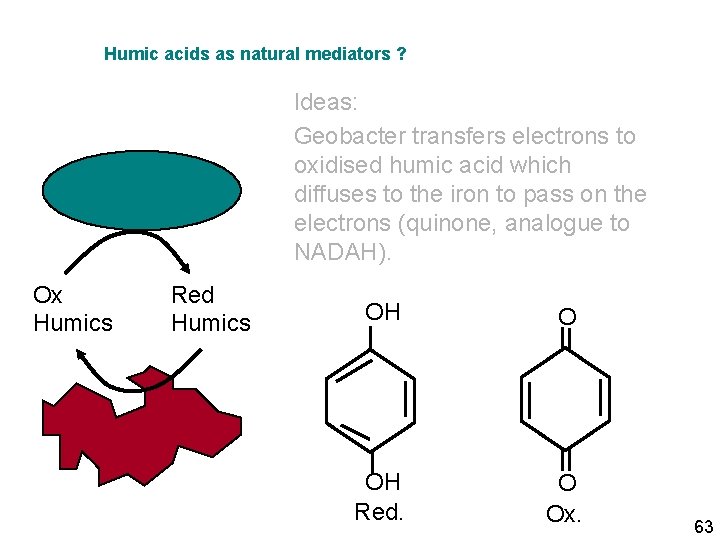
Humic acids as natural mediators ? Ideas: Geobacter transfers electrons to oxidised humic acid which diffuses to the iron to pass on the electrons (quinone, analogue to NADAH). Ox Humics Red Humics OH O OH Red. O Ox. 63
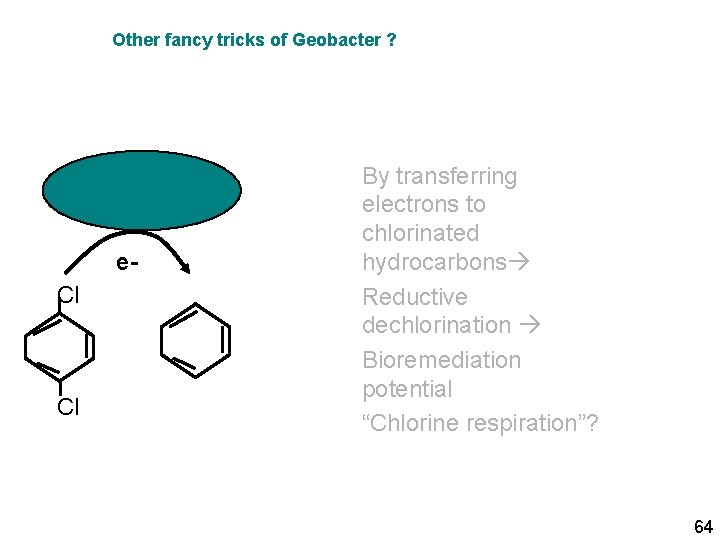
Other fancy tricks of Geobacter ? e. Cl Cl By transferring electrons to chlorinated hydrocarbons Reductive dechlorination Bioremediation potential “Chlorine respiration”? 64
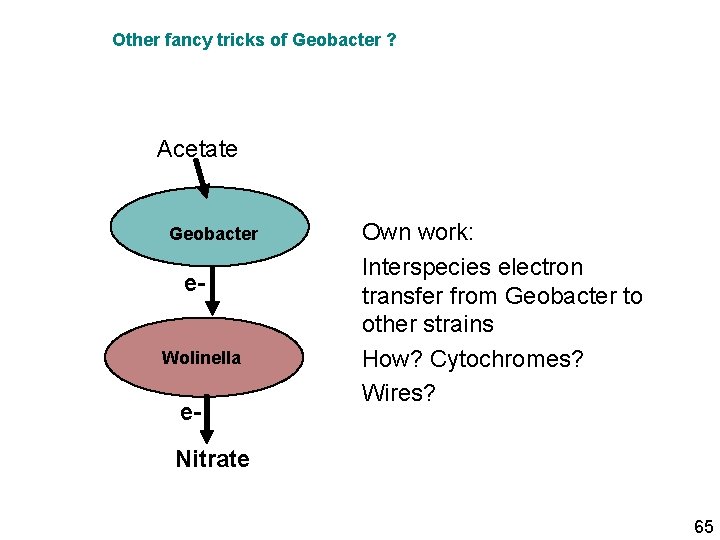
Other fancy tricks of Geobacter ? Acetate Geobacter e. Wolinella e- Own work: Interspecies electron transfer from Geobacter to other strains How? Cytochromes? Wires? Nitrate 65

