Micro RNAs in Ex Vivo Stored Blood Cells

Micro. RNAs in Ex Vivo Stored Blood Cells: What Future Do They Hold in Transfusion Medicine? C. D. Atreya, Ph. D. Associate Director for Research Office of Blood Research and Review Center for Biologics Evaluation and Research US Food and Drug Administration US Department of Health and Human Services 5 th World Hematologists Congress, Aug 18 -19, 2016 ***This presentation reflects the views of Dr. C. D. Atreya and should not be construed to represent FDA’s views or policies***

Ø Micro. RNAs Small Single-stranded noncoding regulatory RNAs which represent ~4% of the genes in the human genome Ø Ex Vivo Stored Blood Cells Platelets and RBCs collected, processed, and stored in bags as PC and p. RBC units available for transfusion Ø Transfusion Medicine A branch of medicine that is concerned with safe and effective transfusion of blood and blood components (e. g. Platelets and RBC)

Topics covered…. Ø mi. RNAs –Biogenesis and mechanism of action Ø Changes associated with ex vivo Storage of blood Cells (PCs and p. RBC) Ø Roll of mi. RNAs in stored blood cells Ø Extracellular release of mi. RNAs in the context of ex vivo stored blood cells Ø Conclusions

Ø mi. RNAs –Biogenesis and mechanism of action Ø Changes associated with ex vivo Storage of blood Cells (PCs and p. RBC) Ø Roll of mi. RNAs in stored blood cells Ø Extracellular release of mi. RNAs in the context of ex vivo stored blood cells Ø Conclusions
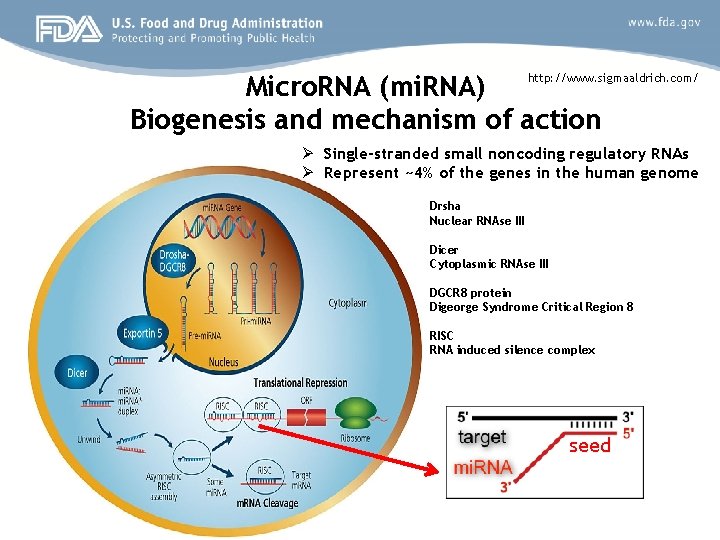
Micro. RNA (mi. RNA) http: //www. sigmaaldrich. com/ Biogenesis and mechanism of action Ø Single-stranded small noncoding regulatory RNAs Ø Represent ~4% of the genes in the human genome Drsha Nuclear RNAse III Dicer Cytoplasmic RNAse III DGCR 8 protein Digeorge Syndrome Critical Region 8 RISC RNA induced silence complex seed

Single mi. RNA can control multiple m. RNAs

Multiple mi. RNAs can effectively control a single m. RNA by targeting at different sites

Ø mi. RNAs –Biogenesis and mechanism of action Ø Changes associated with ex vivo Storage of blood Cells (PCs and p. RBC) Ø Roll of mi. RNAs in stored blood cells Ø Extracellular release of mi. RNAs in the context of ex vivo stored blood cells Ø Conclusions
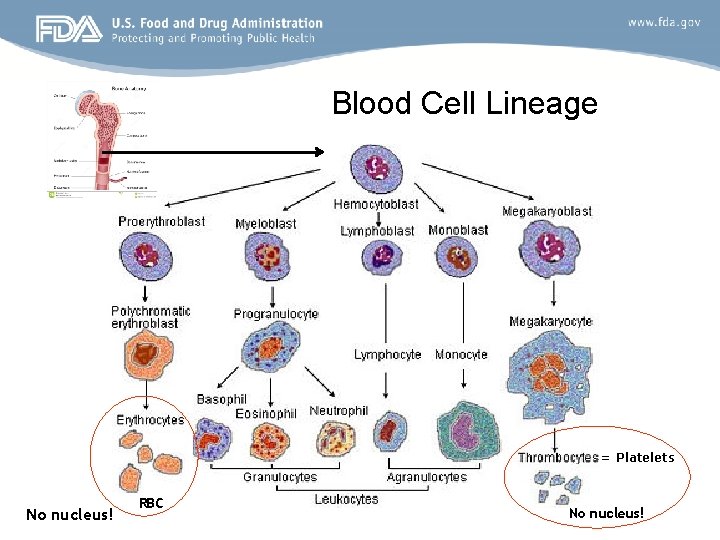
Blood Cell Lineage = Platelets No nucleus! RBC No nucleus!
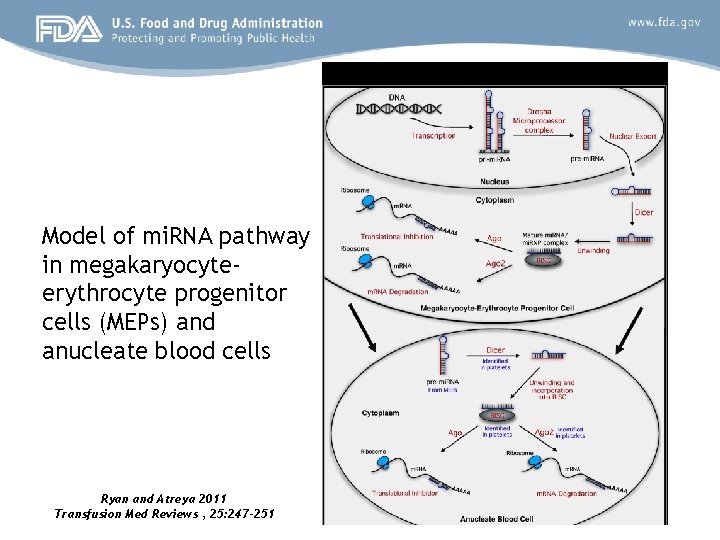
Model of mi. RNA pathway in megakaryocyteerythrocyte progenitor cells (MEPs) and anucleate blood cells Ryan and Atreya 2011 Transfusion Med Reviews , 25: 247 -251
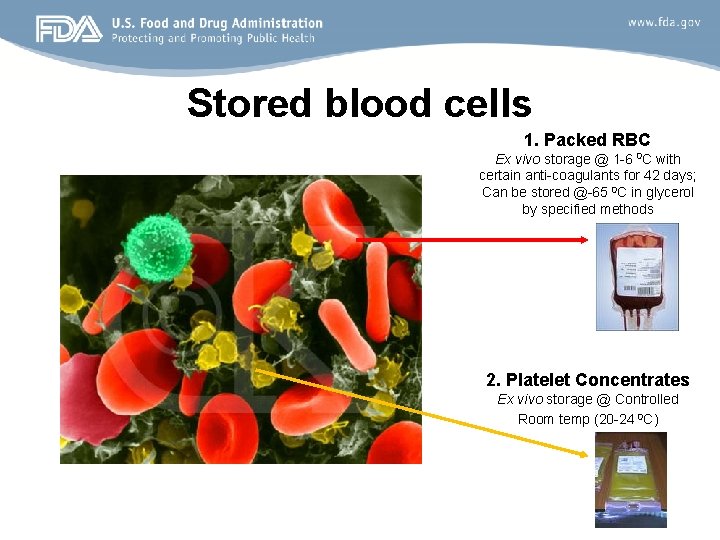
Stored blood cells 1. Packed RBC Ex vivo storage @ 1 -6 0 C with certain anti-coagulants for 42 days; Can be stored @-65 0 C in glycerol by specified methods 2. Platelet Concentrates Ex vivo storage @ Controlled Room temp (20 -24 0 C)

RBC ex vivo storage Ø Currently licensed RBC additive solutions (AS) have acidic p. H (~5. 5 -5. 8) Ø The acidic intracellular environment rapidly decreases ATP (necessary for RBC survival) and 2, 3 -DPG (necessary for oxygen delivery) and triggers apoptosislike processes (eryptosis) Ø Research on RBC storage has repeatedly demonstrated that fundamental biology of RBC is still not well understood Sparrow, Blood Transfusion 2012