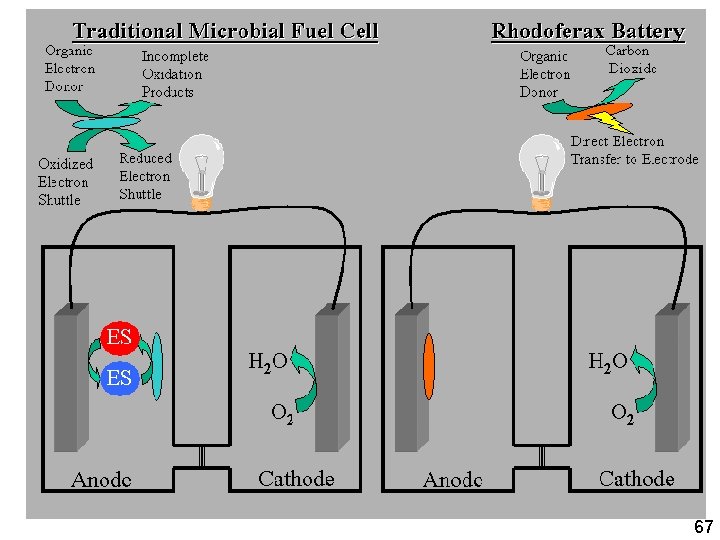
Geobacter Headlines (Geobacter. com) : Bioelectricity If Geobacter can transfer electrons to almost anything, why not to a carbon anode. Electricity generation by Geobacter Outback batteries Driving force: Organic wastes Key: Sugar degrading Geobacter type (Rhodoferax) 66
67

68

Simple approach to bio -electricity 69

Geobacter Headlines 2: Cleanup of Uranium Geobacters capability of reducing other metal species includes uranium. By reducing U(VI) to U(IV) which is less soluble limitation of contamination has been applied in situ (2003) Tod Anderson 70

Geobacter Headlines 3: Hottest Bug Strain 121 Geobacters presence of deep ocean vents (black smokers) has been shown. Temperature tolerance to 121 (autoclave) Interesting Genome. Suggestions of iron reducing Archeae to be one of the oldest lifeforms rather then sulfur reducers. Very old magnetite formations are seen to support this view. 71
Strain 121 72

Lecture Summary 1. Sulfate reduction § P- release from sediments, Sulfur deposits, Corrosion of submerged steel, Lethal gas emissions 2. NO 3 - reduction to N 2 § black precipitate, magnetite (Fe 3 O 4), is produced when Fe 3+ is reduced to Fe 2+ 3. CO 2 reduction to CH 4 § Methane - highly energetic e-donor (fuel) in aerobic areas 4. Prolific life at the anaerobic/aerobic interface § High activity at chemocline, Black smokers, Life on CH 4 73

Lecture Summary 1. Sulfate reduction § P- release from sediments, Sulfur deposits, Corrosion of submerged steel, Lethal gas emissions 2. Fe 3+ reduction to Fe 2+ § black precipitate, magnetite (Fe 3 O 4), is produced when Fe 3+ is reduced to Fe 2+ 3. CO 2 reduction to CH 4 § Methane - highly energetic e-donor (fuel) in aerobic areas 4. Prolific life at the anaerobic/aerobic interface § High activity at chemocline, Black smokers, Life on CH 4 74