RBC Storage Lesion (SL) “Biochemical, morphological and immunologic changes which occur within RBC and in the associated storage media during ex vivo preservation of RBC are collectively known as RBC storage lesion” Biochemical: rapid depletion of 2, 3 -diphosphoglycerate (DPG) and Adenosine triphosphate (ATP) levels in RBC Morphological: Change in corpuscle shape affecting microcirculation in the recipients Immunological: Immuno-modulatory effects in the recipients
Why Study RBC-SL? Ex Vivo Stored RBC with SL Clinical consequences Biochemical morphological immunologic changes Acute lung injury Multi-organ failure Mortality Modified Fig. 1 from Kor et al, BJBMC 2009

Human RBC What we know 1. Mature RBC is enucleated 2. During ex vivo storage, RBC undergo changes (SL) 3. RBC-SL has serious clinical consequences What we didn’t know 1. How are the cellular processes regulated in enucleated RBC, especially in RBC-SL? 2. Are there other ‘command & controls’ to compensate for the loss of nucleus in mature RBC? 2. Can we manipulate these controls to improve the quality and shelf-life of RBC in storage?

Topics covered…. Ø mi. RNAs –Biogenesis and mechanism of action Ø Changes associated with ex vivo Storage of blood Cells (PCs and p. RBC) Ø Roll of mi. RNAs in stored blood cells Ø Extracellular release of mi. RNAs in the context of ex vivo stored blood cells Ø Conclusions

Recent research advances in RBC and platelets Ø Enucleated mature RBC and platelets have m. RNAs, splicing factors etc. in addition to proteins (Denis et al, 2005) Ø Seminal observations are, RBC and platelets have abundant and diverse array of micro. RNAs (Landry et al, 2009; Chen et al, 2009; Kannan et al 2009; Kannan and Atreya, 2010; Sarachana et al 2015; Dahiya et al 2016) Ø Micro. RNAs (mi. RNAs) are established as the key cellular negative regulators of genes (m. RNAs) in eukaryotic cells Ø In enucleated blood cells by far the mi. RNA is the only major regulatory nucleic acid identified that can regulate cellular processes! Ø Therefore, understanding the blood cell SL processes through mi. RNA regulation in ex vivo storage will provide clues towards enhancing the quality and perhaps shelf-life of these cells during storage

Understanding the regulatory mechanisms of RBC storage lesion Some of the measurable parameters during storage ØRBC shape change (affects microcirculation) as measured by Mean Corpuscular Volume (MCV) ØCellular ATP loss ØApoptosis-like (eryptosis) symptoms

mi. RNA analysis in stored blood cells Study design and methods • Leukocyte-reduced p. RBC units collected from 11 donors were stored under conventional blood bank conditions • Samples were collected from each bag at day 0, 7, 14, 28, 42, and 56 and mature RBCs were enriched and isolated • Three known RBC-SL parameters were chosen for the study [Mean Corpuscular Volume (MCV), eryptosis and ATP loss] • Purified RNA from each mature RBC sample was subjected to highthroughput mi. RNA microarray* differential expression profiling and the data was subjected to various bioinformatics programs *Affymetrix Gene. Chip mi. RNA 3. 0 microarrays representing 19, 724 probes covering over 5, 600 mi. RNAs, pre-mi. RNAs, sno. RNAs etc.
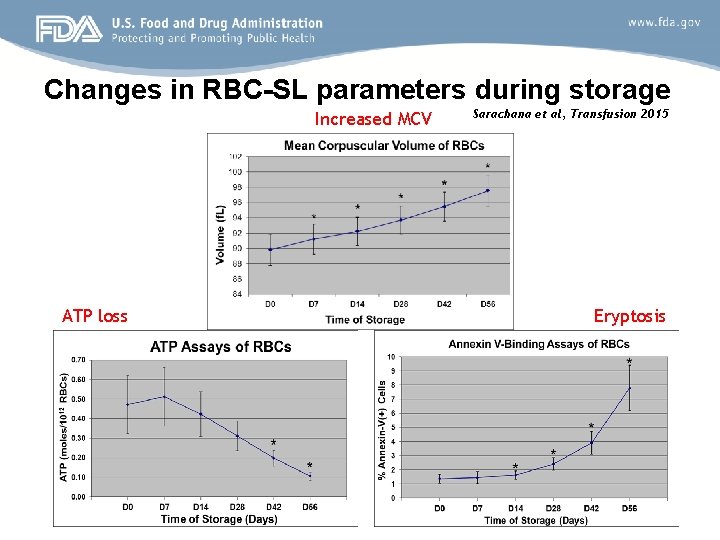
Changes in RBC-SL parameters during storage Increased MCV ATP loss Sarachana et al, Transfusion 2015 Eryptosis
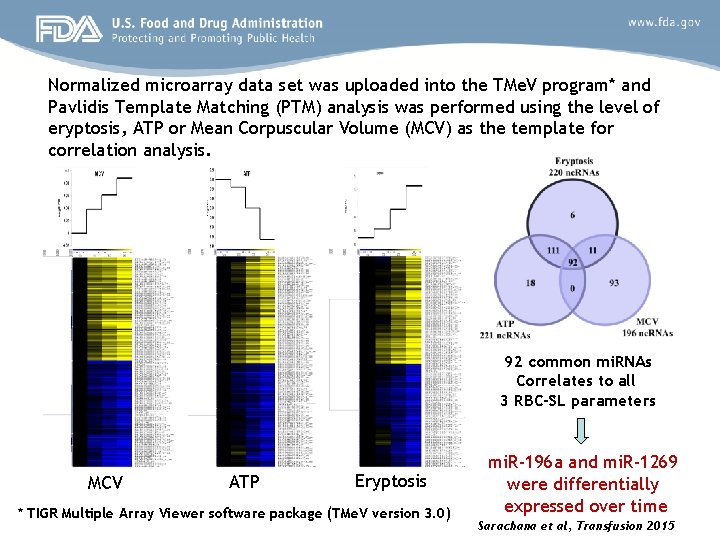
Normalized microarray data set was uploaded into the TMe. V program* and Pavlidis Template Matching (PTM) analysis was performed using the level of eryptosis, ATP or Mean Corpuscular Volume (MCV) as the template for correlation analysis. 92 common mi. RNAs Correlates to all 3 RBC-SL parameters MCV ATP Eryptosis * TIGR Multiple Array Viewer software package (TMe. V version 3. 0) mi. R-196 a and mi. R-1269 were differentially expressed over time Sarachana et al, Transfusion 2015

Top two cellular functions associated with potential targets of mi. R-196 a and mi. R-1269 were identified Identified Function P-value No. of genes (m. RNAs) Cell morphology Fisher’s exact test, <0. 05 74 Cell death and survival Fisher’s exact test, <0. 05 97 These data were obtained using Ingenuity Pathway Analysis (IPA) software. P-values were calculated using Fisher’s exact test, which was performed using the entire set of genes within the Ingenuity Knowledge Base as the reference set. We selected mi. R-196 a and evaluated its effects on cell death and/or survival Sarachana et al, Transfusion 2015
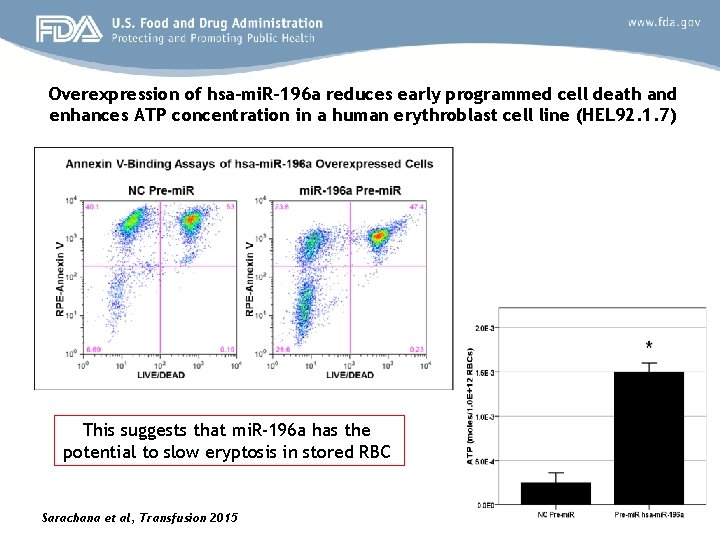
Overexpression of hsa-mi. R-196 a reduces early programmed cell death and enhances ATP concentration in a human erythroblast cell line (HEL 92. 1. 7) This suggests that mi. R-196 a has the potential to slow eryptosis in stored RBC Sarachana et al, Transfusion 2015