End of lecture, below only for personal interest 75

Methane hydrate • Methane hydrate, a curiosity or a significant global phenomenon? • Needed formation: low temperature and high pressure • Why are hydrates mainly on the continental shelfes ? • Deep oceans lack • high biologic productivity (CH 4) • rapid sedimentation rates (bury the organic matter) 76
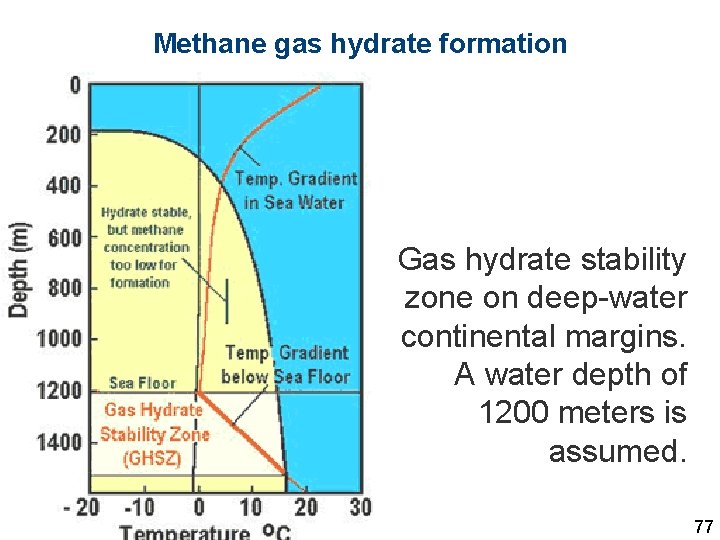
Methane gas hydrate formation Gas hydrate stability zone on deep-water continental margins. A water depth of 1200 meters is assumed. 77
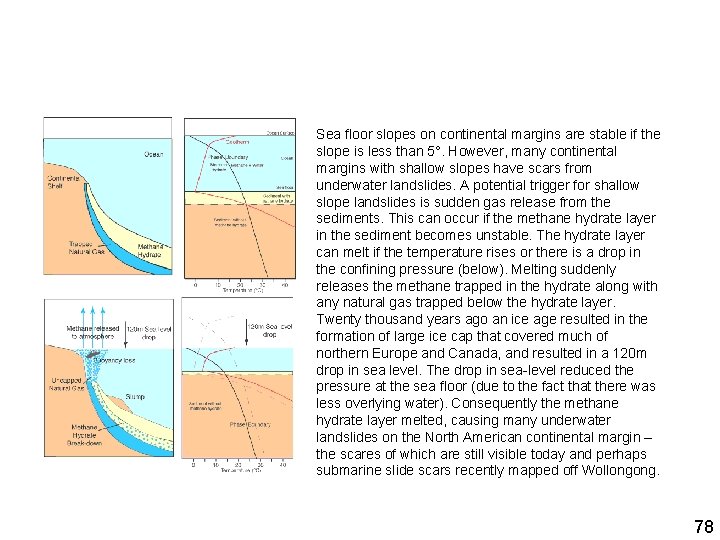
Sea floor slopes on continental margins are stable if the slope is less than 5°. However, many continental margins with shallow slopes have scars from underwater landslides. A potential trigger for shallow slope landslides is sudden gas release from the sediments. This can occur if the methane hydrate layer in the sediment becomes unstable. The hydrate layer can melt if the temperature rises or there is a drop in the confining pressure (below). Melting suddenly releases the methane trapped in the hydrate along with any natural gas trapped below the hydrate layer. Twenty thousand years ago an ice age resulted in the formation of large ice cap that covered much of northern Europe and Canada, and resulted in a 120 m drop in sea level. The drop in sea-level reduced the pressure at the sea floor (due to the fact that there was less overlying water). Consequently the methane hydrate layer melted, causing many underwater landslides on the North American continental margin – the scares of which are still visible today and perhaps submarine slide scars recently mapped off Wollongong. 78

Gas hydrates and bubbles in the bermuda triangle ^ A drop in sea-level reduces the pressure at the sea floor and causes the melting of methane hydrate. The sudden release of gas results in landslides and slumps. It can also result in a plume of gas rapidly rising to the ocean surface. Gas in the water reduces the density of water leading to the loss of buoyancy of ocean going craft. Is this what causes the mysterious sinking of ships in the Bermuda Triangle? When sea level dropped during the last ice age, the destabilisation of hydrate and the release of methane may have been sufficient to heat the atmosphere via greenhouse effects and turn back the ice age. At atmospheric pressure the concentration of methane in hydrate is over 600 times greater than in the free gas form. Methane hydrate is also significantly denser than liquid natural gas. Methane hydrate may provide a cost effective way of transporting and storing methane. 79
- Slides: 79