mi. RNA analysis in stored blood cells Summary Ø Our analysis identified two RNAs (mi. R-196 a and mi. R-1269) whose changes in the expression levels were correlated with the selected three SL parameters that we evaluated. Ø Overexpression of one such mi. RNA, the mi. R-196 a, in a human erythroblast cell line (HEL 97. 1. 2) confirmed its protective effects against cell death and ATP loss. In support of our concept that mi. RNAs can protect RBC from eryptosis, Yu et al, 2010 have demonstrated that mi. R-451 protects erythrocytes against oxidant stress (Yu et al, Genes and Development, 2010, 24: 1620 -33)
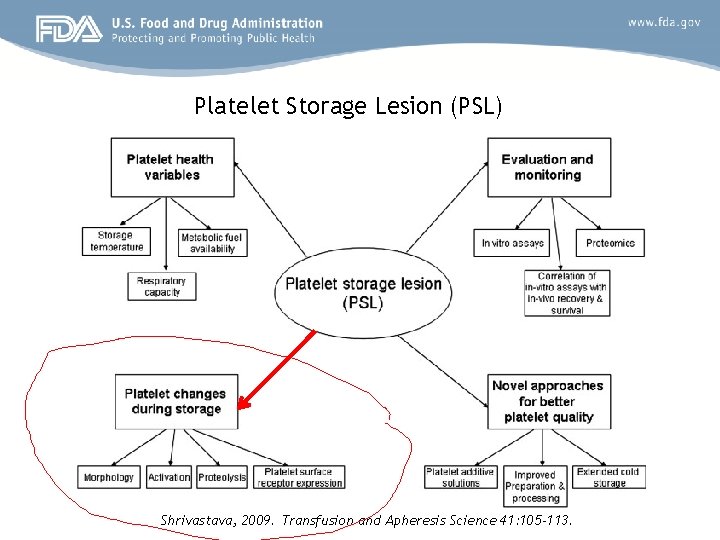
Platelet Storage Lesion (PSL) Shrivastava, 2009. Transfusion and Apheresis Science 41: 105 -113.
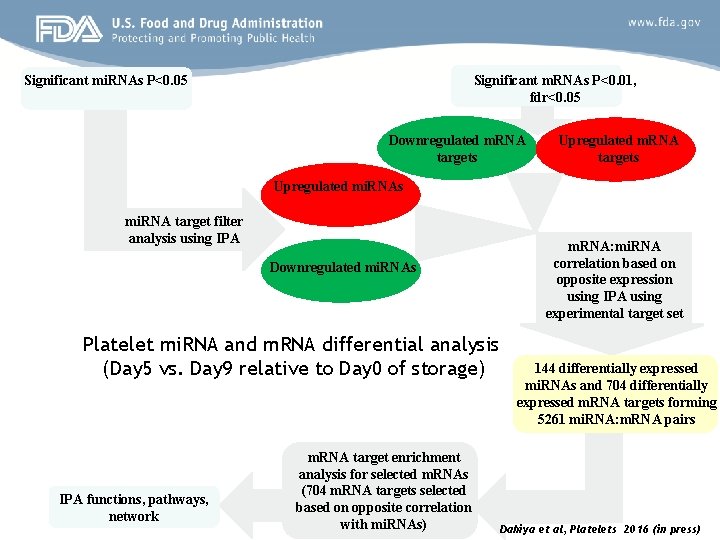
Significant m. RNAs P<0. 01, fdr<0. 05 Significant mi. RNAs P<0. 05 Downregulated m. RNA targets Upregulated mi. RNAs mi. RNA target filter analysis using IPA Downregulated mi. RNAs Platelet mi. RNA and m. RNA differential analysis (Day 5 vs. Day 9 relative to Day 0 of storage) IPA functions, pathways, network m. RNA target enrichment analysis for selected m. RNAs (704 m. RNA targets selected based on opposite correlation with mi. RNAs) m. RNA: mi. RNA correlation based on opposite expression using IPA using experimental target set 144 differentially expressed mi. RNAs and 704 differentially expressed m. RNA targets forming 5261 mi. RNA: m. RNA pairs Dahiya et al, Platelets 2016 (in press)
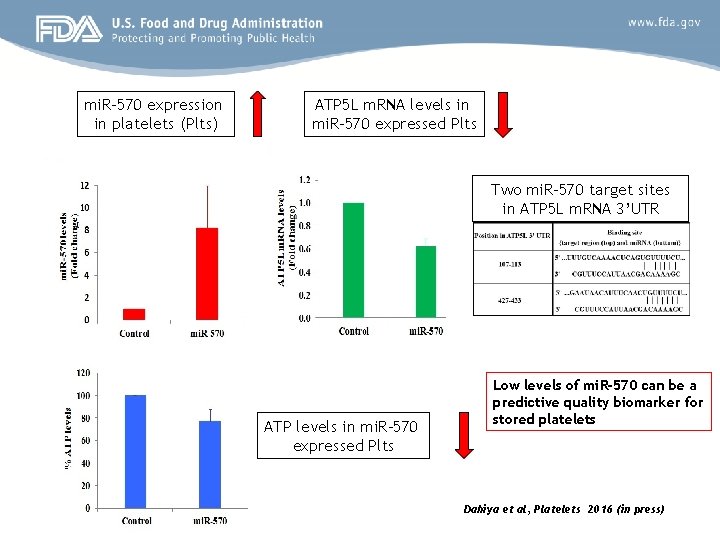
mi. R-570 expression in platelets (Plts) ATP 5 L m. RNA levels in mi. R-570 expressed Plts Two mi. R-570 target sites in ATP 5 L m. RNA 3’UTR ATP levels in mi. R-570 expressed Plts Low levels of mi. R-570 can be a predictive quality biomarker for stored platelets Dahiya et al, Platelets 2016 (in press)

Another example of mi. RNAs in stored platelets Our mi. RNA analysis (Day 0 -Day 9 of storage) identified Ras-related protein 1 (Rap 1) signaling pathway as one of the potential mi. RNA targets in stored platelets It is known that during platelet storage Rap 1 increases and triggers platelet activation as monitored by Flow Cytometry using the activation marker CD 62 P (Shubert et al, Transfusion 2009) Testable Hypothesis: RAP 1 (m. RNA) down-regulation by mi. RNAs should suppress platelet activation during storage and provide opportunities to improve platelet quality in storage Dahiya et al, Unpublished data
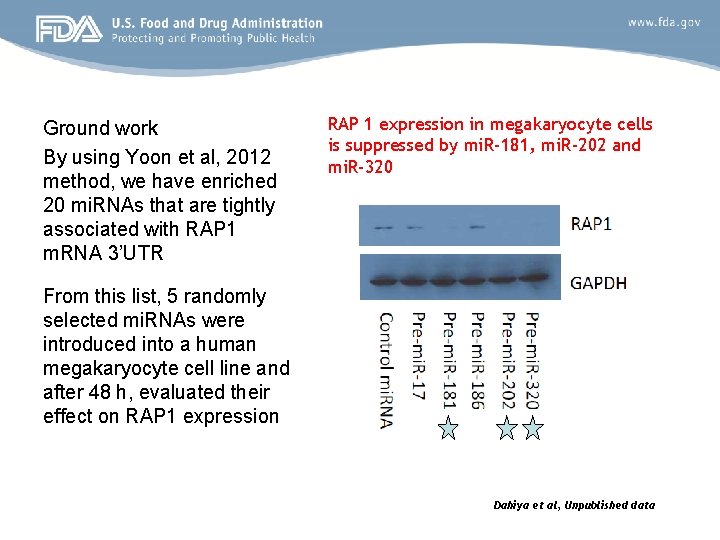
Ground work By using Yoon et al, 2012 method, we have enriched 20 mi. RNAs that are tightly associated with RAP 1 m. RNA 3’UTR RAP 1 expression in megakaryocyte cells is suppressed by mi. R-181, mi. R-202 and mi. R-320 From this list, 5 randomly selected mi. RNAs were introduced into a human megakaryocyte cell line and after 48 h, evaluated their effect on RAP 1 expression Dahiya et al, Unpublished data

Topics covered…. Ø mi. RNAs –Biogenesis and mechanism of action Ø Changes associated with ex vivo Storage of blood Cells (PCs and p. RBC) Ø Roll of mi. RNAs in stored blood cells Ø Extracellular release of mi. RNAs in the context of ex vivo stored blood cells Ø Conclusions
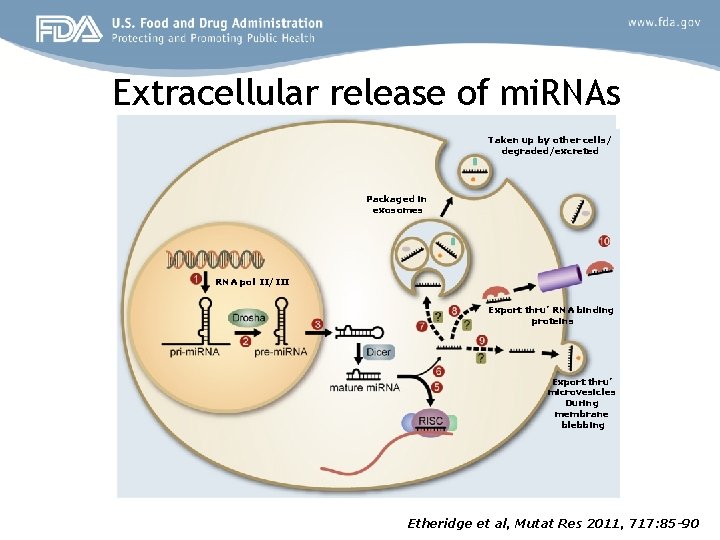
Extracellular release of mi. RNAs Taken up by other cells/ degraded/excreted Packaged in exosomes RNA pol II/III Export thru’ RNA binding proteins Export thru’ microvesicles During membrane blebbing Etheridge et al, Mutat Res 2011, 717: 85 -90

Mechanism of mi. RNA release from cells in the peripheral blood (Ago 2=Argonaute 2; NPM 1=nucleophosmin 1; HDL=high density lipoproteins) Stefano et al, Vascul Pharmacol 2011 Oct; 55(4): 111 -8. Epub 2011 Aug 6.

Ø Platelet MP-derived mi. R-223 can regulate human umbilical vein endothelial cells (HUVEC) gene expression at the protein level Ø Regulates FBXW 7 (a F-Box protein, subunit of ubiquitin protein) Ø Regulates EFNA 1 protein, a member of ephrin family of proteins Ø Platelet MPs (containing mi. Rs, e. g. mi. R-126 -3 p) reprogram macrophage gene expression and function Ø Platelet MPs induced up-regulation of 34 mi. Rs and downregulation of 367 m. RNAs Laffont et al, Blood 2013, 122: 253 -261; Thrombosis and Haemostasis 2016, 115: 311 -323.

Topics covered…. Ø mi. RNAs –Biogenesis and mechanism of action Ø Changes associated with ex vivo Storage of blood Cells (PCs and p. RBC) Ø mi. RNAs’ Roll in stored blood cells Ø Extracellular release of mi. RNAs in the context of ex vivo stored blood cells Ø Conclusions

Micro. RNAs in Stored Blood Cells: What Future Do They Hold in Transfusion Medicine? Ø mi. RNAs regulate physiological events of mature (enucleated) blood cells Ø reversing this regulation may help enhance their quality and shelf-life during storage Ø some of the mi. RNAs could serve as quality biomarkers of storage Ø Enhancing the quality and shelf-life of stored blood cells will increase inter-donation intervals for repeat donors, especially for women whose iron deficiencies can be mitigated by long interdonation intervals! Ø Blood cell-derived MP mi. RNAs can regulate gene expression of other cells Ø Can transfusion of ex vivo stored blood cells with mi. RNA containing MPs develop measurable transient alteration of gene expression in other cells of recipients?

Enhancing the quality and shelf-life of stored blood cells will increase inter-donation intervals for repeat donors, especially for women whose iron deficiencies can be mitigated by long inter-donation intervals!

Advancing innovation is fundamental to US FDA’s core mission of protecting and promoting the public health -FDA’s Regulatory Science Priorities (2014 -2018) Thank you!
- Slides: 37